CIE AS/A Level Chemistry 1.1 Particles in the atom and atomic radius Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 1.1 Particles in the atom and atomic radius Study Notes – New Syllabus
CIE AS/A Level Chemistry 1.1 Particles in the atom and atomic radius Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- understand that atoms are mostly empty space surrounding a very small, dense nucleus that contains protons and neutrons; electrons are found in shells in the empty space around the nucleus
- identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses
- understand the terms atomic and proton number; mass and nucleon number
- describe the distribution of mass and charge within an atom
- describe the behaviour of beams of protons, neutrons and electrons moving at the same velocity in an electric field
- determine the numbers of protons, neutrons and electrons present in both atoms and ions given atomic or proton number, mass or nucleon number and charge
- state and explain qualitatively the variations in atomic radius and ionic radius across a period and down a group
Atomic Structure — Empty Space, Nucleus and Electron Shells
Atoms consist of a very small, dense nucleus surrounded by mostly empty space in which electrons move in shells or orbitals.

\( \text{atom} = \text{very small dense nucleus} + \text{electrons in shells in the surrounding space} \)
- Nucleus contains protons and neutrons and holds nearly all the atom’s mass.
- Electrons are found in shells (energy levels) in the space around the nucleus and have negligible mass compared with protons and neutrons.
- The atom is mostly empty space because the nucleus is tiny compared with the overall atomic size.
Key points
- Typical atomic radius is about \( \mathrm{1\times10^{-10}\ m} \) (0.1 nm).
- Typical nuclear radius is about \( \mathrm{1\times10^{-15}\ m} \) (1 fm).
- Mass of a proton or neutron \( \approx \mathrm{1.67\times10^{-27}\ kg} \); mass of an electron \( \approx \mathrm{9.11\times10^{-31}\ kg} \) — electrons contribute very little to atomic mass.
Why we say atoms are mostly empty space
Compare radii:
\( \dfrac{\text{nuclear radius}}{\text{atomic radius}} \approx \dfrac{1\times10^{-15}}{1\times10^{-10}} = 1\times10^{-5} \)
Volume scales with the cube of radius, so the fraction of volume occupied by the nucleus is roughly
\( \left(1\times10^{-5}\right)^3 = 1\times10^{-15} \)
Therefore the nucleus occupies about \( \mathrm{10^{-15}} \) of the atom’s volume — the rest is essentially empty space occupied by electron probability clouds.
Mass distribution
- Nearly all the mass of the atom is in the nucleus because protons and neutrons are ~1836 times more massive than electrons.
- Example: a neutral carbon atom (6 protons, 6 neutrons, 6 electrons) — mass is almost entirely from the 12 nucleons.
Example
State why most of an atom’s volume is considered empty space.
▶️ Answer / Explanation
The nucleus is extremely small compared with the overall atom (nuclear radius \( \approx \mathrm{1\times10^{-15}\ m} \) vs atomic radius \( \approx \mathrm{1\times10^{-10}\ m} \)).
Because volume scales with the cube of radius, the nucleus occupies about \( \mathrm{10^{-15}} \) of the atom’s volume, so nearly all the atom’s volume is empty space where electrons reside.
Example
Calculate the approximate fraction of an atom’s volume occupied by the nucleus, using atomic radius \( \mathrm{1.0\times10^{-10}\ m} \) and nuclear radius \( \mathrm{1.0\times10^{-15}\ m} \).
▶️ Answer / Explanation
Fraction \( = \left(\dfrac{r_{\text{nucleus}}}{r_{\text{atom}}}\right)^3 = \left(\dfrac{1.0\times10^{-15}}{1.0\times10^{-10}}\right)^3 = (1.0\times10^{-5})^3 = 1.0\times10^{-15} \).
The nucleus occupies about \( \mathrm{1\times10^{-15}} \) of the atom’s volume.
Example
A neutral lithium atom has 3 protons, 4 neutrons and 3 electrons. Explain where the mass and the charge are located in the atom and estimate the percentage of mass carried by the electrons.
▶️ Answer / Explanation
Mass is concentrated in the nucleus (3 protons + 4 neutrons = 7 nucleons). Each nucleon has mass \( \approx \mathrm{1.67\times10^{-27}\ kg} \), so total nuclear mass \( \approx 7\times1.67\times10^{-27} = 1.169\times10^{-26}\ kg \).
Mass of 3 electrons \( \approx 3\times9.11\times10^{-31} = 2.73\times10^{-30}\ kg \).
Percentage of mass in electrons \( \approx \dfrac{2.73\times10^{-30}}{1.169\times10^{-26}+2.73\times10^{-30}}\times100\% \approx \dfrac{2.73\times10^{-30}}{1.1693\times10^{-26}}\times100\% \)
\( \approx 2.34\times10^{-2}\% \) (about \( \mathrm{0.023\%} \) ).
Charge is distributed as positive charge in the nucleus due to protons and negative charge from electrons in shells; the net charge of the neutral atom is zero.
Conclusion: almost all mass is in the nucleus while electrons determine the atom’s size and chemical behaviour.
Subatomic Particles — Protons, Neutrons and Electrons
Atoms are made of three fundamental particles: protons, neutrons and electrons. These particles differ in their relative charges and relative masses.

Relative Charge and Relative Mass
- Protons have a relative charge of +1 and a relative mass of 1.
- Neutrons have no charge (0) and a relative mass of 1.
- Electrons have a relative charge of -1 and a very small relative mass of \( \mathrm{\tfrac{1}{1836}} \approx 0.0005 \).
Location in the Atom
- Protons are found in the nucleus.
- Neutrons are found in the nucleus.
- Electrons move rapidly in shells in the space around the nucleus.
| Sub atomic particle | Symbol | Location in the atom | Relative Charge | Relative Mass | Actual Mass (kg) | Absolute Mass (g) |
|---|---|---|---|---|---|---|
| Electron | e⁻ | Outside the nucleus | -1 | \( \mathrm{\tfrac{1}{1840}} \) | 9.1 × 10⁻³¹ | 9 × 10⁻²⁸ |
| Proton | \( \mathrm{p^+} \) | Inside the nucleus | +1 | 1 a.m.u. | 1.673 × 10⁻²⁷ | 1.6 × 10⁻²⁴ |
| Neutron | n | Inside the nucleus | 0 | 1 a.m.u. | 1.675 × 10⁻²⁷ | 1.6 × 10⁻²⁴ |
Example
State the relative charge and relative mass of an electron.
▶️ Answer / Explanation
Electrons have a relative charge of -1 and a relative mass of \( \mathrm{\tfrac{1}{1836}} \), which is much smaller than that of protons or neutrons.
Example
Explain why the mass of an atom is almost entirely due to its protons and neutrons.
▶️ Answer / Explanation
Protons and neutrons each have a relative mass of 1, while electrons have a relative mass of only \( \mathrm{\tfrac{1}{1836}} \). Because electrons are so light, their mass contribution is negligible compared with the nucleons in the nucleus.
Example
An atom contains 11 protons, 12 neutrons and 11 electrons. Calculate the total relative mass and the net charge of the atom.
▶️ Answer / Explanation
Total relative mass = protons + neutrons = \( 11 + 12 = 23 \).
Total charge = charge from protons + charge from electrons = \( 11(+1) + 11(-1) = 0 \).
The atom has a relative mass of 23 and is electrically neutral.
Atomic Number, Proton Number, Mass Number and Nucleon Number
Atoms can be described using several important numbers that give information about the number of particles they contain. These include the atomic (proton) number and the mass (nucleon) number.

Atomic Number / Proton Number (Z)
- Atomic number is the number of protons in the nucleus of an atom.
- It is represented by the symbol \( \mathrm{Z} \).
- Since all atoms are electrically neutral, the number of electrons equals the number of protons.
- Atomic number determines the identity of the element.
Mass Number / Nucleon Number (A)
- Mass number is the total number of protons and neutrons in the nucleus.
- It is represented by the symbol \( \mathrm{A} \).
- Protons and neutrons together are called nucleons.
- Mass number always equals: \( \mathrm{A = Z + \text{number of neutrons}} \).
Representing Atoms Using A and Z
\( ^A_Z\text{X} \) where X is the chemical symbol.
Example: \( ^{23}_{11}\text{Na} \) has 11 protons and 12 neutrons.
Example
Magnesium has the symbol \( ^{24}_{12}\text{Mg} \). State its number of protons, neutrons and electrons.
▶️ Answer / Explanation
Protons = 12 (from Z).
Neutrons = \( 24 – 12 = 12 \).
Electrons = 12 (atom is neutral).
Example
An atom of chlorine is written as \( ^{35}_{17}\text{Cl} \). Determine its nucleon number and explain what this number represents.
▶️ Answer / Explanation
Nucleon number = 35.
It represents the total number of particles in the nucleus, which are protons and neutrons.
Example
Two isotopes of carbon are \( ^{12}_{6}\text{C} \) and \( ^{14}_{6}\text{C} \). Compare their atomic numbers, mass numbers and numbers of neutrons.
▶️ Answer / Explanation
Both isotopes have the same atomic (proton) number, 6.
Their mass numbers differ: 12 and 14.
Neutrons in \( ^{12}_{6}\text{C} \): \( 12 – 6 = 6 \).
Neutrons in \( ^{14}_{6}\text{C} \): \( 14 – 6 = 8 \).
They differ only in number of neutrons.
Distribution of Mass and Charge in an Atom
Atoms have an uneven distribution of mass and charge. Nearly all the mass is concentrated in the nucleus, while the charges are distributed between the nucleus and the surrounding electrons.
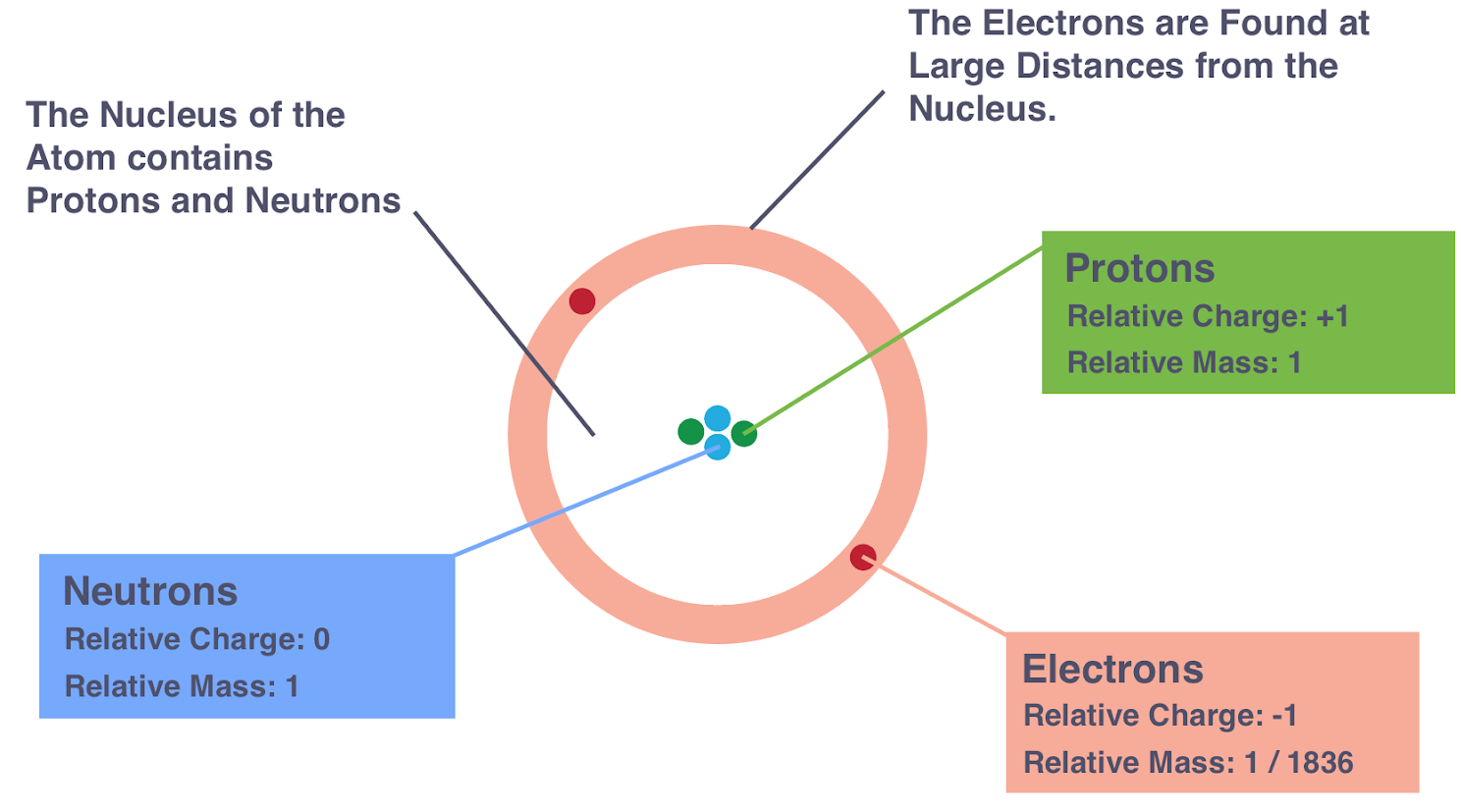
Mass Distribution
- The nucleus contains protons and neutrons, each with a relative mass of 1, so the nucleus holds almost all the atom’s mass.
- Electrons have a very tiny mass \( \mathrm{\left(\tfrac{1}{1836}\right)} \), so their contribution to the total mass is negligible.
- Thus, the atom’s mass is almost entirely located at the centre of the atom.
Charge Distribution
- Protons in the nucleus carry positive charge.
- Neutrons have no charge and contribute nothing to the electrical charge of the atom.
- Electrons carry negative charge and are found in shells around the nucleus.
- In a neutral atom, the total positive charge of protons equals the total negative charge of electrons.
Example
Explain why the nucleus accounts for almost all the mass of an atom.
▶️ Answer / Explanation
Protons and neutrons each have a relative mass of 1, while electrons have a negligible mass of \( \mathrm{\tfrac{1}{1836}} \). Since protons and neutrons are located in the nucleus, almost all the mass is concentrated there.
Example
Describe the charge distribution in a neutral atom of sodium (\( ^{23}_{11}\text{Na} \)).
▶️ Answer / Explanation
Sodium has 11 protons, giving the nucleus a charge of +11. It also has 11 electrons, each with a charge of -1, distributed in shells. The total charge is +11 + (-11) = 0, so the atom is neutral.
Example
An atom of chlorine has 17 protons, 18 neutrons and 17 electrons. Explain where its mass and charge are located and identify its overall charge.
▶️ Answer / Explanation
Mass: Located almost entirely in the nucleus because protons and neutrons each have a relative mass of 1. Total mass number = \( 17 + 18 = 35 \).
Charge: Positive charge (+17) is in the nucleus from protons. Negative charge (-17) is around the nucleus due to electrons in shells.
Total charge = \( +17 + (-17) = 0 \), so the atom is neutral.
Conclusion: Mass is centralised in the nucleus while charges are separated between nucleus (positive) and shells (negative).
Behaviour of Beams of Protons, Neutrons and Electrons in an Electric Field
When beams of charged or uncharged particles pass through an electric field, they behave differently depending on their charge and mass. Protons, neutrons and electrons moving at the same velocity experience different degrees of deflection.
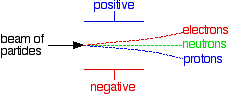
Electric Field Effect on Each Particle
- Protons are positively charged, so they are attracted toward the negative plate and repelled by the positive plate.
- Electrons are negatively charged, so they are attracted toward the positive plate and repelled by the negative plate.
- Neutrons have no charge, so they are unaffected and continue in a straight line.
![]()
Relative Deflection
- Electrons experience the greatest deflection because they have a very small mass.
- Protons are deflected in the opposite direction but much less than electrons because they are much heavier.
- Neutrons experience no deflection at all.
Example
A beam of electrons passes between two charged parallel plates. Describe and explain its path.
▶️ Answer / Explanation
Electrons have a negative charge and are attracted to the positive plate. Because their mass is very small, they undergo strong deflection toward the positive plate.
Example
Protons and electrons of the same velocity enter a uniform electric field. Compare their deflections.
▶️ Answer / Explanation
Both are deflected in opposite directions because their charges are opposite. Electrons show a much larger deflection because their mass is far smaller. Protons are deflected slightly due to their much larger mass.
Example
Three beams (protons, neutrons and electrons) enter an electric field with the same speed. Predict and explain the relative curvature of their paths.
▶️ Answer / Explanation
Electrons curve sharply toward the positive plate due to their negative charge and tiny mass.
Protons curve gently toward the negative plate because they are positively charged but far heavier.
Neutrons do not curve at all because they have no charge.
Therefore, electron path is most curved, proton path is slightly curved, and neutron path remains straight.
Determining Numbers of Protons, Neutrons and Electrons
Given the atomic (proton) number \( \mathrm{Z} \), the mass (nucleon) number \( \mathrm{A} \), and the ionic charge, you can find the numbers of protons, neutrons and electrons in atoms and ions using a few simple relationships.

Key relationships
- Number of protons \( = \mathrm{Z} \). (Atomic number defines the element.)
- Number of neutrons \( = \mathrm{A – Z} \).
- Number of electrons \( = \mathrm{Z – q} \), where \( \mathrm{q} \) is the ion charge written with its sign (for example \( \mathrm{q = +2} \) for \( 2^+ \) and \( \mathrm{q = -1} \) for \( 1^- \)).
Notes:
- For a neutral atom \( \mathrm{q = 0} \), so electrons \( = \mathrm{Z} \).
- When the ion is positive (cation), \( \mathrm{q} \) is positive and electrons \( = \mathrm{Z – q} \) (fewer electrons than protons).
- When the ion is negative (anion), \( \mathrm{q} \) is negative and electrons \( = \mathrm{Z – (negative\;q)} = \mathrm{Z + |q|} \) (more electrons than protons).
- Mass number \( \mathrm{A} \) does not change when an atom gains or loses electrons; it only changes if nucleons are added/removed.
How to proceed — quick method
- Step 1: read off \( \mathrm{Z} \) — that is the number of protons.
- Step 2: if \( \mathrm{A} \) is given, compute neutrons \( = \mathrm{A – Z} \).
- Step 3: use the ionic charge \( \mathrm{q} \) and compute electrons \( = \mathrm{Z – q} \) (treat \( \mathrm{q} \) with its sign).
Other common question forms and tips
- If you are given only the element symbol and the ionic charge (for example \( \mathrm{Ca^{2+}} \)), use the periodic table to find \( \mathrm{Z} \) (calculation of neutrons needs \( \mathrm{A} \)).
- If you are given total number of electrons and the charge is not stated explicitly, you can deduce the charge: \( \mathrm{q = Z – number\ of\ electrons} \).
- Always check sign conventions carefully — a positive q means fewer electrons than protons; a negative q means extra electrons.
Quick summary
- Protons = \( \mathrm{Z} \).
- Neutrons = \( \mathrm{A – Z} \) (if \( \mathrm{A} \) given).
- Electrons = \( \mathrm{Z – q} \) (use signed charge \( \mathrm{q} \)).
Example
Find the numbers of protons, neutrons and electrons in the neutral atom \( ^{23}_{11}\text{Na} \).
▶️ Answer / Explanation
Protons \( = \mathrm{Z} = 11 \).
Neutrons \( = \mathrm{A – Z} = 23 – 11 = 12 \).
Charge \( \mathrm{q = 0} \) for a neutral atom, so electrons \( = \mathrm{Z – q} = 11 – 0 = 11 \).
Result: protons 11, neutrons 12, electrons 11.
Example
An ion is \( ^{35}_{17}\text{Cl}^- \). Determine protons, neutrons and electrons.
▶️ Answer / Explanation
Protons \( = \mathrm{Z} = 17 \).
Neutrons \( = \mathrm{A – Z} = 35 – 17 = 18 \).
Ion charge is \( \mathrm{^-} \) so \( \mathrm{q = -1} \). Electrons \( = \mathrm{Z – q} = 17 – (-1) = 17 + 1 = 18 \).
Result: protons 17, neutrons 18, electrons 18.
Example
An ion of iron is written as \( \mathrm{Fe^{2+}} \) and its mass number is 56 (so \( ^{56}\mathrm{Fe^{2+}} \)). Determine numbers of protons, neutrons and electrons.
▶️ Answer / Explanation
Step 1 — protons: for iron \( \mathrm{Z = 26} \), so protons = 26.
Step 2 — neutrons: \( \mathrm{A – Z = 56 – 26 = 30} \).
Step 3 — electrons: ion has charge \( \mathrm{q = +2} \). Electrons \( = \mathrm{Z – q} = 26 – 2 = 24 \).
Result: protons 26, neutrons 30, electrons 24.
Variations in Atomic Radius and Ionic Radius Across a Period and Down a Group

Atomic and ionic radii change in predictable ways when moving across a period or down a group. These trends can be explained qualitatively by considering nuclear charge, electron shielding and the number of occupied energy levels.
Atomic Radius Across a Period

- Atomic radius decreases from left to right across a period.
- This is because the number of protons increases while electrons are added to the same shell.
- Shielding remains almost constant, so the effective nuclear charge increases.
- The stronger attraction pulls the electron cloud closer to the nucleus, reducing the radius.
A-Level Exception (Across a Period)
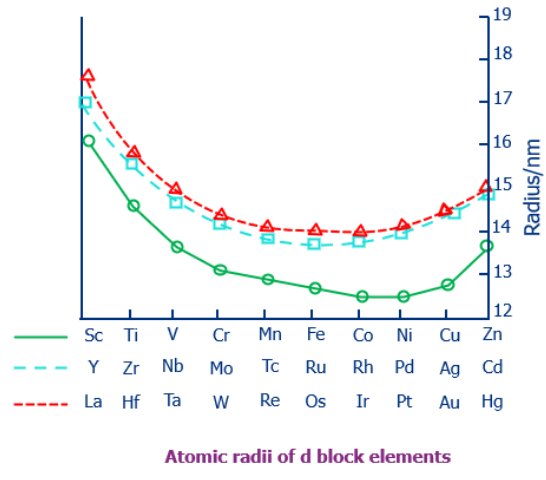
- Elements involving d-block contraction: Atomic radius does not always decrease smoothly across a period once transition metals appear.
- Across Period 4 (K to Kr), the transition metals (Sc → Zn) show a much smaller decrease in radius compared with the s- and p-block elements.
- This is because added 3d electrons provide poor shielding while being close to the nucleus, causing a slight contraction known as d-block contraction.
- As a result, Ga is smaller than expected compared with Al, and similar effects appear down Group 13.
Atomic Radius Down a Group

- Atomic radius increases down a group.
- More electron shells are added, so the outer electrons are further from the nucleus.
- Shielding increases significantly and reduces the attraction of the nucleus.
Ionic Radius Across a Period

- For metals forming cations, ionic radius decreases as nuclear charge increases while electrons are removed.
- For non metals forming anions, ionic radius decreases left to right for ions of equal charge because increased nuclear charge pulls electrons more strongly.
- A large jump occurs between cations and anions since anions have more electrons and greater repulsion.
Ionic Radius Down a Group
- Ionic radius increases down the group for both cations and anions.
- New shells increase the size and shielding decreases attraction.

Ionic radius of ions decreases in each isoelectronic series across the third period
Example
Explain why atomic radius decreases across Period 3 from sodium to chlorine.
▶️ Answer / Explanation
Proton number increases from Na to Cl. Electrons are added to the same shell, shielding stays constant and increased nuclear charge pulls electrons in more strongly.
Example
Explain why ionic radius increases from \( \mathrm{Li^+} \) to \( \mathrm{K^+} \).
▶️ Answer / Explanation
Down the group, ions have more electron shells. Increased shielding and greater distance from the nucleus increase ionic radius.
Example
Compare the radii of \( \mathrm{Mg^{2+}} \), \( \mathrm{Na^+} \) and \( \mathrm{F^-} \), which are isoelectronic.
▶️ Answer / Explanation
All have the same number of electrons but different nuclear charges.
Nuclear charge increases in the order \( \mathrm{F^-} \) (9 p) → \( \mathrm{Na^+} \) (11 p) → \( \mathrm{Mg^{2+}} \) (12 p).
Greater nuclear charge pulls electrons more strongly, so radius decreases.
Order: \( \mathrm{F^- > Na^+ > Mg^{2+}} \).
