CIE AS/A Level Chemistry 1.4 Ionisation energy Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 1.4 Ionisation energy Study Notes – New Syllabus
CIE AS/A Level Chemistry 1.4 Ionisation energy Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- define and use the term first ionisation energy, IE
- construct equations to represent first, second and subsequent ionisation energies
- identify and explain the trends in ionisation energies across a period and down a group of the Periodic Table
- identify and explain the variation in successive ionisation energies of an element
- understand that ionisation energies are due to the attraction between the nucleus and the outer electron
- explain the factors influencing the ionisation energies of elements in terms of nuclear charge, atomic/ionic radius, shielding by inner shells and sub-shells and spin-pair repulsion
- deduce the electronic configurations of elements using successive ionisation energy data
- deduce the position of an element in the Periodic Table using successive ionisation energy data
First Ionisation Energy (IE)
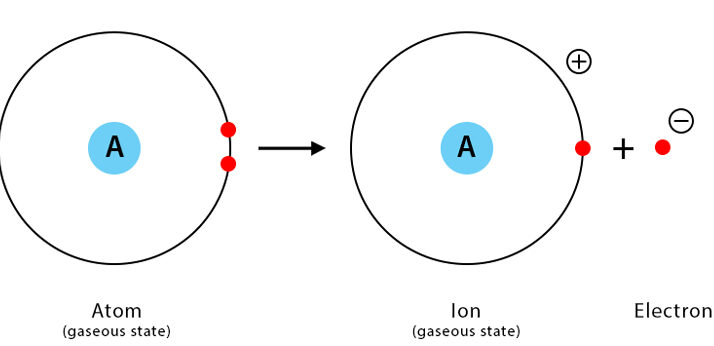
The first ionisation energy is the energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous singly charged positive ions.
\( \mathrm{X(g) \rightarrow X^+(g) + e^-} \)
Key points
- The atom must be in the gaseous state.
- Only one electron is removed from each atom.
- Energy is always required (endothermic process) because electrons are attracted to the positive nucleus.
Example
Write an equation to represent the first ionisation energy of magnesium.
▶️ Answer / Explanation
\( \mathrm{Mg(g) \rightarrow Mg^+(g) + e^-} \)
One electron is removed from each gaseous magnesium atom to produce a \( \mathrm{Mg^+} \) ion.
Example
Explain why the first ionisation energy of sodium is lower than that of magnesium.
▶️ Answer / Explanation
Magnesium has a higher nuclear charge (12 protons vs 11 in sodium).
The outer electron in magnesium is more strongly attracted to the nucleus.
Therefore, more energy is needed to remove it → higher IE.
Example
Write equations for the first and second ionisation energies of aluminium and explain why the second ionisation energy is higher.
▶️ Answer / Explanation
First IE:
\( \mathrm{Al(g) \rightarrow Al^+(g) + e^-} \)
Second IE:
\( \mathrm{Al^+(g) \rightarrow Al^{2+}(g) + e^-} \)
After the first electron is removed, the ion becomes \( \mathrm{Al^+} \), which has a higher positive charge and holds its remaining electrons more tightly.
Therefore, the second ionisation energy is higher than the first.
Equations for First, Second and Subsequent Ionisation Energies
Ionisation energies are always written for atoms or ions in the gaseous state. Each successive ionisation removes one more electron, producing a more highly charged positive ion.
General forms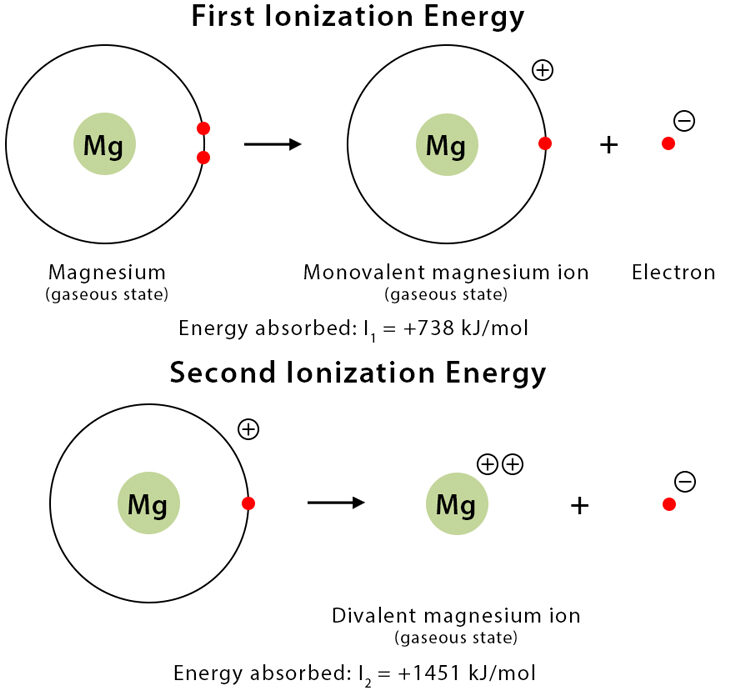
First ionisation energy : removal of one electron from a gaseous atom:
\( \mathrm{X(g) \rightarrow X^+(g) + e^-} \)
Second ionisation energy :removal of an electron from a singly charged ion:
\( \mathrm{X^+(g) \rightarrow X^{2+}(g) + e^-} \)
Third ionisation energy :removal of an electron from a doubly charged ion:
\( \mathrm{X^{2+}(g) \rightarrow X^{3+}(g) + e^-} \)
And so on for subsequent stages.
Example
Write equations for the first and second ionisation energies of magnesium.
▶️ Answer / Explanation
First IE:
\( \mathrm{Mg(g) \rightarrow Mg^+(g) + e^-} \)
Second IE:
\( \mathrm{Mg^+(g) \rightarrow Mg^{2+}(g) + e^-} \)
Each step removes one electron and the species must be gaseous.
Example
Construct equations for the first three ionisation energies of aluminium.
▶️ Answer / Explanation
First IE:
\( \mathrm{Al(g) \rightarrow Al^+(g) + e^-} \)
Second IE:
\( \mathrm{Al^+(g) \rightarrow Al^{2+}(g) + e^-} \)
Third IE:
\( \mathrm{Al^{2+}(g) \rightarrow Al^{3+}(g) + e^-} \)
Aluminium can lose three electrons to form \( \mathrm{Al^{3+}} \).
Example
Titanium can form ions up to \( \mathrm{Ti^{4+}} \). Write equations for the first four ionisation energies of titanium.
▶️ Answer / Explanation
First IE:
\( \mathrm{Ti(g) \rightarrow Ti^+(g) + e^-} \)
Second IE:
\( \mathrm{Ti^+(g) \rightarrow Ti^{2+}(g) + e^-} \)
Third IE:
\( \mathrm{Ti^{2+}(g) \rightarrow Ti^{3+}(g) + e^-} \)
Fourth IE:
\( \mathrm{Ti^{3+}(g) \rightarrow Ti^{4+}(g) + e^-} \)
Each successive electron requires more energy because the ion becomes more positively charged.
Trends in Ionisation Energies Across a Period and Down a Group
Ionisation energy (IE) changes in predictable ways across periods and down groups of the Periodic Table. These trends can be explained using nuclear charge, atomic radius and electron shielding.
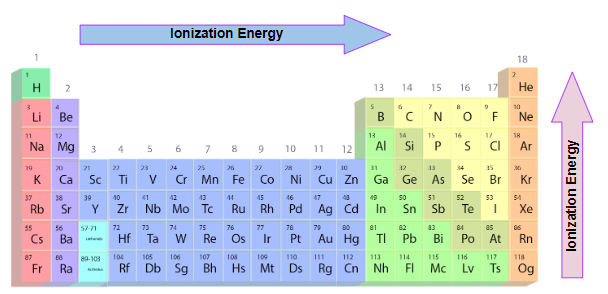
Trend Across a Period (left → right)
- Ionisation energy generally increases across a period.
- Nuclear charge increases (more protons).
- Atomic radius decreases, so the outer electrons are held more tightly.
- Shielding remains roughly constant because electrons are added to the same shell.
- Result: electrons are harder to remove → higher IE.
Exceptions across a period
- Drop between Group 2 → Group 13
Outer electron enters a higher-energy p subshell, slightly further from nucleus → easier to remove.
- Drop between Group 15 → Group 16
Electron pairing occurs in the p subshell → repulsion makes the paired electron easier to remove.
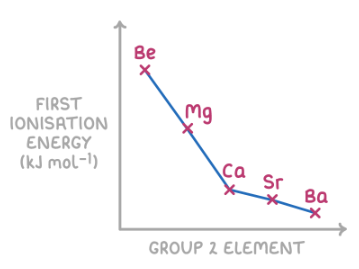
Trend Down a Group (top → bottom)
- Ionisation energy generally decreases down a group.
- Atomic radius increases (more electron shells).
- Shielding increases due to more inner shells.
- Outermost electrons are further from the nucleus and less strongly attracted.
- Result: electrons are easier to remove → lower IE.
Example
Why is the first ionisation energy of sulfur slightly lower than that of phosphorus?
▶️ Answer / Explanation
In phosphorus, the three p electrons are unpaired. In sulfur, one p orbital contains a pair of electrons.
Electron-electron repulsion in the paired orbital of sulfur makes it easier to remove an electron.
Therefore sulfur has a lower IE.
Example
Explain why the first ionisation energy of magnesium is higher than that of sodium.
▶️ Answer / Explanation
Magnesium has a higher nuclear charge (12 protons vs 11 in sodium).
The outer electron in magnesium is held more strongly and is harder to remove.
Therefore Mg has a higher IE.
Example
Explain why ionisation energy decreases from fluorine to iodine.
▶️ Answer / Explanation
Down Group 17, atoms gain more electron shells.
This increases shielding and increases atomic radius.
The outer electron becomes less strongly attracted to the nucleus.
Therefore ionisation energy decreases from F → Cl → Br → I.
Variation in Successive Ionisation Energies
Successive ionisation energies refer to the energies required to remove electrons one by one from the same atom. These values always increase and show characteristic jumps that provide important information about electronic structure.
General Pattern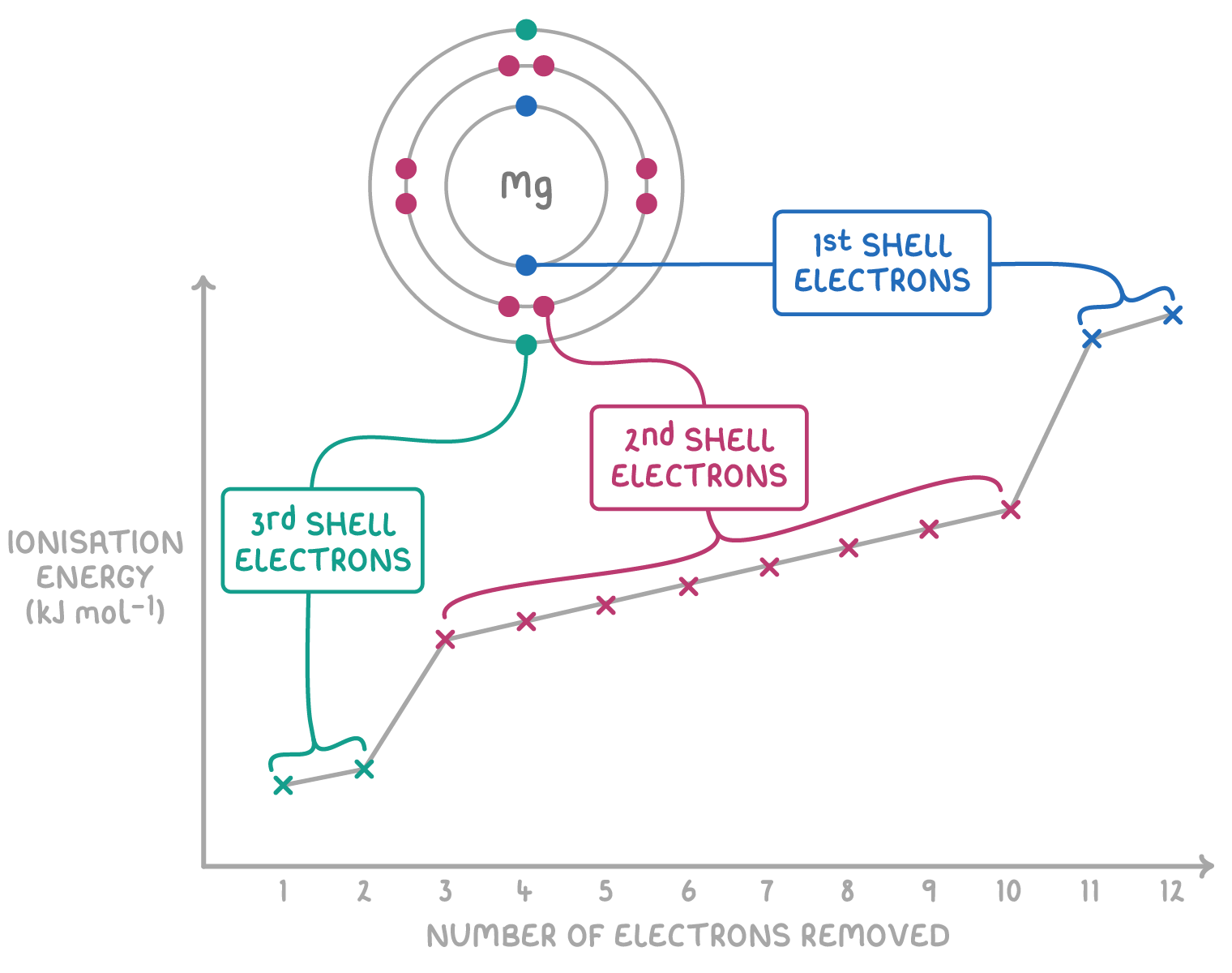
- Successive ionisation energies always increase.
- Each time an electron is removed, the ion becomes more positively charged.
- Remaining electrons are held more strongly, so more energy is required.
Large Jumps and What They Mean
- A large jump in successive IE values indicates that a new inner electron shell is being broken into.
- Electrons closer to the nucleus are much harder to remove because they experience stronger nuclear attraction and less shielding.
- The number of electrons removed before the large jump indicates the number of outer-shell electrons → identifies the group.
Example of the pattern
Small increases → removing electrons from same shell.
Huge jump → removing an electron from a shell closer to nucleus.
Example
The first and second ionisation energies of magnesium are relatively low, but the third is extremely high. Explain why.
▶️ Answer / Explanation
Magnesium has the electron configuration 2,8,2.
The first two IE values correspond to removing the two outer-shell electrons (easier to remove).
The third IE removes an electron from a full inner shell, which is much closer to the nucleus and more strongly attracted.
Therefore the third IE is extremely high.
Example
An element has the following successive ionisation energies (in arbitrary units):
IE₁ = 500, IE₂ = 1250, IE₃ = 2400, IE₄ = 3900, IE₅ = 25 000.
Identify the group of the element.
▶️ Answer / Explanation
The first four ionisation energies gradually increase.
A huge jump occurs after IE₄ (from 3900 to 25 000).
This means the fifth electron is being removed from a new inner shell.
Therefore, the atom has 4 outer-shell electrons → Group 14.
Example
Successive ionisation energies of an element are:
IE₁ = 762, IE₂ = 1580, IE₃ = 3230, IE₄ = 4350, IE₅ = 16 100, IE₆ = 19 500.
Use the data to deduce the group of the element and explain your reasoning.
▶️ Answer / Explanation
There is a large jump between IE₄ (4350) and IE₅ (16 100).
This means the first four electrons removed were in the outer shell.
The fifth electron belongs to a new inner shell, which is much harder to remove.
Therefore the element has 4 outer electrons → it is in Group 14.
Electronic Configurations in Terms of Energy and Electron–Electron Repulsion
The arrangement of electrons in atoms is governed by their energies and by repulsion between electrons. These two ideas explain the order in which electrons fill shells, subshells and orbitals.
1. Electrons fill the lowest-energy levels first
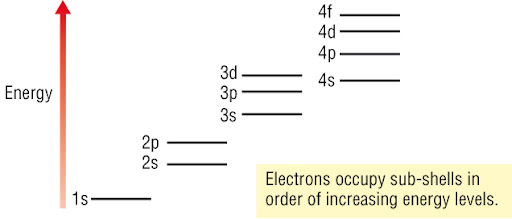
- Electrons occupy the lowest available energy level (orbital or subshell) to achieve maximum stability.
- This is why electrons fill the 1s subshell before 2s, and 2s before 2p.
- The energy order of subshells is:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p
2. Electron–electron repulsion affects how orbitals fill

- Electrons repel each other because they are negatively charged.
- To minimise repulsion, electrons spread out before pairing up.
- This is why p orbitals fill singly first (Hund’s rule).
- Pairing electrons in the same orbital increases repulsion, raising the energy.
3. Subshell energy differences lead to exceptions

- Some elements (such as chromium and copper) have unusual configurations because half-filled or fully filled subshells reduce electron repulsion and lower energy.
- This creates more stable arrangements.
Example
Explain why the 2p orbitals fill with one electron each before any pairing occurs.
▶️ Answer / Explanation
Electrons repel one another. By occupying separate p orbitals, the electrons stay further apart and minimise repulsion.
This lowers the energy of the atom, making the arrangement more stable.
Example
Why is the 4s subshell filled before the 3d subshell?
▶️ Answer / Explanation
The 4s subshell has a slightly lower energy than the 3d subshell in neutral atoms.
Electrons occupy the lowest energy level available, so 4s fills first.
Example
Chromium has the electron configuration \( \mathrm{[Ar]\;3d^5\;4s^1} \), not \( \mathrm{[Ar]\;3d^4\;4s^2} \). Explain this in terms of energy and electron repulsion.
▶️ Answer / Explanation
A half-filled d subshell (3d⁵) is more stable because electrons are spread out among the five d orbitals, minimising repulsion.
Moving one electron from 4s to 3d reduces repulsion and lowers the overall energy.
Therefore the configuration becomes \( \mathrm{3d^5\;4s^1} \) instead of \( \mathrm{3d^4\;4s^2} \).
Factors Influencing Ionisation Energies
Ionisation energy depends on how strongly an atom holds its electrons. This strength of attraction is controlled by several key factors: nuclear charge, atomic/ionic radius, shielding, subshell energy and spin-pair repulsion.
Nuclear charge
- A higher nuclear charge means more protons in the nucleus.

- Stronger attraction between nucleus and the outer electron.
- Result: higher ionisation energy.
Atomic or ionic radius
- Larger radius means the outer electron is further from the nucleus.
- Attraction is weaker, so less energy is needed to remove the electron.
- Result: lower ionisation energy.
Shielding by inner shells and subshells
- Inner electrons repel outer electrons.
- This reduces the effective nuclear charge felt by the outer electron.
- More inner shells = more shielding.
- Result: lower ionisation energy.
Subshell energy differences
- Electrons in higher-energy subshells (such as p) are easier to remove than those in lower-energy subshells (such as s).
- This explains the drop in IE from Group 2 (s electron) to Group 13 (p electron).
- Higher-energy subshell → lower ionisation energy.
Spin-pair repulsion
- Paired electrons in the same orbital repel each other strongly.
- An electron in a paired orbital is easier to remove than one in a singly occupied orbital.
- This explains the drop in IE from Group 15 → Group 16.
- More repulsion → lower ionisation energy.
Example
Explain why potassium has a lower first ionisation energy than sodium.
▶️ Answer / Explanation
Potassium has an extra electron shell, increasing its atomic radius and shielding.
The outer electron is further from the nucleus and less strongly attracted.
Therefore potassium has a lower IE than sodium.
Example
Why is the first ionisation energy of boron lower than that of beryllium?
▶️ Answer / Explanation
Beryllium removes a 2s electron, but boron removes a higher-energy 2p electron.
The 2p electron is less strongly attracted to the nucleus because it is in a higher-energy subshell.
Therefore boron has a lower IE.
Example
Explain why oxygen has a lower first ionisation energy than nitrogen, even though it has a higher nuclear charge.
▶️ Answer / Explanation
Nitrogen has three unpaired p electrons, but oxygen has one paired p orbital.
The paired electrons in oxygen experience spin-pair repulsion, making one electron easier to remove.
Therefore oxygen has a lower IE despite its greater nuclear charge.
Deducing Electronic Configurations from Successive Ionisation Energy Data
Successive ionisation energies provide strong evidence for the number of electrons in each shell of an atom. By examining where the large jumps occur in the data, we can work out the electronic configuration of an element.
Key idea: locating large jumps

- Successive ionisation energies always increase, but not evenly.
- A large jump indicates the point where electrons begin to be removed from a new, inner shell.
- The number of electrons removed before the large jump shows how many electrons are in the outer shell.
- This allows deduction of the group and the full electron configuration.
How to deduce configuration from data
- Step 1: Look at the successive IE values.
- Step 2: Identify where the large jump occurs.
- Step 3: The number of electrons removed before the jump = number of valence electrons.
- Step 4: Determine the group, then reconstruct the full configuration.
Example
Successive ionisation energies of an element show a huge jump between IE₁ and IE₂. Deduce the group and configuration.
▶️ Answer / Explanation
A large jump after the first IE means the first electron was removed from the outer shell, but the second electron would be removed from an inner shell.
Thus the atom has only 1 valence electron.
The element is in Group 1.
Example configuration: \( 2,8,1 \) (for sodium).
Example
The successive ionisation energies (arbitrary units) for an element are:
IE₁ = 800
IE₂ = 1250
IE₃ = 4100
IE₄ = 5500
IE₅ = 7000
IE₆ = 22 000
Deduce the number of valence electrons and the electronic configuration.
▶️ Answer / Explanation
There is a huge jump between IE₅ (7000) and IE₆ (22 000).
This means the first 5 electrons were outer-shell electrons.
Valence electrons = 5 → Group 15.
Electronic configuration (example): \( 2,8,5 \) (for phosphorus).
Example
An element has the following successive ionisation energies (kJ mol⁻¹):
IE₁ = 738
IE₂ = 1450
IE₃ = 7730
IE₄ = 10 500
IE₅ = 13 600
Use the data to deduce the electronic configuration.
▶️ Answer / Explanation
There is a very large jump between IE₂ (1450) and IE₃ (7730).
This means the first 2 electrons are valence electrons, and removing the third electron would break into an inner shell.
Valence electrons = 2 → Group 2.
Electronic configuration (example): \( 2,8,2 \) (for magnesium).
Using Successive Ionisation Energies to Deduce the Position of an Element in the Periodic Table
Successive ionisation energy data show how much energy is needed to remove each electron one at a time. By examining the pattern of these values, we can determine the number of electrons in the outer shell, and therefore identify the element’s group.
Key idea

- A large jump in successive ionisation energies indicates that an electron is being removed from a new, inner shell.
- The number of electrons removed before the large jump equals the number of electrons in the outer shell.
- The number of outer-shell electrons = the group number (for Groups 1–18).
Simple method
- Step 1: Look for where the large increase between successive ionisation energies occurs.
- Step 2: Count how many electrons were removed before that jump.
- Step 3: That number is the group number.
- Step 4: Use total electrons (from atomic number) to determine the period if needed.
Example
Successive IEs (arbitrary units): IE₁ = 420, IE₂ = 920, IE₃ = 28 500. Deduce the group of the element.
▶️ Answer / Explanation
A very large jump occurs between IE₂ and IE₃. Two electrons are removed before the jump, so there are 2 valence electrons.
Conclusion: the element is a Group 2 element (an alkaline earth metal).
Example
IE data (kJ mol\(^{-1}\)): IE₁ = 789, IE₂ = 1 500, IE₃ = 6 900, IE₄ = 8 100, IE₅ = 10 200, IE₆ = 39 000. Deduce group and suggest a likely period.
▶️ Answer / Explanation
There is a very large jump between IE₅ (10 200) and IE₆ (39 000): five electrons removed before the jump → 5 valence electrons.
Group: 5 valence electrons → Group 15 (pnictogens).
To suggest a period, compare absolute IE magnitudes: the relatively low values for IE₁–IE₅ (hundreds to ~10³ kJ mol\(^{-1}\)) indicate valence electrons are not as tightly bound as for first-row elements, so this element is likely in Period 3 or Period 4 rather than Period 2. If you know typical IE ranges, IE₁ ≈ 789 is consistent with a Period 3 or 4 main-group element (for example phosphorus, Period 3, has IE₁ ≈ 1012 kJ mol\(^{-1}\) — here the numbers are illustrative).
Practical note: for a firm period assignment you normally need either the atomic number or to match the IE magnitudes with known elements; the jump gives group reliably, period usually needs extra data.
Example
IEs (kJ mol\(^{-1}\)): IE₁ = 762, IE₂ = 1 580, IE₃ = 7 730, IE₄ = 10 500, IE₅ = 13 600. Deduce the group and the likely electronic configuration (main-group assumption).
▶️ Answer / Explanation
A very large jump occurs between IE₂ and IE₃ (1450 → 7730). Two electrons are removed easily, then a large jump → 2 valence electrons.
Group: Group 2.
Likely electronic configuration (main-group): \( 2,8,2 \) (for example magnesium). If absolute IE magnitudes fit known values (IE₁ ≈ 738 kJ mol\(^{-1}\) for Mg), that confirms Period 3 and the configuration \( 2,8,2 \).