CIE AS/A Level Chemistry 20.1 Addition polymerisation Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 20.1 Addition polymerisation Study Notes – New Syllabus
CIE AS/A Level Chemistry 20.1 Addition polymerisation Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
describe addition polymerisation as exemplified by poly(ethene) and poly(chloroethene), PVC
deduce the repeat unit of an addition polymer obtained from a given monomer
identify the monomer(s) present in a given section of an addition polymer molecule
recognise the difficulty of the disposal of poly(alkene)s, i.e. non-biodegradability and harmful
combustion products
Addition Polymerisation
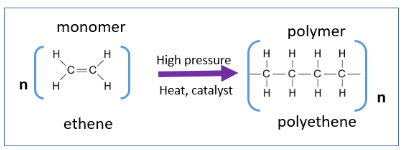
Addition polymerisation is a reaction in which many small molecules, called monomers, join together to form a long-chain molecule, called a polymer.
This type of polymerisation occurs with alkenes, which contain a C=C double bond.
General Features of Addition Polymerisation
- Monomers are unsaturated molecules (usually alkenes)
- The C=C double bond breaks during the reaction
- No other products are formed
- Only one type of monomer is involved
(a) Formation of Poly(ethene)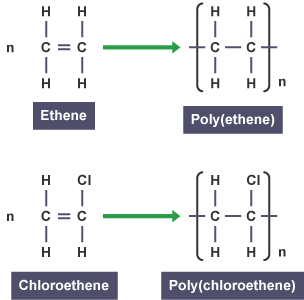
Ethene undergoes addition polymerisation to form poly(ethene).
Monomer: \( \mathrm{CH_2=CH_2} \)
Polymer repeating unit: \( \mathrm{[-CH_2-CH_2-]_n} \)
(b) Formation of Poly(chloroethene) (PVC)
Chloroethene undergoes addition polymerisation to form poly(chloroethene), also known as PVC.
Monomer: \( \mathrm{CH_2=CHCl} \)
Polymer repeating unit: \( \mathrm{[-CH_2-CHCl-]_n} \)
In both reactions, the C=C double bond in the monomer is broken and replaced by C–C single bonds in the polymer chain.
Example
Explain why ethene can undergo addition polymerisation.
▶️ Answer / Explanation
Ethene contains a C=C double bond.
The double bond can break and allow the molecules to join together.
This forms a long-chain polymer with only single C–C bonds.
Example
Describe how the structure of the monomer chloroethene determines the structure of PVC.
▶️ Answer / Explanation
Chloroethene contains a C=C double bond, which breaks during polymerisation.
This allows monomers to join together to form a long chain.
The chlorine atom remains attached to every other carbon in the polymer chain.
Deducing the Repeat Unit of an Addition Polymer
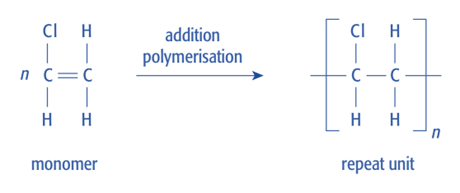
In addition polymerisation, the repeat unit of a polymer can be deduced directly from the structure of the alkene monomer.
Method for Deducing the Repeat Unit
- Identify the C=C double bond in the monomer
- Break the C=C double bond
- Replace it with a C–C single bond
- Keep all other atoms attached to the same carbon atoms
- Place brackets around the repeat unit and add subscript \( \mathrm{n} \)
Only the atoms directly involved in the double bond change; all side groups remain unchanged.
Example
The monomer is propene, \( \mathrm{CH_2=CHCH_3} \). Deduce the repeat unit of the addition polymer formed.
▶️ Answer / Explanation
The C=C double bond in propene breaks.
The two carbon atoms become joined by a single bond.
The \( \mathrm{CH_3} \) group remains attached to the second carbon.
The repeat unit is:
\( \mathrm{[-CH_2-CH(CH_3)-]_n} \)
Example
The monomer shown has the structure \( \mathrm{CH_2=C(CH_3)Cl} \). Deduce the repeat unit of the polymer formed by addition polymerisation.
▶️ Answer / Explanation
The C=C double bond breaks during polymerisation.
Both the \( \mathrm{CH_3} \) group and the chlorine atom remain attached to the same carbon.
The repeat unit is:
\( \mathrm{[-CH_2-C(CH_3)(Cl)-]_n} \)
Brackets and subscript \( \mathrm{n} \) indicate a repeating polymer chain.
Identifying the Monomer from an Addition Polymer
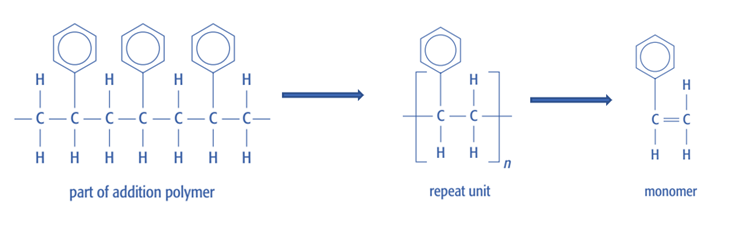
In addition polymerisation, the monomer can be identified from a section of the polymer by reversing the polymerisation process.
Method for Identifying the Monomer
- Identify the repeat unit within the polymer chain
- Remove the single C–C bond between the two backbone carbon atoms
- Insert a C=C double bond between these two carbon atoms
- Keep all side groups attached to the same carbon atoms
- The resulting structure is the monomer
Addition polymers are formed from one type of monomer only.
Example
A section of an addition polymer contains the repeat unit \( \mathrm{[-CH_2-CH_2-]_n} \). Identify the monomer used to form this polymer.
▶️ Answer / Explanation
The polymer backbone contains two carbon atoms joined by a single bond.
Reinserting a C=C double bond gives \( \mathrm{CH_2=CH_2} \).
The monomer is ethene.
Example
A section of a polymer has the structure \( \mathrm{-CH_2-CH(CH_3)-CH_2-CH(CH_3)-} \). Identify the monomer from which this polymer was formed.
▶️ Answer / Explanation
The repeating unit is \( \mathrm{-CH_2-CH(CH_3)-} \).
Replacing the single C–C bond with a C=C double bond gives \( \mathrm{CH_2=CHCH_3} \).
The monomer is propene.
Environmental Issues Associated with Poly(alkene)s
Poly(alkene)s, such as poly(ethene) and poly(propene), are widely used plastics. However, their disposal presents significant environmental problems.
Non-biodegradability
Poly(alkene)s are non-biodegradable because they contain strong C–C and C–H bonds that cannot be broken down by microorganisms.
- They persist in landfill for many years
- They accumulate in the environment
- They contribute to plastic pollution
Combustion of Poly(alkene)s
Poly(alkene)s can be disposed of by incineration, but this can produce harmful gases.
- Complete combustion produces carbon dioxide and water
- Carbon dioxide is a greenhouse gas
- Incomplete combustion produces carbon monoxide and soot
Carbon monoxide is toxic and reduces the oxygen-carrying capacity of blood.
Example
Explain why poly(ethene) is described as non-biodegradable.
▶️ Answer / Explanation
Poly(ethene) contains strong carbon–carbon and carbon–hydrogen bonds.
Microorganisms do not have enzymes that can break these bonds.
As a result, poly(ethene) does not decompose naturally.
Example
Discuss two environmental problems associated with the disposal of poly(alkene)s.
▶️ Answer / Explanation
Poly(alkene)s are non-biodegradable and accumulate in landfill and natural environments.
Incineration produces carbon dioxide, which contributes to global warming.
Incomplete combustion can produce toxic carbon monoxide.
