CIE AS/A Level Chemistry 1.2 Isotopes Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 1.2 Isotopes Study Notes – New Syllabus
CIE AS/A Level Chemistry 1.2 Isotopes Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- define the term isotope in terms of numbers of protons and neutrons
- understand the notation x y A for isotopes, where x is the mass or nucleon number and y is the atomic or proton number
- state that and explain why isotopes of the same element have the same chemical properties
- state that and explain why isotopes of the same element have different physical properties, limited to mass and density
Isotopes
Isotopes are different forms of the same element that have the same number of protons but different numbers of neutrons.
![]()
- Same number of protons → same atomic number → same element.
- Different number of neutrons → different mass numbers.
For example, \( ^{12}_{6}\text{C} \) and \( ^{14}_{6}\text{C} \) are isotopes because both have 6 protons but 6 and 8 neutrons respectively.
Example
Explain why \( ^{35}_{17}\text{Cl} \) and \( ^{37}_{17}\text{Cl} \) are isotopes.
▶️ Answer / Explanation
Both atoms have 17 protons, so they are chlorine atoms.
They have different numbers of neutrons: \( 35 – 17 = 18 \) neutrons and \( 37 – 17 = 20 \) neutrons.
Same protons, different neutrons → isotopes.
Example
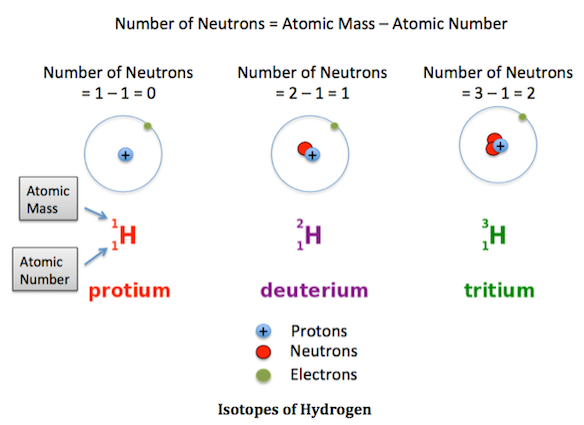
Hydrogen has three isotopes: protium \( ^1_1\text{H} \), deuterium \( ^2_1\text{H} \) and tritium \( ^3_1\text{H} \). Describe their proton and neutron numbers.
▶️ Answer / Explanation
All three have 1 proton → they are hydrogen.
Neutrons:
Protium: \( 1 – 1 = 0 \) neutrons.
Deuterium: \( 2 – 1 = 1 \) neutron.
Tritium: \( 3 – 1 = 2 \) neutrons.
They differ only in neutrons → isotopes.
Example
Two atoms of an element X are written as \( ^{62}_{28}\text{X} \) and \( ^{64}_{28}\text{X} \). Explain why they are isotopes and calculate the number of neutrons in each.
▶️ Answer / Explanation
Both atoms have 28 protons, so they are the same element.
Neutrons in \( ^{62}_{28}\text{X} \): \( 62 – 28 = 34 \).
Neutrons in \( ^{64}_{28}\text{X} \): \( 64 – 28 = 36 \).
Same protons, different neutrons → isotopes.
Notation \( ^A_Z\text{X} \) for Isotopes
Isotopes of an element are represented using the notation \( ^A_Z\text{X} \). This notation shows the mass (nucleon) number, the atomic (proton) number, and the chemical symbol of the element.
Meaning of Each Part![]()
- \( \mathrm{A} \) = mass (nucleon) number
total number of protons + neutrons in the nucleus. - \( \mathrm{Z} \) = atomic (proton) number
number of protons in the nucleus. - \( \mathrm{X} \) = chemical symbol
symbol of the element.
General Format
\( ^A_Z\text{X} \)
For example, the isotope \( ^{23}_{11}\text{Na} \) has:
- \( \mathrm{A = 23} \) → protons + neutrons = 23
- \( \mathrm{Z = 11} \) → protons = 11
- Neutrons = \( 23 – 11 = 12 \)
Example
Identify the values of A and Z in the isotope \( ^{40}_{20}\text{Ca} \).
▶️ Answer / Explanation
Mass number \( A = 40 \).
Atomic number \( Z = 20 \).
Therefore the atom has 20 protons and \( 40 – 20 = 20 \) neutrons.
Example
Explain what information is shown by the isotope notation \( ^{14}_{6}\text{C} \).
▶️ Answer / Explanation
The symbol C shows the element is carbon.
\( Z = 6 \) → it has 6 protons.
\( A = 14 \) → total nucleons = 14.
Neutrons = \( 14 – 6 = 8 \).
Example
Two isotopes of an element are written as \( ^{62}_{30}\text{X} \) and \( ^{64}_{30}\text{X} \). Use the notation to determine whether they are isotopes and find their neutron numbers.
▶️ Answer / Explanation
Both have the same atomic number \( Z = 30 \), so they are the same element.
Neutrons in \( ^{62}_{30}\text{X} = 62 – 30 = 32 \).
Neutrons in \( ^{64}_{30}\text{X} = 64 – 30 = 34 \).
Same protons, different neutrons → isotopes.
Isotopes and Their Chemical Properties
Isotopes of the same element have identical chemical properties. This is because chemical behaviour depends on electrons, not neutrons.
![]()
Why isotopes have the same chemical properties
- Isotopes of an element have the same number of protons.
- Therefore, they also have the same number of electrons in the outer shell.
- Chemical reactions involve the gain, loss or sharing of these outer-shell electrons.
- Since isotopes have identical electron arrangements, their chemical behaviour is the same.
Differences in neutron number do not affect chemical reactions — they only affect mass and nuclear stability.
Example
Explain why \( ^{35}_{17}\text{Cl} \) and \( ^{37}_{17}\text{Cl} \) react in the same way with sodium metal.
▶️ Answer / Explanation
Both isotopes have 17 electrons arranged as 2,8,7.
Chemical reactions depend on outer-shell electrons.
Since they have the same electron configuration, they both form \( \mathrm{Cl^-} \) ions and react identically with sodium.
Example
Carbon has two stable isotopes: \( ^{12}\text{C} \) and \( ^{13}\text{C} \). Explain why both isotopes undergo the same chemical reactions.
▶️ Answer / Explanation
Both isotopes have 6 protons and 6 electrons.
They share the same electron configuration: 2,4.
Because chemical reactions depend on electron structure, both isotopes behave chemically the same.
Example
Isotopes of hydrogen include \( ^1\text{H} \), \( ^2\text{H} \) and \( ^3\text{H} \). Despite their large mass differences, they all react similarly. Explain why.
▶️ Answer / Explanation
All hydrogen isotopes have 1 proton and 1 electron.
Their electron configuration is the same: 1 electron in the 1s subshell.
Chemical reactions involve the electron, not the neutrons.
Therefore, all isotopes of hydrogen show identical chemical behaviour.
Physical Properties of Isotopes
Isotopes of the same element have the same chemical properties but different physical properties. These differences arise because isotopes have different numbers of neutrons, which changes their mass.
Why isotopes have different physical properties
- Isotopes of an element have the same number of protons and electrons but different numbers of neutrons.
- A different number of neutrons means the isotopes have different mass numbers.
- Because mass affects physical behaviour, isotopes show different physical properties.
Key differences (A-level focus)
- Mass: Heavier isotopes have a greater mass because they contain more neutrons.
- Density: Density = mass ÷ volume. Since isotopes have different masses but occupy the same volume, heavier isotopes have higher densities.
These differences do not affect chemical reactions because chemistry depends on electrons, not on mass.
Example
Explain why \( ^{35}\text{Cl} \) and \( ^{37}\text{Cl} \) have different densities.
▶️ Answer / Explanation
Both isotopes of chlorine occupy the same volume because they have the same electron arrangement.
However, \( ^{37}\text{Cl} \) has two more neutrons and therefore a greater mass.
Since density = mass ÷ volume, the heavier isotope has a higher density.
Example
Hydrogen has isotopes \( ^1\text{H} \), \( ^2\text{H} \) and \( ^3\text{H} \). State why they have different masses.
▶️ Answer / Explanation
All three isotopes have 1 proton, but they contain 0, 1 and 2 neutrons respectively.
More neutrons increase the mass, so \( ^3\text{H} \) is the heaviest and \( ^1\text{H} \) the lightest.
Example
Two isotopes of an element X are \( ^{62}\text{X} \) and \( ^{64}\text{X} \). Explain why one has a higher density than the other.
▶️ Answer / Explanation
Both isotopes occupy the same volume because they have identical electron arrangements.
However, \( ^{64}\text{X} \) has two more neutrons, so its mass is greater.
Since density depends on mass and they have equal volume, \( ^{64}\text{X} \) has the higher density.
