CIE AS/A Level Chemistry 23.4 Gibbs free energy change, $\Delta G$ Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 23.4 Gibbs free energy change, $\Delta G$ Study Notes – New Syllabus
CIE AS/A Level Chemistry 23.4 Gibbs free energy change, $\Delta G$ Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
state and use the Gibbs equation ΔG⦵ = ΔH⦵ – TΔS⦵
perform calculations using the equation ΔG⦵ = ΔH⦵ – TΔS⦵
state whether a reaction or process will be feasible by using the sign of ΔG
predict the effect of temperature change on the feasibility of a reaction, given standard enthalpy and entropy changes
Gibbs Free Energy
The Gibbs free energy change, \( \mathrm{\Delta G^\circ} \), is used to predict whether a reaction is feasible under standard conditions.
Gibbs Equation![]()
The relationship between enthalpy, entropy and Gibbs free energy is:
\( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \)
Where:
- \( \mathrm{\Delta G^\circ} \) is in \( \mathrm{kJ\,mol^{-1}} \)
- \( \mathrm{\Delta H^\circ} \) is in \( \mathrm{kJ\,mol^{-1}} \)
- \( \mathrm{\Delta S^\circ} \) must be in \( \mathrm{kJ\,mol^{-1}\,K^{-1}} \)
- \( \mathrm{T} \) is the temperature in kelvin, \( \mathrm{K} \)
Standard entropy values are usually given in \( \mathrm{J\,mol^{-1}\,K^{-1}} \) and must be converted to \( \mathrm{kJ\,mol^{-1}\,K^{-1}} \) before substitution.
Example
For a reaction at 298 K, \( \mathrm{\Delta H^\circ = -92\ kJ\,mol^{-1}} \) and \( \mathrm{\Delta S^\circ = -198\ J\,mol^{-1}\,K^{-1}} \). Calculate \( \mathrm{\Delta G^\circ} \) and comment on feasibility.
▶️ Answer / Explanation
Convert entropy to \( \mathrm{kJ\,mol^{-1}\,K^{-1}} \):
\( \mathrm{\Delta S^\circ = -0.198\ kJ\,mol^{-1}\,K^{-1}} \)
Substitute into the Gibbs equation:
\( \mathrm{\Delta G^\circ = -92 – (298 \times -0.198)} \)
\( \mathrm{\Delta G^\circ = -92 + 59.0 = -33.0\ kJ\,mol^{-1}} \)
The reaction is feasible.
Example
A reaction has \( \mathrm{\Delta H^\circ = +45\ kJ\,mol^{-1}} \) and \( \mathrm{\Delta S^\circ = +120\ J\,mol^{-1}\,K^{-1}} \). Calculate the minimum temperature at which the reaction becomes feasible.
▶️ Answer / Explanation
At feasibility threshold, \( \mathrm{\Delta G^\circ = 0} \).
Convert entropy:
\( \mathrm{\Delta S^\circ = 0.120\ kJ\,mol^{-1}\,K^{-1}} \)
Rearrange the equation:
\( \mathrm{0 = \Delta H^\circ – T\Delta S^\circ} \)
\( \mathrm{T = \dfrac{\Delta H^\circ}{\Delta S^\circ}} \)
\( \mathrm{T = \dfrac{45}{0.120} = 375\ K} \)
The reaction is feasible above 375 K.
Feasibility and the Sign of Gibbs Free Energy
The sign of the Gibbs free energy change, \( \mathrm{\Delta G} \), is used to determine whether a reaction or process is feasible under specified conditions.
Using the Sign of \( \mathrm{\Delta G} \)
- If \( \mathrm{\Delta G < 0} \), the reaction or process is feasible
- If \( \mathrm{\Delta G > 0} \), the reaction or process is not feasible
- If \( \mathrm{\Delta G = 0} \), the system is at equilibrium
A feasible reaction is one that can occur spontaneously, but it may still be slow.
Example
A reaction has a Gibbs free energy change of \( \mathrm{\Delta G = -18\ kJ\,mol^{-1}} \) at 298 K. State whether the reaction is feasible and explain your answer.
▶️ Answer / Explanation
The reaction is feasible.
The Gibbs free energy change is negative.
A negative \( \mathrm{\Delta G} \) indicates that the reaction can occur spontaneously.
Example
For a process, \( \mathrm{\Delta H = +40\ kJ\,mol^{-1}} \) and \( \mathrm{\Delta S = +150\ J\,mol^{-1}\,K^{-1}} \). State whether the process is feasible at high temperature and explain your answer.
▶️ Answer / Explanation
At high temperature, the \( \mathrm{T\Delta S} \) term becomes large.
Since \( \mathrm{\Delta S} \) is positive, \( \mathrm{T\Delta S} \) is positive.
This can make \( \mathrm{\Delta G} \) negative.
The process becomes feasible at sufficiently high temperature.
Effect of Temperature on Feasibility
The effect of temperature on the feasibility of a reaction can be predicted using the Gibbs free energy equation: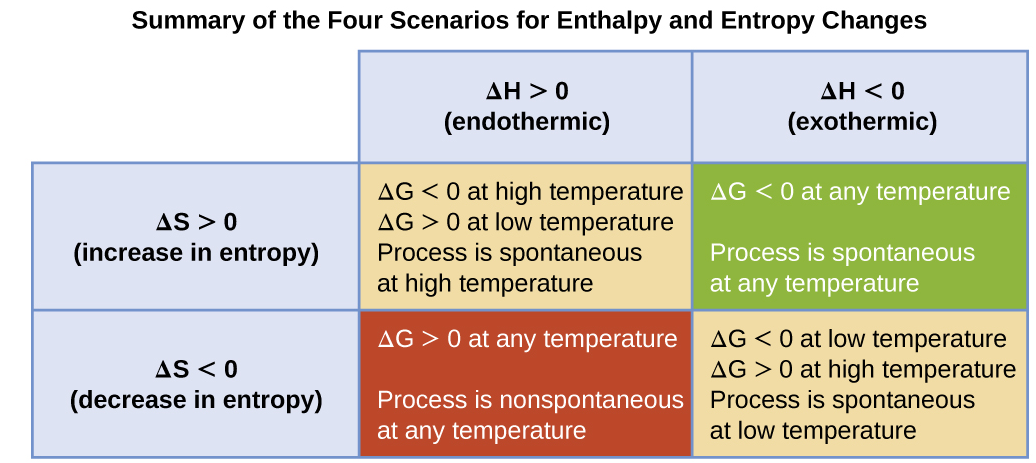
\( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \)
The signs of \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \) determine how temperature affects feasibility.
General Rules
- If increasing temperature makes \( \mathrm{T\Delta S^\circ} \) more positive, it can make \( \mathrm{\Delta G^\circ} \) more negative
- If increasing temperature makes \( \mathrm{T\Delta S^\circ} \) more negative, it can make \( \mathrm{\Delta G^\circ} \) more positive
Feasibility Based on Signs of \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \)
| Sign of \( \mathrm{\Delta H^\circ} \) | Sign of \( \mathrm{\Delta S^\circ} \) | Feasibility |
|---|---|---|
| \( \mathrm{\Delta H^\circ < 0} \) | \( \mathrm{\Delta S^\circ > 0} \) | Feasible at all temperatures |
| \( \mathrm{\Delta H^\circ > 0} \) | \( \mathrm{\Delta S^\circ < 0} \) | Not feasible at any temperature |
| \( \mathrm{\Delta H^\circ < 0} \) | \( \mathrm{\Delta S^\circ < 0} \) | Feasible at low temperatures |
| \( \mathrm{\Delta H^\circ > 0} \) | \( \mathrm{\Delta S^\circ > 0} \) | Feasible at high temperatures |
These predictions come from how the \( \mathrm{T\Delta S^\circ} \) term changes with temperature.
Example
A reaction has \( \mathrm{\Delta H^\circ = -80\ kJ\,mol^{-1}} \) and \( \mathrm{\Delta S^\circ = -120\ J\,mol^{-1}\,K^{-1}} \). Predict whether the reaction is more feasible at low or high temperature.
▶️ Answer / Explanation
The reaction is exothermic and has a negative entropy change.
At low temperature, the \( \mathrm{T\Delta S^\circ} \) term is small.
This keeps \( \mathrm{\Delta G^\circ} \) negative.
The reaction is more feasible at low temperature.
Example
For a reaction, \( \mathrm{\Delta H^\circ = +65\ kJ\,mol^{-1}} \) and \( \mathrm{\Delta S^\circ = +180\ J\,mol^{-1}\,K^{-1}} \).
Explain how increasing temperature affects feasibility.
▶️ Answer / Explanation
The reaction is endothermic with a positive entropy change.
As temperature increases, the \( \mathrm{T\Delta S^\circ} \) term becomes larger.
This reduces \( \mathrm{\Delta G^\circ} \).
At sufficiently high temperature, \( \mathrm{\Delta G^\circ} \) becomes negative.
The reaction becomes feasible at high temperature.
