Topic: 23.1
(a) (i) Describe the trend in the solubility of the hydroxides of magnesium, calcium and strontium. Explain your answer.

(ii) Suggest the variation in pH of saturated solutions of the hydroxides of magnesium, calcium and strontium. Explain your answer.
(b) Barium hydroxide, Ba(OH)₂, is a strong base. A 250.0 cm³ solution of Ba(OH)₂ with a pH of 12.2 is made by dissolving Ba(OH)₂ in distilled water. Calculate the mass of Ba(OH)₂ required to make this solution. Show your working.
[\(M_r\): Ba(OH)₂, 171.3]
(c) The solubility of iron(II) hydroxide, Fe(OH)₂, is 5.85 × 10⁻⁶ moldm⁻³ at 298K.
(i) Write the expression for the solubility product, Ksp, of Fe(OH)₂.
(ii) Calculate the value of Ksp of Fe(OH)₂. Include its units.
▶️ Answer/Explanation
(a)(i) Strontium hydroxide > Calcium hydroxide > Magnesium hydroxide (most soluble to least soluble).
Explanation: Solubility of Group 2 hydroxides increases down the group due to decreasing lattice energy and increasing ionic size, making hydration more favorable.
(a)(ii) pH increases from magnesium hydroxide to strontium hydroxide.
Explanation: Higher solubility means more OH⁻ ions in solution, resulting in a higher pH for strontium hydroxide compared to magnesium hydroxide.
(b) Mass of Ba(OH)₂ = 0.0219 g.
Explanation:
- pH = 12.2 ⇒ pOH = 1.8 ⇒ [OH⁻] = 10⁻¹·⁸ = 0.0158 mol/dm³.
- Since Ba(OH)₂ dissociates as Ba(OH)₂ → Ba²⁺ + 2OH⁻, [Ba(OH)₂] = 0.0158/2 = 0.0079 mol/dm³.
- Moles in 250 cm³ = 0.0079 × 0.25 = 0.001975 mol.
- Mass = 0.001975 × 171.3 = 0.0219 g.
(c)(i) Ksp = [Fe²⁺][OH⁻]².
Explanation: The solubility product expression is derived from the dissociation equation Fe(OH)₂ ⇌ Fe²⁺ + 2OH⁻.
(c)(ii) Ksp = 8.00 × 10⁻¹⁶ mol³dm⁻⁹.
Explanation:
- [Fe²⁺] = 5.85 × 10⁻⁶ mol/dm³, [OH⁻] = 2 × 5.85 × 10⁻⁶ = 1.17 × 10⁻⁵ mol/dm³.
- Ksp = (5.85 × 10⁻⁶)(1.17 × 10⁻⁵)² = 8.00 × 10⁻¹⁶ mol³dm⁻⁹.

Topic: 28.2
(a) (i) Define transition element.
(ii) Explain why transition elements can form complex ions.
(b) The 3d orbitals in an isolated Ag⁺ ion are degenerate.
(i) Define degenerate d orbitals.
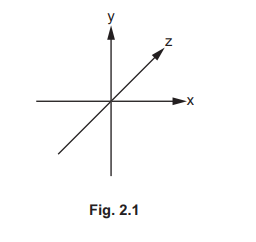
(ii) Sketch the shape of a 3dxy orbital in Fig. 2.1.
(c) Tollens’ reagent can be used to distinguish between aldehydes and ketones. Tollens’ reagent contains [Ag(NH₃)₂]OH, which can be prepared in a two-step process.
step 1 Aqueous NaOH is added dropwise to aqueous AgNO₃ to form Ag₂O as a brown precipitate.
step 2 Aqueous NH₃ is added dropwise to Ag₂O to form a colourless solution containing [Ag(NH₃)₂]OH.
Construct equations for each of the steps in the preparation of [Ag(NH₃)₂]OH.
(d) Name the shape of the complex ion \([Ag(NH_3)_2]^+\). State the bond angle for H-N-Ag and for N-Ag-N.
(e) An electrochemical cell uses Ag₂O as the positive electrode and Zn as the negative electrode immersed in an alkaline electrolyte.
The overall cell reaction is shown.
Ag₂O + Zn + H₂O → 2Ag + Zn(OH)₂
Complete the half-equation for the reaction at each electrode.
at the positive electrode Ag₂O + …………
at the negative electrode Zn + …………..
(f) Coordination polymers are made when a bidentate ligand acts as a bridge between different metal ions. Under certain conditions \(Ru^{3+}\)(aq) and the bidentate ligand dps can form a coordination polymer containing \(([Ru(dps)Cl_4]^–)_n\) chains.
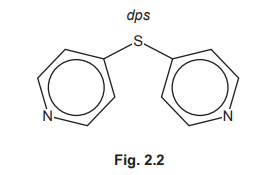
The bidentate ligand dps uses each of the nitrogen atoms to bond to a different \(Ru^{3+}\). Complete Fig. 2.3 by drawing the structure for the coordination polymer \(([Ru(dps)Cl_4]^–)_n\). Show two repeat units. The dps ligand can be represented using 
▶️ Answer/Explanation
(a)(i) A transition element is an element that forms one or more stable ions with incomplete d orbitals.
Explanation: Transition elements are characterized by their ability to form ions with partially filled d subshells, such as Fe²⁺ (3d⁶) or Cu²⁺ (3d⁹).
(a)(ii) Transition elements can form complex ions because they have vacant d orbitals that are energetically accessible for bonding with lone pairs from ligands.
Explanation: The presence of empty d orbitals allows transition metals to accept electron pairs from ligands, forming coordinate bonds in complex ions like \([Fe(CN)_6]^{4-}\).
(b)(i) Degenerate d orbitals are orbitals of the same energy level.
Explanation: In an isolated Ag⁺ ion, all five 3d orbitals (dxy, dxz, dyz, dx²-y², dz²) have identical energy.
(b)(ii)
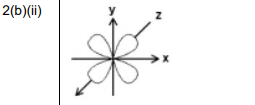
Explanation: The 3dxy orbital has four lobes lying in the xy plane between the axes, with a node at the nucleus.
(c)
Step 1: \(2AgNO_3 + 2NaOH → Ag_2O + 2NaNO_3 + H_2O\)
Step 2: \(Ag_2O + 4NH_3 + H_2O → 2[Ag(NH_3)_2]OH\)
Explanation: In step 1, silver oxide precipitates, while in step 2, it dissolves in ammonia to form the diamminesilver(I) complex.
(d) Linear; H-N-Ag angle: 180°; N-Ag-N angle: 180°.
Explanation: The \([Ag(NH_3)_2]^+\) ion has two ammonia ligands arranged linearly around the central Ag⁺ ion, giving bond angles of 180°.
(e)
Positive electrode: \(Ag_2O + H_2O + 2e^- → 2Ag + 2OH^-\)
Negative electrode: \(Zn + 2OH^- → Zn(OH)_2 + 2e^-\)
Explanation: At the positive electrode, Ag₂O is reduced to Ag, while at the negative electrode, Zn is oxidized to Zn(OH)₂.
(f)
Explanation: The dps ligand bridges between Ru³⁺ ions, with each nitrogen coordinating to a different metal center, forming a polymeric chain with \([Ru(dps)Cl_4]^-\) repeating units.
Topic: 33.2
(a) When a sample of hydrated lithium ethanedioate, Li₂C₂O₄•H₂O, is gently heated, two gaseous products are formed and a white solid residue remains. The residue is added to HNO3(aq). A gas is produced that turns limewater milky. Complete the equation for the decomposition of Li₂C₂O₄•H₂O.![]()
(b) The trend in the decomposition temperatures of the Group 2 ethanedioates is similar to that of the Group 2 nitrates. Suggest which of CaC₂O₄ and BaC₂O₄ will decompose at the lower temperature. Explain your answer.
(c) Potassium iron(III) ethanedioate, K₃[Fe(C₂O₄)₃], dissolves in water to form a green solution. Explain why transition elements can form coloured complexes.
(d) The anhydrous iron(III) compound K₃[Fe(C₂O₄)₃] decomposes on heating to form a mixture of K₂[Fe(C₂O₄)₂], K₂C₂O₄, and CO₂. Complete the equation for the decomposition of K₃[Fe(C₂O₄)₃].
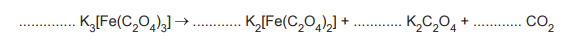
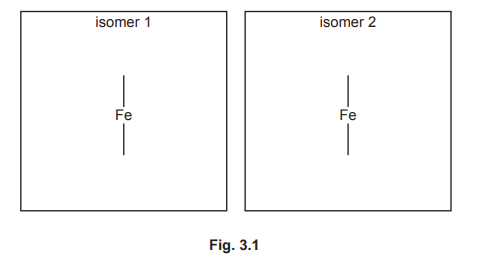
(e) The \([Fe(C_2O_4)_3]^{3–}\) complex ion shows stereoisomerism. Complete the three-dimensional diagrams in Fig. 3.1 to show the two stereoisomers of \([Fe(C_2O_4)_3]^{3–}\). The \(C_2O_4^{2–}\) ligand can be represented using ![]() .
.
(f) Buffer solutions are used to regulate pH. Write two equations to describe how a solution containing HC₂O₄⁻ ions acts as a buffer solution when small amounts of acid or alkali are added.
(g) A fuel cell is an electrochemical cell that can be used to generate electrical energy by using oxygen to oxidise a fuel. Ethanedioic acid, (COOH)₂, dissolved in an alkaline electrolyte, is being investigated as a fuel. The relevant standard electrode potentials, \(E^\Theta \), for the cell are shown.
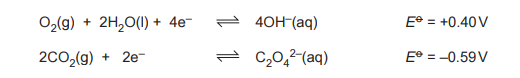
Use these equations to deduce the overall cell reaction. Calculate the value of \(E^{\Theta}_{cell} \).
▶️ Answer/Explanation
(a) \(\text{Li}_2\text{C}_2\text{O}_4 \cdot \text{H}_2\text{O} \rightarrow \text{Li}_2\text{CO}_3 + \text{CO} + \text{H}_2\text{O}\)
Explanation: Heating lithium ethanedioate releases CO and H₂O gases, leaving Li₂CO₃ as residue. When HNO₃ is added, CO₂ is produced (turns limewater milky).
(b) BaC₂O₄ decomposes at a lower temperature than CaC₂O₄.
Explanation: Larger cations (Ba²⁺) stabilize the carbonate ion less effectively than smaller cations (Ca²⁺), so BaC₂O₄ decomposes more easily.
(c) Transition elements form coloured complexes due to d-d electron transitions.
Explanation: The partially filled d-orbitals in transition metals split in the ligand field, absorbing specific wavelengths of light and transmitting the complementary colour (green in this case).
(d) \(\text{2K}_3[\text{Fe}(\text{C}_2\text{O}_4)_3] \rightarrow \text{2K}_2[\text{Fe}(\text{C}_2\text{O}_4)_2] + \text{K}_2\text{C}_2\text{O}_4 + \text{2CO}_2\)
Explanation: The decomposition breaks down the complex into a simpler iron(II) complex, potassium oxalate, and CO₂ gas.
(e) 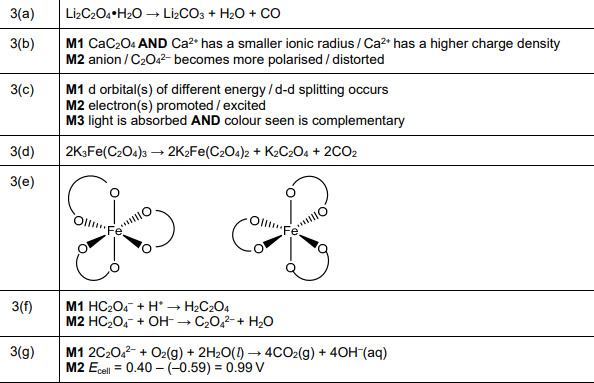
Explanation: The two stereoisomers are the Λ (lambda) and Δ (delta) forms, which are non-superimposable mirror images (enantiomers).
(f)
For acid: \(\text{HC}_2\text{O}_4^- + \text{OH}^- \rightarrow \text{C}_2\text{O}_4^{2-} + \text{H}_2\text{O}\)
For base: \(\text{HC}_2\text{O}_4^- + \text{H}^+ \rightarrow \text{H}_2\text{C}_2\text{O}_4\)
Explanation: HC₂O₄⁻ acts as a buffer by neutralizing added OH⁻ or H⁺, maintaining pH stability.
(g) Overall reaction: \((\text{COOH})_2 + \text{O}_2 \rightarrow 2\text{CO}_2 + 2\text{H}_2\text{O}\)
\(E^{\Theta}_{cell} = 1.23 – (-0.49) = 1.72 \text{ V}\)
Explanation: The cell potential is calculated by subtracting the anode potential from the cathode potential (oxygen reduction).
Topic: 24.1
(a) Define standard electrode potential, \(E^{\Theta}\), including a description of standard conditions.
(b) (i) An electrochemical cell is set up to measure \(E^{\Theta}\) of the \(Ag^+(aq)/Ag(s)\) electrode. Draw a labelled diagram of this electrochemical cell. Include all necessary substances. It is not necessary to state conditions used.
(ii) A separate electrochemical cell is set up using a lower concentration of \(Ag^+(aq)\) than that used in (b)(i). Suggest how the electrode potential, E, for the \(Ag^+(aq)/Ag(s)\) electrode would change from its \(E^{\Theta}\). Explain your answer.
(c) Define enthalpy change of solution, \(\Delta H ^{\Theta}_{sol}\).
(d) Some relevant energy changes for AgNO₃ are shown in Table 4.1.
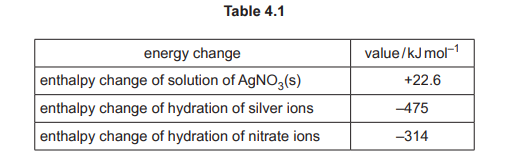
(i) Complete the energy cycle in Fig. 4.1 to show the relationship between the lattice energy, \(\Delta H ^{\Theta}_{latt}\), of AgNO₃(s) and the energy changes shown in Table 4.1. Include state symbols for all the species.
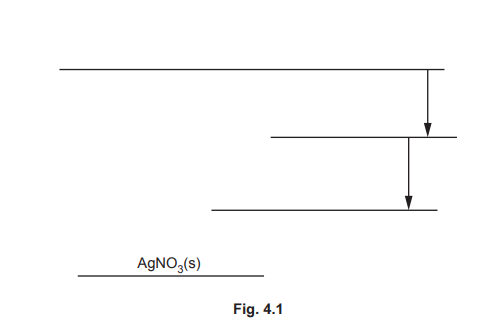
(ii) Calculate the lattice energy, \(\Delta H ^{\Theta}_{latt}\), of AgNO₃(s).
(e) Suggest the trend in the magnitude of the lattice energies of the metal nitrates, NaNO₃(s), Mg(NO₃)₂(s), and RbNO₃(s). Explain your answer.

▶️ Answer/Explanation
(a) The standard electrode potential (\(E^{\Theta}\)) is the voltage of a half-cell compared to the Standard Hydrogen Electrode (SHE) under standard conditions (1 mol dm⁻³ concentration, 101 kPa pressure, and 298 K temperature).
(b)(i)
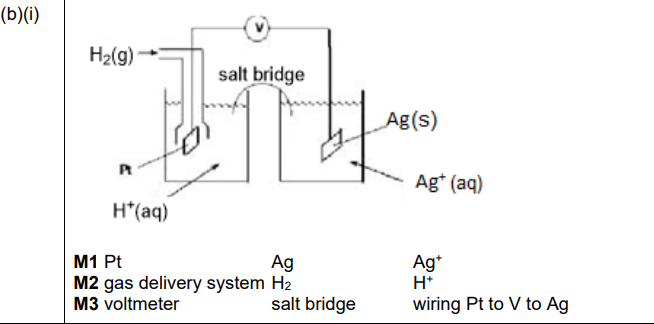
Explanation: The diagram shows the \(Ag^+(aq)/Ag(s)\) half-cell connected to the SHE. The cell includes a salt bridge, voltmeter, and electrodes (Ag and Pt).
(b)(ii) The electrode potential (\(E\)) would become more negative. Explanation: Lower \(Ag^+\) concentration shifts the equilibrium \(Ag^+ + e^- \leftrightarrow Ag\) to the left (Le Chatelier’s Principle), reducing the tendency for reduction.
(c) The enthalpy change of solution (\(\Delta H^{\Theta}_{sol}\)) is the energy change when one mole of a solute dissolves in water to form an infinitely dilute solution.
(d)(i)
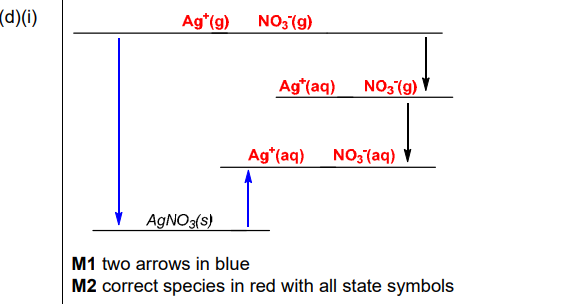
Explanation: The cycle links lattice energy (\(\Delta H^{\Theta}_{latt}\)), enthalpy of hydration (\(\Delta H^{\Theta}_{hyd}\)), and enthalpy of solution (\(\Delta H^{\Theta}_{sol}\)).
(d)(ii) \(\Delta H^{\Theta}_{latt} = -811.6 \ \text{kJ mol}^{-1}\). Explanation: Using Hess’s Law: \(\Delta H^{\Theta}_{latt} = \Delta H^{\Theta}_{sol} – \Delta H^{\Theta}_{hyd} = +22.6 – (+834.2) = -811.6 \ \text{kJ mol}^{-1}\).
(e) The trend is Mg(NO₃)₂(s) > NaNO₃(s) > RbNO₃(s). Explanation: Mg²⁺ has a higher charge density than Na⁺ or Rb⁺, leading to stronger ionic attraction. Na⁺ is smaller than Rb⁺, so NaNO₃ has a higher lattice energy than RbNO₃.
Topic: 26.1
(a) In aqueous solution, persulfate ions, \(S_2O_8^{2–}\), react with iodide ions, as shown in reaction 1.
![]()
The rate of reaction 1 is investigated. A sample of \(S_2O_8^{2–}\) is mixed with a large excess of iodide ions of known concentration. The graph in Fig. 5.1 shows the results obtained.
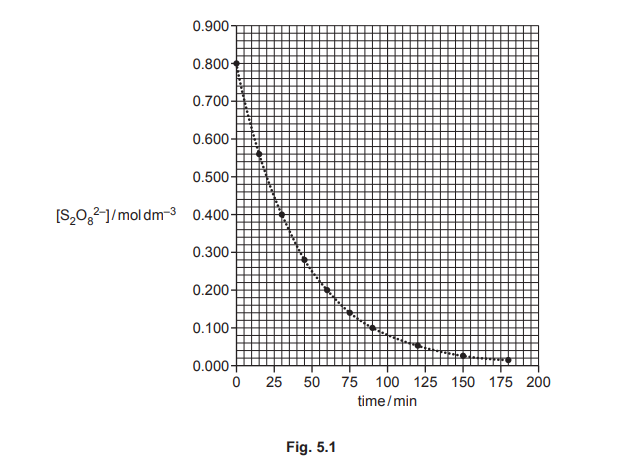
(i) Use Fig. 5.1 to determine the initial rate of reaction 1. Show your working.
(ii) The rate equation for reaction 1 is rate = \(k [S_2O_8^{2–}] [I^–]\). Suggest why a large excess of iodide ions allows the rate constant to be determined from the half-life in this investigation.
(b) The reaction of persulfate ions, \(S_2O_8^{2–}\), with iodide ions is catalysed by \(Fe^{2+}\) ions. Write two equations to show how \(Fe^{2+}\) catalyses reaction 1.
(c) Describe the effect of an increase in temperature on the rate constant and the rate of reaction 1.
(d) In aqueous solution, thiosulfate ions, \(S_2O_3^{2–}\), react with hydrogen ions, as shown in reaction 2.
![]()
The rate of reaction is first order with respect to [\(S_2O_3^{2–}\)] and zero order with respect to [\(H^+\)] under certain conditions. The rate constant, k, for this reaction is \(1.58 \times 10^{–2} s^{–1}\). Calculate the half-life, \(t_{\frac{1}{2}}\) , for reaction 2.
(e) The compound nitrosyl bromide, NOBr, can be formed as shown in reaction 3. ![]()
The rate is first order with respect to [NO] and first order with respect to [\(Br_2\)]. The reaction mechanism has two steps. Suggest equations for the two steps of this mechanism. State which is the rate-determining step.
▶️ Answer/Explanation
(a)(i)
Answer: The initial rate is determined from the tangent at \( t = 0 \). From the graph, the slope is approximately \(-0.0125 \, \text{mol dm}^{-3} \, \text{s}^{-1}\).
Explanation: The initial rate is the change in concentration of \( S_2O_8^{2-} \) per unit time at \( t = 0 \). The tangent at \( t = 0 \) gives a slope of \(-0.0125\), so the rate is \( 0.0125 \, \text{mol dm}^{-3} \, \text{s}^{-1} \).
(a)(ii)
Answer: A large excess of \( I^- \) ensures its concentration remains approximately constant, making the reaction pseudo-first order with respect to \( S_2O_8^{2-} \). The half-life then depends only on \( k \).
Explanation: Since \( [I^-] \) is constant, the rate equation simplifies to \( \text{rate} = k’ [S_2O_8^{2-}] \), where \( k’ = k[I^-] \). The half-life of a first-order reaction is \( t_{1/2} = \frac{\ln 2}{k’} \), allowing \( k \) to be determined.
(b)
Answer: \[ Fe^{2+} + S_2O_8^{2-} \rightarrow Fe^{3+} + 2SO_4^{2-} \] \[ Fe^{3+} + I^- \rightarrow Fe^{2+} + \frac{1}{2} I_2 \]
Explanation: \( Fe^{2+} \) is oxidised to \( Fe^{3+} \) by \( S_2O_8^{2-} \), and \( Fe^{3+} \) is reduced back to \( Fe^{2+} \) by \( I^- \), regenerating the catalyst.
(c)
Answer: An increase in temperature increases the rate constant \( k \) (Arrhenius equation) and thus the rate of reaction.
Explanation: The Arrhenius equation \( k = Ae^{-E_a/RT} \) shows that \( k \) increases with temperature. Since rate \( = k[S_2O_8^{2-}][I^-] \), the rate also increases.
(d)
Answer: The half-life \( t_{1/2} = \frac{\ln 2}{k} = \frac{0.693}{1.58 \times 10^{-2}} \approx 43.9 \, \text{s} \).
Explanation: For a first-order reaction, \( t_{1/2} = \frac{\ln 2}{k} \). Substituting \( k = 1.58 \times 10^{-2} \, \text{s}^{-1} \) gives \( t_{1/2} \approx 43.9 \, \text{s} \).
(e)
Answer: Step 1 (slow, rate-determining): \( NO + Br_2 \rightarrow NOBr_2 \)
Step 2 (fast): \( NOBr_2 + NO \rightarrow 2NOBr \)
Explanation: The rate-determining step involves one NO and one \( Br_2 \) molecule, matching the observed rate law. The second step is fast and does not affect the rate.

Topic: 34.4
(a) (i) State what is meant by partition coefficient, \(K_{pc}\).
(ii) The partition coefficient, \(K_{pc}\), for a compound, X, between carbon disulfide, CS₂, and water is 10.5. 1.85 g of X is dissolved in water and made up to 100.0 cm³ in a volumetric flask. 40.0 cm³ of this aqueous solution is shaken with 25.0 cm³ of CS₂. The mixture is left to reach equilibrium. Calculate the mass of X, in g, extracted into the \(CS_2\) layer.
(b) The compound \(C_6H_6\) has many structural isomers. Four suggested structures of \(C_6H_6\) are shown in Fig. 6.1.
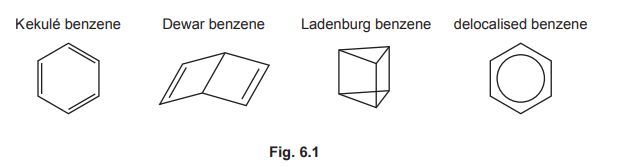
Using Fig. 6.1, complete Table 6.1 to predict the number of carbon atoms that have sp, \(sp^2\), and \(sp^3\) hybridisation in Kekulé benzene, Dewar benzene, and Ladenburg benzene.

(c) Describe the shape of delocalised benzene. Include the geometry of each carbon, the C-C-H bond angle, and the type of bond(s) between the carbon atoms and between the carbon and hydrogen atoms.
(d) Suggest why Dewar benzene and Ladenburg benzene are unstable isomers of \(C_6H_6\).
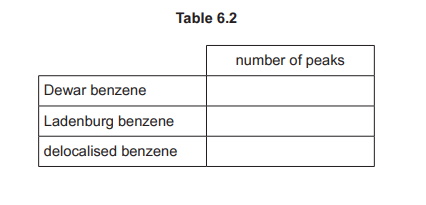
(e) Complete Table 6.2 to predict the number of peaks in the proton \((^1H)\) NMR spectrum for Dewar benzene, Ladenburg benzene and delocalised benzene.
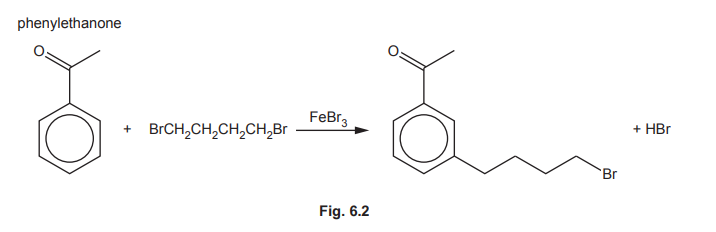
(f) The reaction of phenylethanone with 1,4-dibromobutane, BrCH₂CH₂CH₂CH₂Br, in the presence of FeBr₃ is shown in Fig. 6.2.
The mechanism of this reaction is similar to that of the alkylation of benzene.
(i) Construct an equation for the formation of the electrophile, BrCH₂CH₂CH₂CH₂⁺.
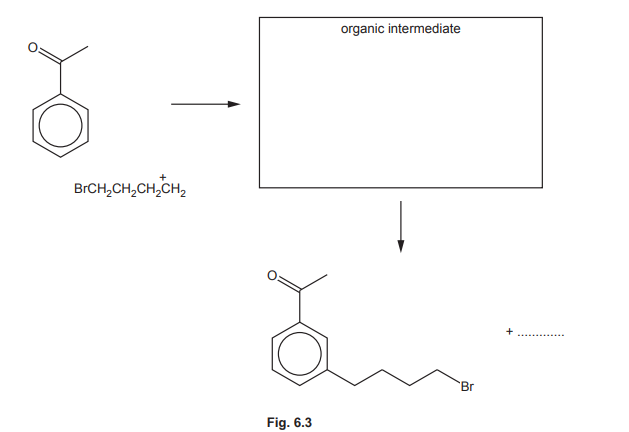
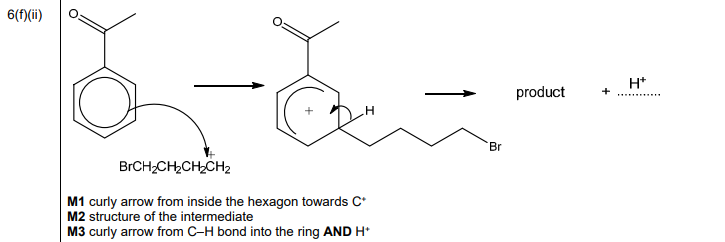
(ii) Complete the mechanism in Fig. 6.3 for the reaction of phenylethanone with BrCH₂CH₂CH₂CH₂⁺ ions. Include all relevant curly arrows and charges. Draw the structure of the organic intermediate.
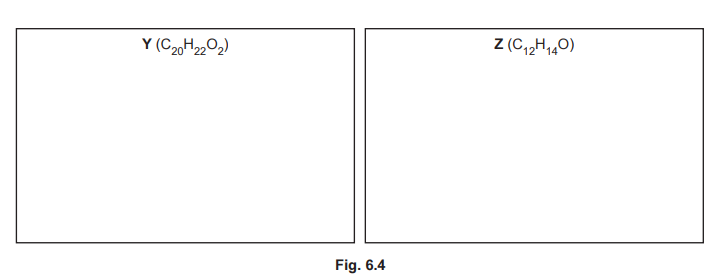
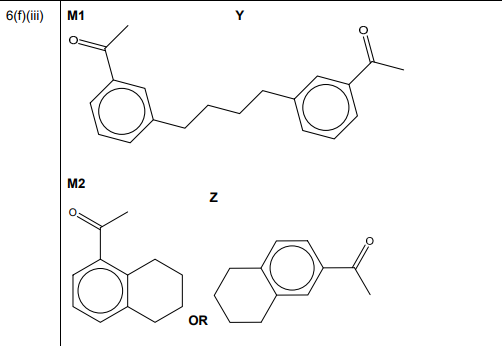
(iii) The reaction shown in Fig. 6.2 forms small amounts of two by-products, Y \((C_{20}H_{22}O_2)\) and Z \((C_{12}H_{14}O)\). Suggest structures for Y and Z in the boxes in Fig. 6.4.
▶️ Answer/Explanation
(a)(i) The partition coefficient, \(K_{pc}\), is the ratio of the concentrations of a solute in two immiscible solvents at equilibrium.
Explanation: It describes how a solute distributes itself between two phases, such as organic and aqueous layers.
(a)(ii) 0.642 g
Explanation: Using the partition coefficient formula \(K_{pc} = \frac{[X]_{CS_2}}{[X]_{H_2O}}\), we set up the equation \(10.5 = \frac{(y/25)}{(0.74 – y)/40}\), where \(y\) is the mass extracted into CS₂. Solving gives \(y = 0.642 \, \text{g}\).
(b)
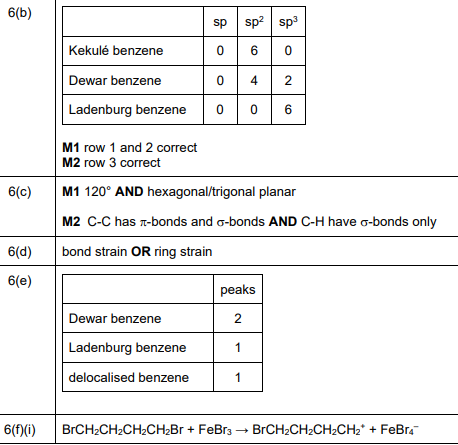
Explanation: Kekulé benzene has 6 \(sp^2\) carbons. Dewar benzene has 4 \(sp^2\) and 2 \(sp^3\) carbons. Ladenburg benzene has 2 \(sp\), 2 \(sp^2\), and 2 \(sp^3\) carbons.
(c) Delocalised benzene is planar with each carbon having trigonal planar geometry. The C-C-H bond angle is 120°. The C-C bonds are delocalised \(\pi\)-bonds, and C-H bonds are sigma (\(\sigma\)) bonds.
Explanation: The ring is flat due to \(sp^2\) hybridisation, with equal bond lengths from electron delocalisation.
(d) Dewar benzene and Ladenburg benzene are unstable due to ring strain and lack of aromaticity (delocalisation).
Explanation: Their structures introduce angle strain and prevent the stability conferred by aromatic \(\pi\)-electron delocalisation in benzene.
(e)
Explanation: Dewar benzene has 2 peaks (2 types of H), Ladenburg benzene has 3 peaks (3 types of H), and delocalised benzene has 1 peak (all H equivalent).
(f)(i) \(\text{BrCH}_2\text{CH}_2\text{CH}_2\text{CH}_2\text{Br} + \text{FeBr}_3 \rightarrow \text{BrCH}_2\text{CH}_2\text{CH}_2\text{CH}_2^+ + \text{FeBr}_4^-\)
Explanation: FeBr₃ acts as a Lewis acid, polarising the C-Br bond to generate the electrophile.
(f)(ii)
Explanation: The mechanism involves electrophilic attack on the benzene ring, forming a carbocation intermediate, followed by deprotonation to restore aromaticity.
(f)(iii) Y is a dimer (two phenylethanone units bridged by the dibromobutane chain). Z is a monosubstituted product (one phenylethanone with a butyl group).
Explanation: Y forms from two electrophilic attacks, while Z results from a single substitution.
Topic: 37.3
Four esters, A, B, C and D, with the molecular formula \(C_6H_{12}O_2\), are shown in Fig. 7.1.
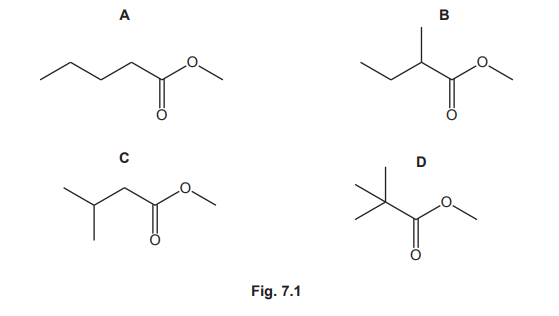
(a) Give the systematic name of ester A.
(b) A mixture of these esters, A, B, C and D, is analysed by gas–liquid chromatography. The chromatogram produced is shown in Fig. 7.2. The number above each peak represents the area under the peak. The area under each peak is proportional to the mass of the respective ester in the mixture.
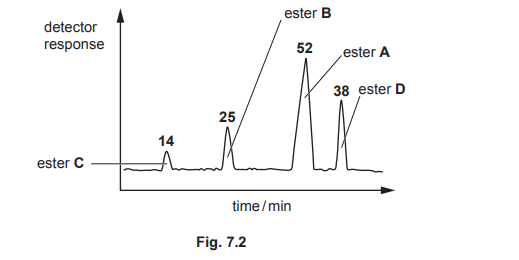
(i) State what is meant by retention time.
(ii) Calculate the percentage by mass of ester D in the original mixture.
(c) Separate samples of the esters, A, B, C and D, are analysed using proton \((^1H)\) NMR and carbon-13 NMR spectroscopy.
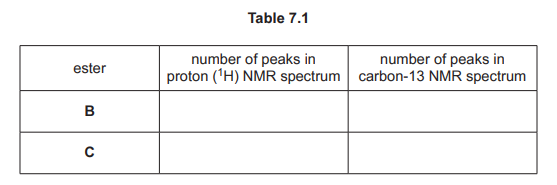
(i) Complete Table 7.1 to show the number of peaks in each NMR spectrum for esters B and C.
(ii) Identify all of the esters from A, B, C and D that have at least one triplet peak in their proton \((^1H)\) NMR spectrum.
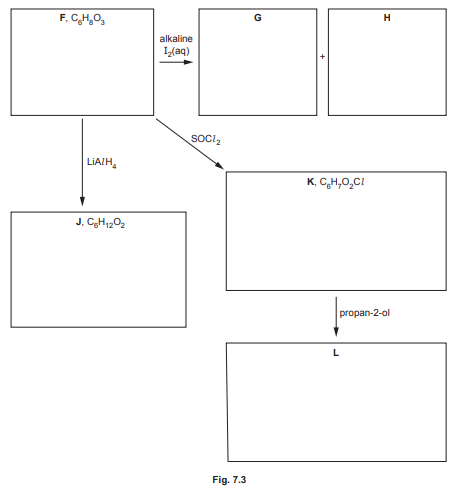
(d) Compound F, \(C_6H_8O_3\), shows stereoisomerism and effervesces with \(Na_2CO_3(aq)\).
Compound F reacts with alkaline \(I_2\)(aq) to form yellow precipitate G and compound H.
Compound F reacts with \(LiAl H_4\) to form compound J, \(C_6H_{12}O_2\).
Compound F reacts with SOCl₂ to form compound K, C₆H₇O₂Cl.
Compound K reacts with propan-2-ol to form compound L.
Draw the structures of compounds F, G, H, J, K and L in the boxes in Fig. 7.3.
▶️ Answer/Explanation
(a) methyl pentanoate
Explanation: Ester A has a 5-carbon chain (pentanoate) and a methyl group (\(-OCH_3\)) attached to the carbonyl carbon, hence the name methyl pentanoate.
(b)(i) Time between injection and detection.
Explanation: Retention time is the time taken for a compound to travel through the chromatography column from injection to detection.
(b)(ii) 29.5%
Explanation: The area under peak D is 59, and the total area is 200. Thus, the percentage of D is \(\frac{59}{200} \times 100 = 29.5\%\).
(c)(i)
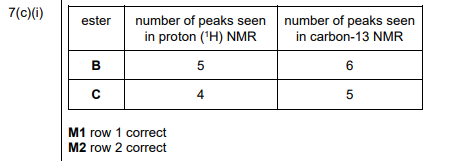
Explanation: Ester B has 4 proton environments (peaks in \(^1H\) NMR) and 5 carbon environments (peaks in \(^{13}C\) NMR). Ester C has 3 proton environments and 4 carbon environments.
(c)(ii) A and B
Explanation: Triplet peaks arise from protons adjacent to \(-CH_2\) groups. Esters A and B have such environments, while C and D do not.
(d)
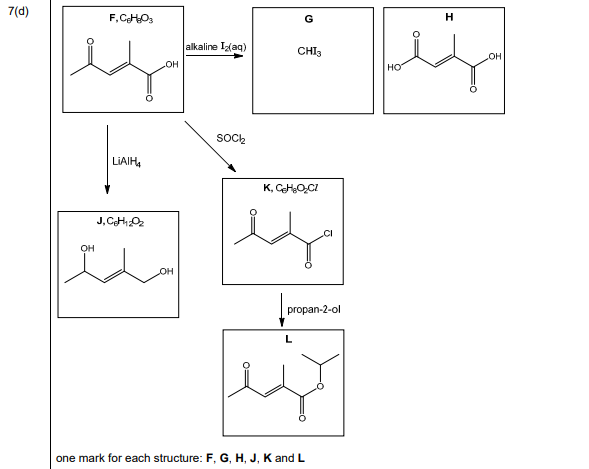
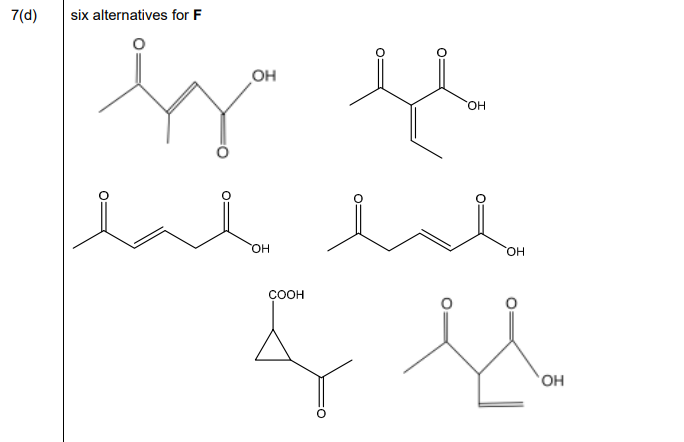
Explanation: Compound F is a \(\beta\)-ketoacid (\(CH_3COCH_2COOH\)) due to its reactions. G is iodoform (\(CHI_3\)), H is sodium salt (\(CH_3COONa\)), J is reduced to a diol (\(HOCH_2CH(OH)CH_3\)), K is the acyl chloride (\(CH_3COCH_2COCl\)), and L is the ester (\(CH_3COCH_2COOCH(CH_3)_2\)).
Topic: 35.3
Neotame is an artificial sweetener added to some foods.
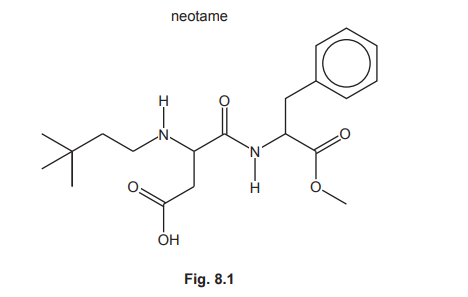
(a) (i) State the number of chiral carbon atoms in a molecule of neotame.
(ii) Neotame contains the arene functional group. Identify all the other functional groups present in neotame.
(b) Neotame reacts with an excess of hot NaOH(aq) to form three organic products.
(i) State the two types of reaction that occur when neotame reacts with hot NaOH(aq).
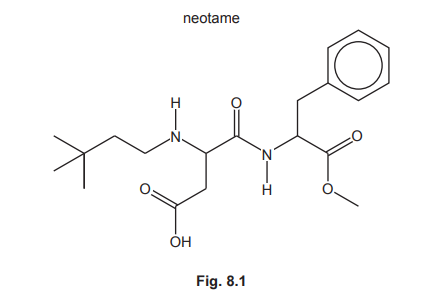
(ii) Draw the structures of the three organic products formed from the reaction of neotame with an excess of hot NaOH(aq).

▶️ Answer/Explanation
(a)(i) Two.
Explanation: Chiral carbons are those attached to four different groups. In neotame, the two carbons marked with asterisks (*) in the structure are chiral because they each have four distinct substituents.
(a)(ii) Amide, amine, ester, carboxylic acid.
Explanation: The functional groups in neotame are identified as follows: the amide (–CONH–), amine (–NH2), ester (–COO–), and carboxylic acid (–COOH) groups are all present in addition to the arene (benzene ring).
(b)(i) Hydrolysis and acid-base reaction.
Explanation: The ester and amide groups undergo hydrolysis in hot NaOH, breaking into smaller molecules. The carboxylic acid group participates in an acid-base reaction with NaOH, forming a carboxylate ion.
(b)(ii)
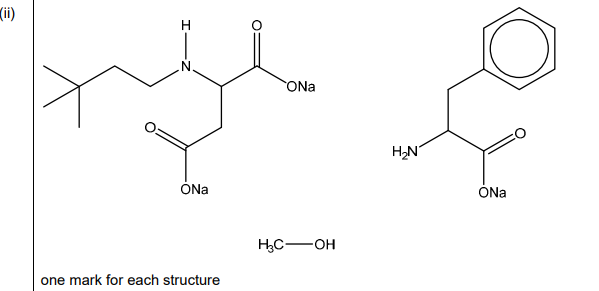
Explanation: The three organic products are: (1) sodium carboxylate (from the carboxylic acid group), (2) an amine (from the amide hydrolysis), and (3) an alcohol (from the ester hydrolysis). The structures are correctly drawn in the image above.
Topic: 34.2
(a) Samples of phenol, C₆H₅OH, are reacted separately with sodium and with dilute nitric acid.
(i) Write the equation for the reaction of C₆H₅OH with Na.
(ii) Draw the structures of the two major isomeric organic products formed in the reaction of phenol with dilute HNO₃.

(b) Salicylic acid can be synthesised from phenol.
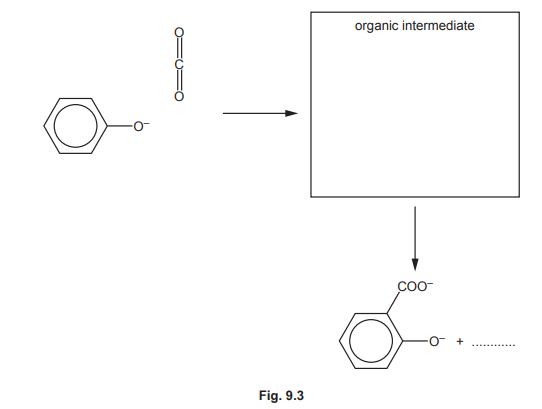
One of the steps in this synthesis is the electrophilic substitution reaction of carbon dioxide with the phenoxide ion, C₆H₅O⁻. Complete the mechanism in Fig. 9.3 for the reaction of C₆H₅O⁻ with \(CO_2\). Include all relevant curly arrows, dipoles and charges. Draw the structure of the organic intermediate.
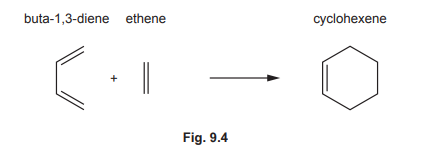
(c) Some syntheses use Diels–Alder reactions, which normally involve a diene and an alkene reacting together to form a cyclohexene.
(i) Draw three curly arrows in Fig. 9.4 to complete the mechanism for the Diels–Alder reaction between buta-1,3-diene and ethene.
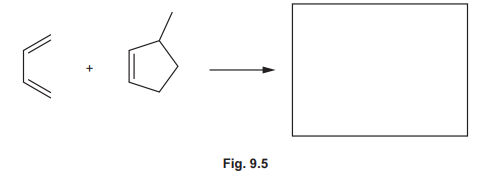
(ii) Another Diels–Alder reaction of buta-1,3-diene is shown in Fig. 9.5. Predict the product formed in this reaction.
▶️ Answer/Explanation
(a)(i) The reaction of phenol with sodium produces sodium phenoxide and hydrogen gas:
\[ 2C_6H_5OH + 2Na \rightarrow 2C_6H_5O^-Na^+ + H_2 \]
Explanation: Phenol reacts with sodium similarly to alcohols, where the acidic hydrogen is replaced by sodium, forming a phenoxide ion and releasing \( H_2 \).
text
(a)(ii) The two major isomeric products from nitration of phenol are 2-nitrophenol and 4-nitrophenol.
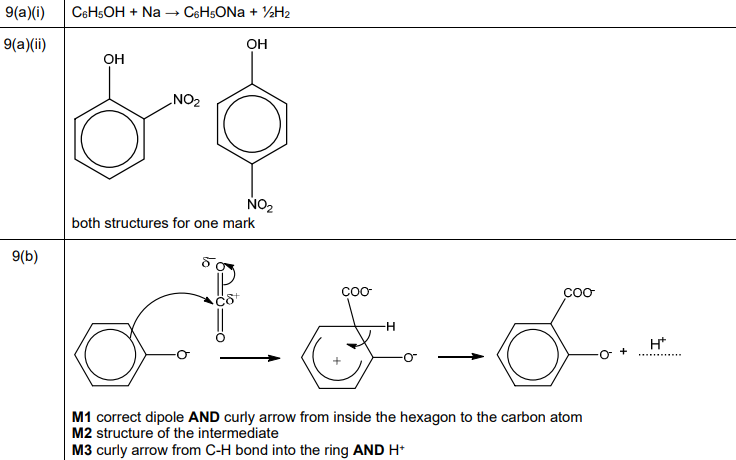
Explanation: Dilute \( HNO_3 \) nitrates phenol at the ortho (2-) and para (4-) positions due to the electron-donating effect of the hydroxyl group.
(b) The mechanism involves electrophilic attack of \( CO_2 \) on the phenoxide ion, forming a carboxylate intermediate.
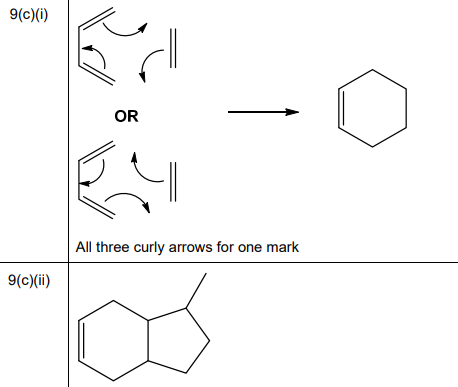
Explanation: \( CO_2 \) acts as an electrophile, attacking the electron-rich ortho position of the phenoxide ion. The intermediate is stabilised by resonance, leading to salicylic acid after protonation.
(c)(i) The Diels-Alder mechanism involves three curly arrows showing the movement of π-electrons to form new σ-bonds.
(c)(ii) The product is a cyclohexene derivative formed by [4+2] cycloaddition.
Explanation: Buta-1,3-diene reacts with ethene to form cyclohexene. The reaction proceeds via a concerted mechanism, where π-electrons rearrange to form a six-membered ring.
