CIE AS/A Level Chemistry 1.3 Electrons, energy levels and atomic orbitals Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 1.3 Electrons, energy levels and atomic orbitals Study Notes – New Syllabus
CIE AS/A Level Chemistry 1.3 Electrons, energy levels and atomic orbitals Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- understand the terms:
• shells, sub-shells and orbitals
• principal quantum number (n)
• ground state, limited to electronic configuration - describe the number of orbitals making up s, p and d sub-shells, and the number of electrons that can fill s, p and d sub-shells
- describe the order of increasing energy of the sub-shells within the first three shells and the 4s and 4p sub-shells
- describe the electronic configurations to include the number of electrons in each shell, sub-shell and orbital
- explain the electronic configurations in terms of energy of the electrons and inter-electron repulsion
- determine the electronic configuration of atoms and ions given the atomic or proton number and charge, using either of the following conventions: e.g. for Fe: 1s2 2s2 2p6 3s2 3p6 3d6 4s2 (full electronic configuration) or [Ar] 3d6 4s2 (shorthand electronic configuration)
- understand and use the electrons in boxes notation e.g. for Fe: [Ar]
- describe and sketch the shapes of s and p orbitals
- describe a free radical as a species with one or more unpaired electrons
Electronic Structure — Shells, Sub-shells, Orbitals & Quantum Numbers
Electrons in atoms are arranged in different energy levels (shells). Each shell contains sub-shells, and each sub-shell contains orbitals — regions where electrons are most likely found.
\( \text{Electron arrangement} = \text{shells} \rightarrow \text{sub-shells} \rightarrow \text{orbitals} \)
- Shells are main energy levels labelled by the principal quantum number \( n \).
- Sub-shells are divisions of each shell (s, p, d, f), with slightly different energies.
- Orbitals are regions of space that hold up to 2 electrons.
Shells (Main Energy Levels)
Shells correspond to values of the principal quantum number \( n = 1,2,3,4\ldots \). The higher the value of \( n \), the higher the energy and the further the electron is from the nucleus.
- Maximum electrons a shell can hold = \( 2n^2 \).
- Examples: \( n=1 \to 2 \) electrons, \( n=2 \to 8 \) electrons, \( n=3 \to 18 \) electrons.
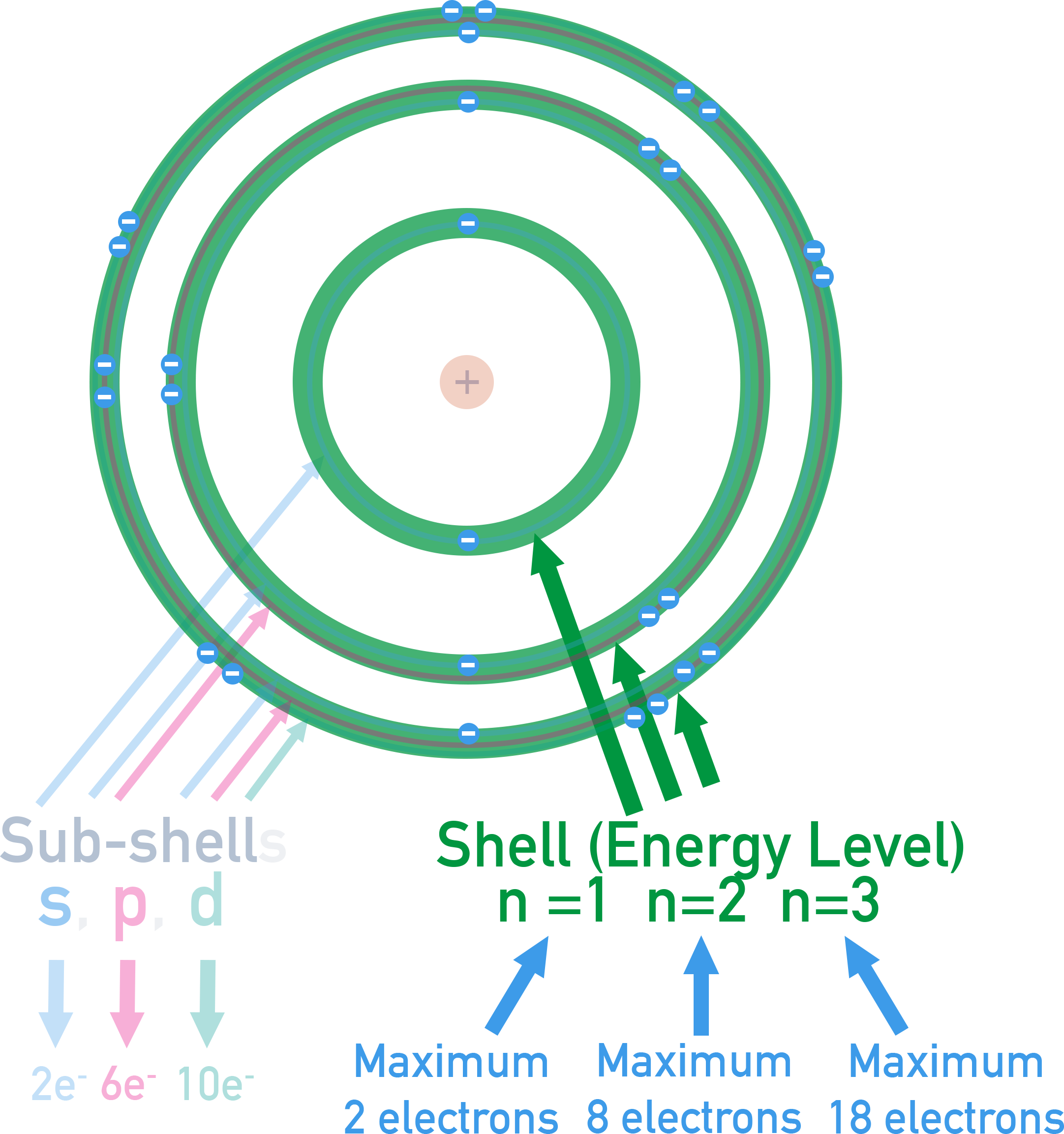
Sub-shells (s, p, d, f)
Each shell contains sub-shells of different shapes and energies.

| Sub-shell | No. of orbitals | Maximum electrons | Shape |
|---|---|---|---|
| s | 1 | 2 | Spherical |
| p | 3 | 6 | Dumbbell |
| d | 5 | 10 | Complex |
Orbitals

- An orbital is a region of space with a high probability of finding an electron.
- Each orbital holds a maximum of 2 electrons with opposite spin.
- Examples: 1s orbital, the three 2p orbitals (px, py, pz), five 3d orbitals.
Principal Quantum Number (n)
The principal quantum number \( n \) indicates:
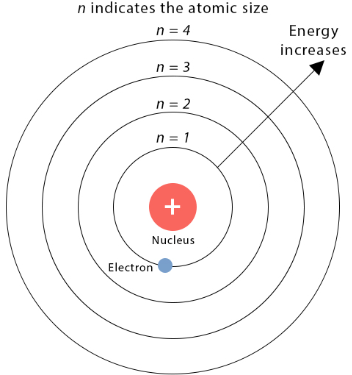
- the main energy level of the electron
- the distance of the electron from the nucleus
- the overall energy of the electron (larger \( n \) = higher energy)
Values: \( n = 1,2,3,4,… \)
Ground State Electronic Configuration
The ground state is the lowest-energy arrangement of electrons.

Electron configurations follow three rules:
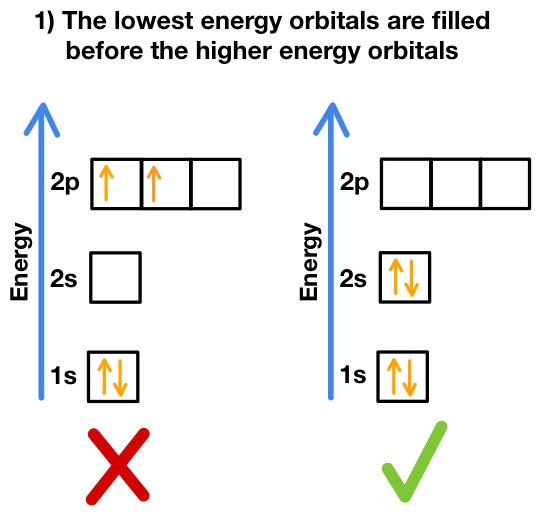
- Aufbau Principle: electrons fill the lowest-energy sub-shells first (1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p …).
- Pauli Exclusion Principle: each orbital holds max 2 electrons with opposite spins.
- Hund’s Rule: electrons fill orbitals singly before pairing in the same sub-shell.

Example: Carbon (6 electrons) → 1s² 2s² 2p²
Example
How many electrons can the n = 2 shell hold?
▶️ Answer / Explanation
Using \( 2n^2 \): \( 2 \to 2(2)^2 = 8 \) electrons.
Example
How many orbitals are in the 3p sub-shell?
▶️ Answer / Explanation
The p sub-shell always contains 3 orbitals, so 3p has 3 orbitals.
Example
Write the ground-state electron configuration for phosphorus (Z = 15) and explain the arrangement of the 3p electrons.
▶️ Answer / Explanation
Phosphorus has 15 electrons. Configuration: 1s² 2s² 2p⁶ 3s² 3p³.
According to Hund’s Rule, the 3 electrons in the 3p sub-shell will occupy the three p orbitals singly with parallel spins.
Sub-shells and Orbitals — s, p and d
Each sub-shell (s, p or d) is made up of a specific number of orbitals. Since each orbital holds a maximum of 2 electrons, the number of orbitals determines how many electrons the sub-shell can contain.

- s sub-shell → 1 orbital → maximum of 2 electrons
- p sub-shell → 3 orbitals → maximum of 6 electrons
- d sub-shell → 5 orbitals → maximum of 10 electrons
These numbers arise from the allowed orientations of orbitals in 3D space.
Table: Number of Orbitals & Maximum Electrons
| Sub-shell | No. of orbitals | Maximum electrons |
|---|---|---|
| s | 1 | 2 |
| p | 3 | 6 |
| d | 5 | 10 |
Example
How many electrons can a p sub-shell hold?
▶️ Answer / Explanation
A p sub-shell contains 3 orbitals. Each orbital holds 2 electrons, so the p sub-shell can hold a maximum of 6 electrons.
Example (Medium)
How many orbitals are there in the n = 3 shell?
▶️ Answer / Explanation
For n = 3, the sub-shells are 3s, 3p and 3d.
3s → 1 orbital
3p → 3 orbitals
3d → 5 orbitals
Total orbitals = 1 + 3 + 5 = 9.
Example
A transition metal atom has electrons filling the 3d sub-shell. Explain why the maximum number of electrons it can contain is 10.
▶️ Answer / Explanation
The d sub-shell contains 5 orbitals. Each orbital can hold 2 electrons with opposite spins, according to the Pauli Exclusion Principle.
Thus, maximum electrons = 5 orbitals × 2 electrons = 10 electrons.
Energy Order of Sub-shells — First Three Shells + 4s and 4p
Sub-shells within an atom do not all have the same energy. Even within the same shell, the order of energy is: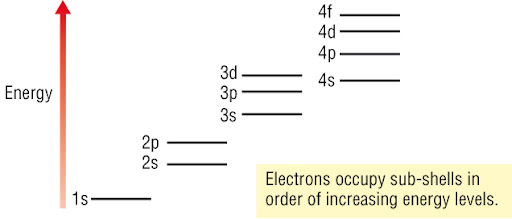
Lowest energy → Highest energy
s < p < d < f
However, the actual order of energies across shells is not simply by shell number. The correct order (up to 4p) is:
Order of Increasing Energy (Up to 4p)
The sub-shells fill in the following sequence of increasing energy:
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p
Notice two important anomalies:
- 4s is lower in energy than 3d, so 4s fills before 3d.
- 3d is higher in energy than 4s but lower than 4p.
Energy Order Within Each of the First Three Shells
| Shell (n) | Sub-shells present | Energy Order (Low → High) |
|---|---|---|
| n = 1 | 1s | 1s (only) |
| n = 2 | 2s, 2p | 2s < 2p |
| n = 3 | 3s, 3p, 3d | 3s < 3p < 3d (but note: 4s comes before 3d when filling) |
Key Points
- Within a shell: s < p < d in energy.
- 4s is unexpectedly lower in energy than 3d → fills first.
- Order of filling (up to 4p): 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p
Example
Which sub-shell has lower energy: 3p or 3d?
▶️ Answer / Explanation
Energy order within n = 3 is: 3s < 3p < 3d. So, 3p has lower energy than 3d.
Example
Which fills first: 4s or 3d? Explain why.
▶️ Answer / Explanation
4s fills before 3d because the 4s sub-shell has slightly lower energy when empty. Thus, electrons enter 4s before 3d during ground-state configuration.
Example
Arrange the following sub-shells in order of increasing energy: 3p, 4s, 2p, 3d, 3s.
▶️ Answer / Explanation
Following the filling order: 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p
The required order is:
2p < 3s < 3p < 4s < 3d
Electronic Configurations — Electrons in Shells, Sub-shells and Orbitals
Electronic configuration is the way electrons are arranged in shells, sub-shells and orbitals in an atom. Electrons fill the available energy levels following the Aufbau Principle, Pauli Exclusion Principle and Hund’s Rule.
Shell → Sub-shell → Orbital → Electrons
- Shells are labelled by principal quantum number \( n \).
- Sub-shells are divisions of shells (s, p, d, f).
- Orbitals are regions holding up to 2 electrons.
Number of Electrons in Shells
Maximum electrons in a shell = \( 2n^2 \).
| Shell (n) | Maximum electrons |
|---|---|
| n = 1 | 2 |
| n = 2 | 8 |
| n = 3 | 18 |
| n = 4 | 32 |
Electrons in Each Sub-shell
Each sub-shell contains a fixed number of orbitals.
| Sub-shell | No. of orbitals | Electrons per orbital | Maximum electrons |
|---|---|---|---|
| s | 1 | 2 | 2 |
| p | 3 | 2 | 6 |
| d | 5 | 2 | 10 |
| f | 7 | 2 | 14 |
Electrons in Each Orbital
- Every orbital can hold a maximum of 2 electrons.
- The two electrons in an orbital must have opposite spins (Pauli Exclusion Principle).
- In p and d sub-shells, electrons fill orbitals singly first (Hund’s Rule).
Writing Electronic Configurations
An electronic configuration shows how many electrons are in each sub-shell. For example:
- 1s² → 2 electrons in the 1s sub-shell
- 2p⁶ → 6 electrons in the 2p sub-shell (fully filled)
- 3d⁵ → 5 electrons in the 3d sub-shell
Electrons fill according to increasing energy:
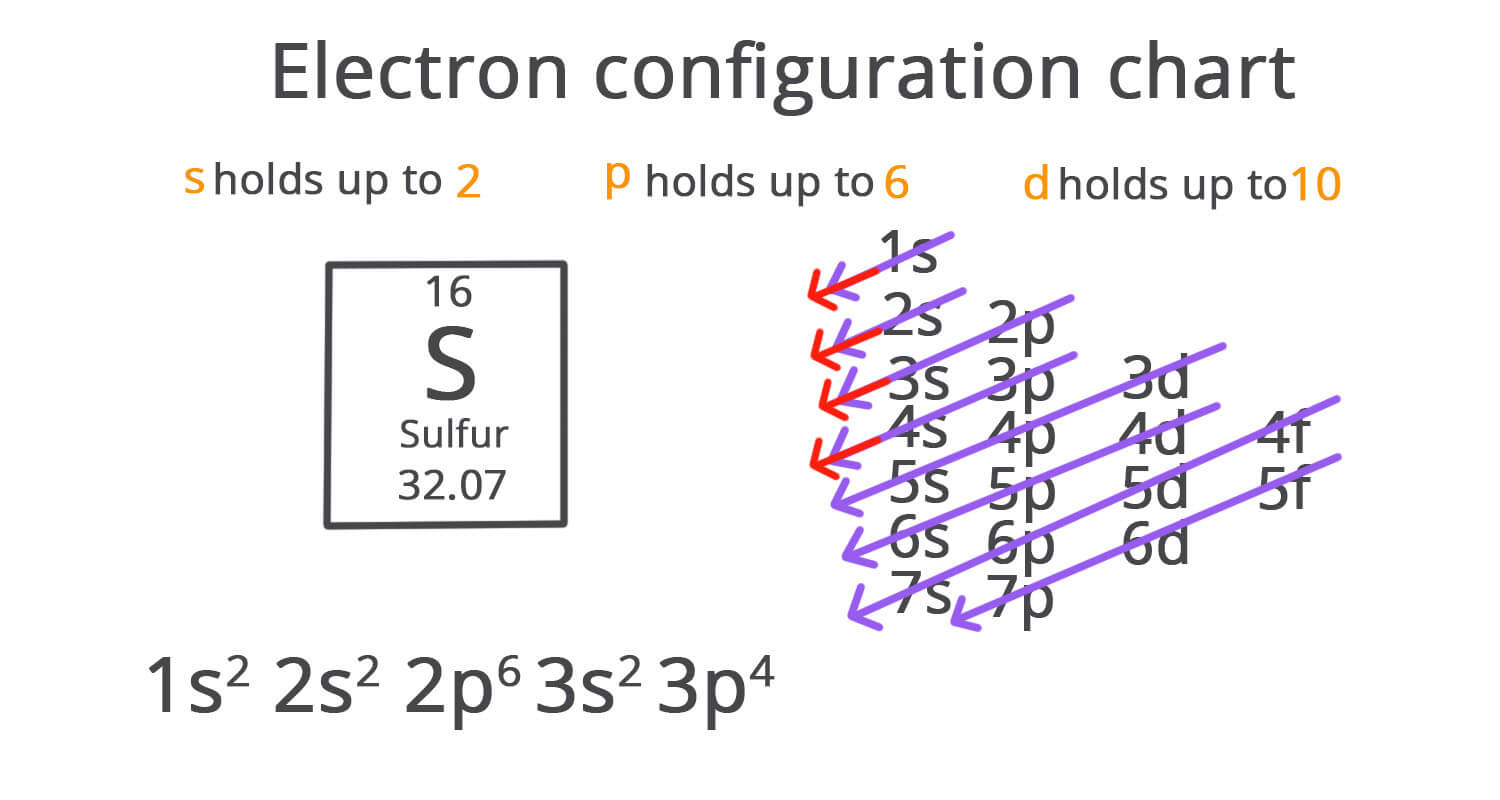
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p
Example
How many electrons can the 3d sub-shell hold?
▶️ Answer / Explanation
The d sub-shell contains 5 orbitals. Each holds 2 electrons. Maximum electrons = 5 × 2 = 10 electrons.
Example
Write the electronic configuration for nitrogen (Z = 7).
▶️ Answer / Explanation
Nitrogen has 7 electrons. Filling order: 1s → 2s → 2p
Configuration: 1s² 2s² 2p³
In 2p, electrons fill 3 separate orbitals singly (Hund’s Rule).
Example
How many electrons are present in all orbitals of the n = 3 shell?
▶️ Answer / Explanation
Sub-shells in n = 3: 3s, 3p, 3d
3s: 1 orbital → 2 electrons 3p: 3 orbitals → 6 electrons 3d: 5 orbitals → 10 electrons
Total electrons = 2 + 6 + 10 = 18 electrons
Electronic Configurations — Energy of Electrons & Inter-Electron Repulsion
Electronic configurations are determined by the energies of electrons in different sub-shells and by the repulsions between electrons. The arrangement adopted is always the one with the lowest possible total energy for the atom.
Electron energies + Electron–electron repulsion → determines the configuration
1. Energy of Electrons in Different Sub-shells
Electrons occupy sub-shells of different energies. Even within the same shell, s, p and d have different energy levels:

- s sub-shell has the lowest energy
- p sub-shell is higher in energy
- d sub-shell is higher still
Electrons fill the lowest-energy sub-shells first because this gives the atom the most stable (lowest-energy) arrangement — the Aufbau Principle.
2. Energy Differences Between 4s and 3d
The 4s sub-shell lies slightly lower in energy than the 3d sub-shell when both are empty. Therefore:

4s fills before 3d.
However, once electrons start filling 3d, the repulsions and shielding change the energies so that:
3d becomes lower in energy than 4s when occupied.
This is why transition metal ions lose 4s electrons before 3d electrons.
3. Inter-Electron Repulsion
Electrons repel each other because they have the same negative charge. This affects how they arrange themselves in orbitals:

- Paired electrons in the same orbital repel strongly.
- Unpaired electrons in separate orbitals repel less.
Because pairing increases repulsion (and therefore energy), electrons prefer to stay unpaired for as long as possible — Hund’s Rule.
4. Hund’s Rule — Minimising Repulsion
Within a sub-shell (such as p or d), electrons fill each orbital singly before pairing. This arrangement gives:

- Lower electron–electron repulsion
- Lower energy for the atom
- Greater stability
Example: In nitrogen (1s² 2s² 2p³), the three 2p electrons occupy three different orbitals, not the same one.
5. Pauli Exclusion Principle — Limiting Paired Electrons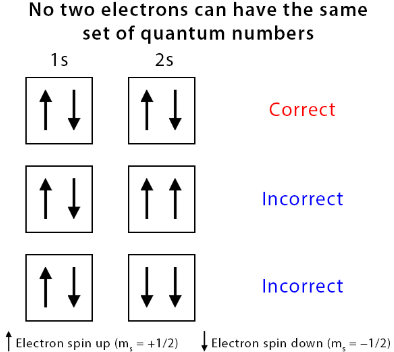
Each orbital can hold only 2 electrons, and they must have opposite spins. Opposite spins slightly reduce repulsion, giving a more stable arrangement.
Electrons only pair up when there is no empty orbital available in the same sub-shell.
Why Electronic Configurations Are the Way They Are
- Electrons occupy lowest-energy sub-shells first to minimise total energy.
- Electrons stay unpaired where possible to reduce repulsion.
- Pairing increases electron–electron repulsion and raises energy.
- Real configurations result from a balance between sub-shell energies and inter-electron repulsion.
Example
Why do electrons fill 2p orbitals singly before pairing?
▶️ Answer / Explanation
Electrons repel each other. If they occupy different orbitals, repulsion is minimised and energy is lower. Pairing increases repulsion, so electrons stay unpaired as long as possible (Hund’s Rule).
Example
Explain why 4s fills before 3d even though both belong to different shells.
▶️ Answer / Explanation
The empty 4s orbital is slightly lower in energy than the empty 3d orbitals. Electrons always occupy the lowest-energy available sub-shell, so 4s fills before 3d.
Example
Explain why chromium has the configuration 3d⁵ 4s¹ instead of 3d⁴ 4s².
▶️ Answer / Explanation
3d⁵ 4s¹ gives chromium a half-filled d sub-shell, which drastically reduces inter-electron repulsion. A half-filled 3d (five unpaired electrons) is more stable than 3d⁴ 4s², where electrons must pair up.
Lower repulsion → lower energy → more stable configuration.
Electronic Configurations of Atoms and Ions
Electronic configurations show how electrons are arranged in shells, sub-shells and orbitals. From the atomic (proton) number and the charge, you can work out the total number of electrons and then assign them to the correct sub-shells in order of increasing energy.
1. Find number of electrons → 2. Fill sub-shells in energy order → 3. Use full or shorthand notation
1. Determine Number of Electrons
- Atom: number of electrons = proton number (atomic number)
- Cation (+ charge): electrons = proton number − charge • e.g., Fe²⁺ → 26 − 2 = 24 electrons
- Anion (− charge): electrons = proton number + charge • e.g., O²⁻ → 8 + 2 = 10 electrons
2. Fill Sub-shells in Energy Order
Electrons fill the lowest-energy available sub-shells first:
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → …
For transition metals: 4s fills before 3d, but 4s empties before 3d when forming ions.
3. Writing Electronic Configurations
Full configuration: lists every sub-shell
e.g. Fe: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁶ 4s²
Shorthand (noble gas) configuration: replace inner electrons with nearest noble gas in brackets.
e.g. Fe: [Ar] 3d⁶ 4s²
Example : Sodium atom, Na (Z = 11)
Find: electronic configuration of Na.
▶️ Answer / Explanation
Electrons = 11
Configuration: 1s² 2s² 2p⁶ 3s¹
Shorthand: [Ne] 3s¹
Example : Sulphide ion, S²⁻ (Z = 16)
Find: electronic configuration of S²⁻.
▶️ Answer / Explanation
S has 16 protons. Charge = 2− → add 2 electrons.
Total electrons = 16 + 2 = 18
Configuration: 1s² 2s² 2p⁶ 3s² 3p⁶
Shorthand: [Ar]
Example : Iron(III) ion, Fe³⁺ (Z = 26)
Find: electronic configuration of Fe³⁺.
▶️ Answer / Explanation
Electrons in Fe atom = 26
Fe³⁺ → remove 3 electrons
Remove from 4s first, then 3d
Start: [Ar] 3d⁶ 4s²
Remove 2 from 4s → [Ar] 3d⁶
Remove 1 from 3d → [Ar] 3d⁵
Final: [Ar] 3d⁵
Full: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁵
Example : Copper atom, Cu (Z = 29)
Find: electronic configuration of Cu.
▶️ Answer / Explanation
Expected: [Ar] 3d⁹ 4s²
Actual: [Ar] 3d¹⁰ 4s¹
Reason: A full 3d¹⁰ sub-shell is more stable due to lower repulsion.
Example : Titanium(IV), Ti⁴⁺ (Z = 22)
Find: electronic configuration of Ti⁴⁺.
▶️ Answer / Explanation
Electrons in Ti atom = 22
Ti⁴⁺ → remove 4 electrons
Start: [Ar] 3d² 4s²
Remove 2 from 4s → [Ar] 3d²
Remove 2 from 3d → [Ar]
Final: [Ar]
Electrons-in-Boxes Notation
Electrons-in-boxes notation (also called orbital box notation) shows how electrons occupy individual orbitals within a sub-shell. It helps visualise:
- pairing of electrons

- unpaired electrons
- Hund’s Rule (electrons occupy orbitals singly first)
- opposite spins in an orbital
1. Structure of Electrons-in-Boxes Notation
Each orbital is represented by a box:
Electrons are shown as arrows:
- ↑ or ↓ represent electrons with opposite spin
- ↑↓ in the same box = paired electrons
Sub-shells have: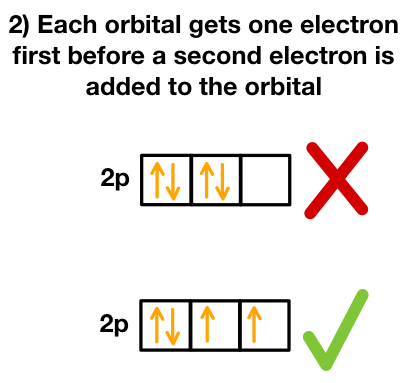
- s: 1 box
- p: 3 boxes
- d: 5 boxes
2. Hund’s Rule in Electrons-in-Boxes Notation
Electrons fill orbitals singly before pairing up. This reduces repulsion between electrons and lowers the atom’s energy.
Correct (Hund’s Rule): ↑ ↑ ↑
Incorrect: ↑↓ ↑
3. Opposite Spins (Pauli Exclusion Principle)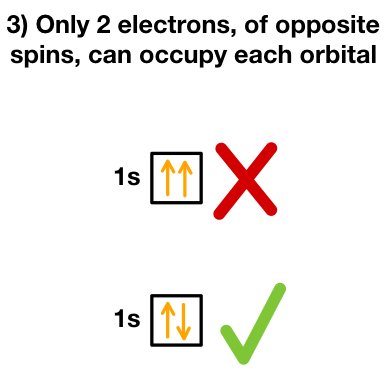
Two electrons in the same orbital must have opposite spins:
↑↓ (never ↑↑ or ↓↓)
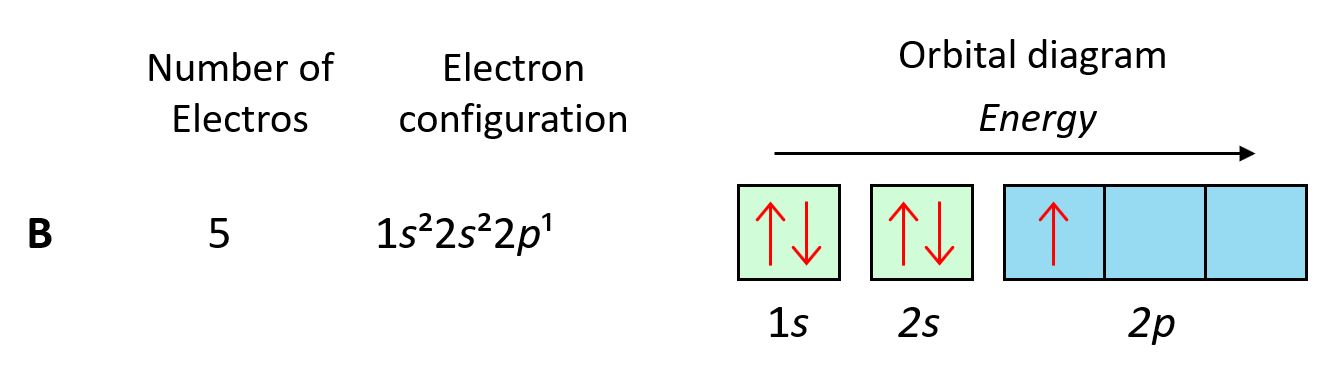
Example
Draw the electrons-in-boxes notation for 1s².
▶️ Answer / Explanation
1s has 1 orbital → 1 box.
2 electrons → paired.
1s: □ → ↑↓
Example
Draw the electrons-in-boxes notation for 2p³.
▶️ Answer / Explanation
p sub-shell has 3 orbitals → 3 boxes.
Three electrons fill singly (Hund’s Rule):
2p: □ □ □ → ↑ ↑ ↑
Example
Draw electrons-in-boxes notation for 2p⁶.
▶️ Answer / Explanation
6 electrons → all p orbitals fully filled.
2p: □ □ □ → ↑↓ ↑↓ ↑↓
Example
Draw electrons-in-boxes notation for 3d⁵.
▶️ Answer / Explanation
d sub-shell has 5 orbitals → 5 boxes.
Five electrons fill singly first:
3d: □ □ □ □ □ → ↑ ↑ ↑ ↑ ↑
Example
Use electrons-in-boxes notation for oxygen (2p⁴).
▶️ Answer / Explanation
Four electrons → fill singly, then pair one.
2p: □ □ □ → ↑↓ ↑ ↑
Shapes of s and p Orbitals
Orbitals are regions of space around the nucleus where there is a high probability of finding an electron. Each orbital has a specific three-dimensional shape, which affects how atoms bond and interact.
Orbitals → hold up to 2 electrons → shapes depend on sub-shell (s or p)
1. Shape of the s Orbital
- All s orbitals (1s, 2s, 3s…) are spherical in shape.
- The sphere becomes larger for higher energy levels (e.g., 2s is larger than 1s).
- Electrons are found with equal probability in all directions at a given distance from the nucleus.

2. Shape of the p Orbitals
- Each p sub-shell has three p orbitals: px, py, pz.
- All p orbitals have a dumbbell shape (two lobes).
- Each p orbital is oriented at 90° to the others along the x, y and z axes.
- There is a node (region of zero probability) at the nucleus.

Summary Table
| Orbital type | No. of orbitals | Shape | Orientation |
|---|---|---|---|
| s | 1 | Spherical | Not directional |
| p | 3 | Dumbbell | Along x, y and z axes |
Example
How many p orbitals are in a p sub-shell, and how are they oriented?
▶️ Answer / Explanation
A p sub-shell contains 3 orbitals. They are oriented at right angles along the x, y and z axes (px, py, pz).
Example
Describe the shape of an s orbital.
▶️ Answer / Explanation
s orbitals are spherical and have no directional orientation. The probability of finding the electron depends only on distance from the nucleus, not direction.
Example
Why do p orbitals have a dumbbell shape?
▶️ Answer / Explanation
The dumbbell shape results from the mathematical solutions to the Schrödinger equation. These solutions produce two lobes separated by a nodal plane through the nucleus.
Free Radicals — Definition & Electron Structure
A free radical is a highly reactive chemical species that contains one or more unpaired electrons in its outer shell. The presence of an unpaired electron makes free radicals unstable and very reactive.
Free radical = species with unpaired electron(s) → very reactive
Characteristics of Free Radicals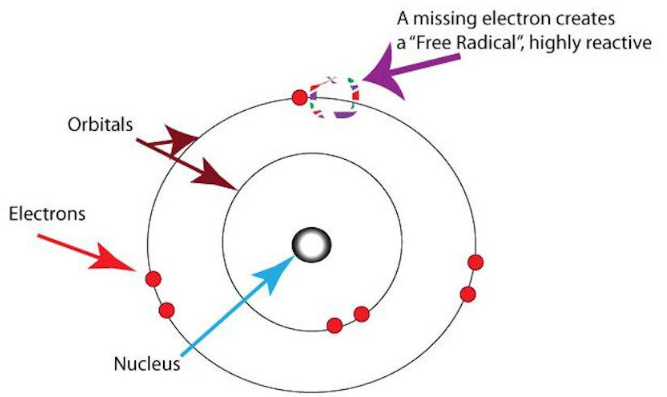
- Contain at least one unpaired electron in an orbital.
- Neutral species (not necessarily ions).
- Extremely reactive because they seek to pair their unpaired electron.
- Often formed by homolytic bond fission, where each atom takes one electron from a bond.
- Represented using a dot “·”, e.g., Cl·
Formation of Free Radicals
Free radicals are commonly formed when a covalent bond breaks evenly (homolytic fission):
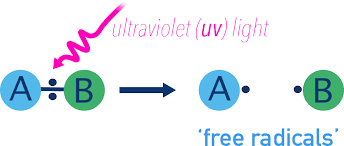
Examples of Free Radicals
- Cl· (chlorine radical)
- OH· (hydroxyl radical)
- CH₃· (methyl radical)
Example
Define a free radical.
▶️ Answer / Explanation
A free radical is a species with one or more unpaired electrons. These unpaired electrons make the radical highly reactive.
Example
Explain why Cl· is more reactive than Cl₂.
▶️ Answer / Explanation
Cl· contains an unpaired electron, making it unstable and extremely reactive. Cl₂ has all electrons paired, so it is far more stable.
Example
Does the methyl radical (CH₃·) contain an unpaired electron? Explain.
▶️ Answer / Explanation
Yes. In CH₃·, one electron on the carbon is unpaired. This unpaired electron makes CH₃· a free radical.