CIE AS/A Level Chemistry 26.1 Simple rate equations, orders of reaction and rate constants Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 26.1 Simple rate equations, orders of reaction and rate constants Study Notes – New Syllabus
CIE AS/A Level Chemistry 26.1 Simple rate equations, orders of reaction and rate constants Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- explain and use the terms rate equation, order of reaction, overall order of reaction, rate constant, half-life, rate-determining step and intermediate
- (a) understand and use rate equations of the form rate = k [A]^m[B]^n (for which m and n are 0, 1 or 2)
(b) deduce the order of a reaction from concentration–time graphs or from experimental data relating to the initial rates method and half-life method
(c) interpret experimental data in graphical form, including concentration–time and rate–concentration graphs
(d) calculate an initial rate using concentration data
(e) construct a rate equation - (a) show understanding that the half-life of a first-order reaction is independent of concentration
(b) use the half-life of a first-order reaction in calculations - calculate the numerical value of a rate constant, for example by:
(a) using the initial rates and the rate equation
(b) using the half-life, t₁⁄₂, and the equation k = 0.693 / t₁⁄₂ - for a multi-step reaction:
(a) suggest a reaction mechanism that is consistent with the rate equation and the equation for the overall reaction
(b) predict the order that would result from a given reaction mechanism and rate-determining step
(c) deduce a rate equation using a given reaction mechanism and rate-determining step for a given reaction
(d) identify an intermediate or catalyst from a given reaction mechanism
(e) identify the rate-determining step from a rate equation and a given reaction mechanism - describe qualitatively the effect of temperature change on the rate constant and hence the rate of a reaction
Kinetics: Rate Equations and Reaction Mechanisms
Chemical kinetics is the study of how fast reactions occur. Key terms such as rate equation, order of reaction, rate constant, half-life, rate-determining step and intermediate are used to describe and analyse reaction rates.
Rate Equation
A rate equation shows how the rate of a reaction depends on the concentration of reactants.![]()
General form:
\( \mathrm{rate = k[A]^m[B]^n} \)
- \( \mathrm{k} \) is the rate constant
- \( \mathrm{m} \) and \( \mathrm{n} \) are the orders of reaction with respect to A and B
Order of Reaction
The order of reaction with respect to a reactant shows how the rate changes when the concentration of that reactant changes.
- First order: rate ∝ concentration
- Second order: rate ∝ (concentration)2
- Zero order: rate independent of concentration
Overall Order of Reaction
The overall order of reaction is the sum of the individual orders in the rate equation.
For example, for \( \mathrm{rate = k[A]^2[B]} \), the overall order is 3.
Rate Constant
The rate constant, \( \mathrm{k} \), is a proportionality constant in the rate equation.
- It is constant for a given temperature
- It changes with temperature and the presence of a catalyst
Half-life
The half-life, \( \mathrm{t_{1/2}} \), is the time taken for the concentration of a reactant to fall to half of its initial value.
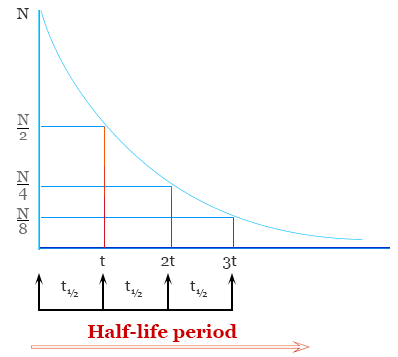
For a first-order reaction, the half-life is constant and independent of concentration.
Reaction Mechanisms
Many reactions occur in a series of steps called a reaction mechanism.
![]()
Rate-Determining Step
The rate-determining step is the slowest step in a reaction mechanism and controls the overall rate of reaction.
![]()
The rate equation is determined by the reactants involved in the rate-determining step.
Intermediate
An intermediate is a species that is formed in one step and used up in a later step.
![]()
- Intermediates do not appear in the overall equation
- They are often highly reactive and short-lived
Example
The rate equation for a reaction is \( \mathrm{rate = k[NO]^2[O_2]} \). State the order with respect to each reactant and the overall order.
▶️ Answer / Explanation
The order with respect to NO is 2.
The order with respect to \( \mathrm{O_2} \) is 1.
The overall order of reaction is \( \mathrm{2 + 1 = 3} \).
Example
A reaction occurs by the following mechanism:
Step 1 (slow): \( \mathrm{A + B \rightarrow C} \)
Step 2 (fast): \( \mathrm{C + D \rightarrow E} \)
Identify the rate-determining step, the intermediate, and write the rate equation.
▶️ Answer / Explanation
The rate-determining step is Step 1 because it is slow.
\( \mathrm{C} \) is an intermediate as it is formed in Step 1 and used up in Step 2.
The rate equation is:
\( \mathrm{rate = k[A][B]} \)
Only reactants in the rate-determining step appear in the rate equation.
Kinetics: Rate Equations and Experimental Determination
Reaction kinetics involves using experimental data to determine how reaction rate depends on reactant concentrations and to construct a rate equation.
(a) Rate Equations![]()
A rate equation has the general form:
\( \mathrm{rate = k[A]^m[B]^n} \)
- \( \mathrm{k} \) is the rate constant
- \( \mathrm{m} \) and \( \mathrm{n} \) are the orders of reaction
- At A level, \( \mathrm{m} \) and \( \mathrm{n} \) are usually 0, 1 or 2
The values of \( \mathrm{m} \) and \( \mathrm{n} \) can only be determined experimentally.
(b) Deducing Order of Reaction![]()
Orders of reaction can be deduced using:
- Concentration–time graphs
- Initial rates method
- Half-life method
Concentration–time graphs
- Zero order: straight-line decrease
- First order: exponential curve
- Second order: curve that becomes less steep rapidly
Half-life method
For a first-order reaction, the half-life is constant and does not depend on initial concentration.
(c) Interpreting Graphical Data
Experimental data may be presented as:![]()
- Concentration–time graphs → slope gives rate
- Rate–concentration graphs → shape indicates order
For example:
- Straight line through origin → first order
- Curve through origin → second order
- Horizontal line → zero order
(d) Calculating an Initial Rate
The initial rate is the rate at the very start of a reaction.
It is calculated using:
\( \mathrm{rate = \dfrac{\Delta [A]}{\Delta t}} \)
The initial rate corresponds to the gradient of the tangent at time = 0 on a concentration–time graph.
(e) Constructing a Rate Equation
To construct a rate equation:
- Use experimental data to find the order of reaction with respect to each reactant
- Write the rate equation using these orders
- Substitute values to calculate the rate constant \( \mathrm{k} \), if required
Example
The following data were obtained for a reaction between A and B. Doubling the concentration of A doubles the rate, while doubling the concentration of B has no effect. Construct the rate equation.
▶️ Answer / Explanation
Doubling the concentration of A doubles the rate, so the reaction is first order with respect to A.
Changing the concentration of B has no effect, so the reaction is zero order with respect to B.
The rate equation is:
\( \mathrm{rate = k[A]} \)
Example
A reaction has an initial rate of \( \mathrm{4.0 \times 10^{-3}\ mol\,dm^{-3}\,s^{-1}} \) when \( \mathrm{[A] = 0.20\ mol\,dm^{-3}} \) and \( \mathrm{[B] = 0.10\ mol\,dm^{-3}} \). When \( \mathrm{[A]} \) is doubled and \( \mathrm{[B]} \) is tripled, the rate increases by a factor of 12. Construct the rate equation.
▶️ Answer / Explanation
Doubling \( \mathrm{[A]} \) and tripling \( \mathrm{[B]} \) increases the rate by 12.
\( \mathrm{2^m \times 3^n = 12} \)
This gives \( \mathrm{m = 1} \) and \( \mathrm{n = 1} \).
The rate equation is:
\( \mathrm{rate = k[A][B]} \)
Half-Life of First-Order Reactions
The concept of half-life is particularly important for first-order reactions. For these reactions, the half-life has a special property that makes calculations straightforward.
(a) Half-Life and Concentration![]()
The half-life, \( \mathrm{t_{1/2}} \), is the time taken for the concentration of a reactant to decrease to half of its initial value.
For a first-order reaction:
\( \mathrm{t_{1/2} = \dfrac{0.693}{k}} \)
This equation shows that the half-life depends only on the rate constant, \( \mathrm{k} \), and does not depend on concentration.
Therefore, for a first-order reaction, the time taken for the concentration to halve is the same at any point during the reaction.
(b) Using Half-Life in Calculations
Because the half-life is constant, first-order reactions can be analysed by counting the number of half-lives.
After each half-life:
- After 1 half-life: concentration = \( \mathrm{\dfrac{1}{2}} \) original
- After 2 half-lives: concentration = \( \mathrm{\dfrac{1}{4}} \) original
- After 3 half-lives: concentration = \( \mathrm{\dfrac{1}{8}} \) original
Alternatively, the half-life can be used to calculate the rate constant using:
\( \mathrm{k = \dfrac{0.693}{t_{1/2}}} \)
Example
The half-life of a first-order reaction is 60 s. Calculate the fraction of the reactant remaining after 180 s.
▶️ Answer / Explanation
180 s corresponds to:
\( \mathrm{\dfrac{180}{60} = 3} \) half-lives.
After 3 half-lives, the fraction remaining is:
\( \mathrm{\left(\dfrac{1}{2}\right)^3 = \dfrac{1}{8}} \).
Example
A radioactive isotope decays by a first-order process and has a half-life of 4.0 days. Calculate the time taken for 87.5% of the isotope to decay.
▶️ Answer / Explanation
If 87.5% has decayed, then 12.5% remains.
\( \mathrm{12.5\% = \dfrac{1}{8}} \) of the original amount.
\( \mathrm{\dfrac{1}{8} = \left(\dfrac{1}{2}\right)^3} \), so this corresponds to 3 half-lives.
Time taken:
\( \mathrm{3 \times 4.0 = 12.0\ days} \)
Calculating the Rate Constant, \( \mathrm{k} \)
The rate constant, \( \mathrm{k} \), can be calculated using experimental data. Two common methods are:
- Using initial rates together with the rate equation
- Using the half-life of a first-order reaction
(a) Using Initial Rates and the Rate Equation
Once the rate equation is known, the rate constant can be found by rearranging:
\( \mathrm{k = \dfrac{rate}{[A]^m[B]^n}} \)
All concentrations are substituted in \( \mathrm{mol\,dm^{-3}} \), and the rate in \( \mathrm{mol\,dm^{-3}\,s^{-1}} \). The units of \( \mathrm{k} \) depend on the overall order of the reaction.
(b) Using the Half-Life of a First-Order Reaction
For a first-order reaction, the half-life is related to the rate constant by:
\( \mathrm{k = \dfrac{0.693}{t_{1/2}}} \)
This method can only be used for first-order reactions, as their half-life is independent of concentration.
Example
A reaction has the rate equation \( \mathrm{rate = k[A]} \). The initial rate is \( \mathrm{2.4 \times 10^{-4}\ mol\,dm^{-3}\,s^{-1}} \) when \( \mathrm{[A] = 0.30\ mol\,dm^{-3}} \). Calculate the value of \( \mathrm{k} \).
▶️ Answer / Explanation
Rearrange the rate equation:
\( \mathrm{k = \dfrac{rate}{[A]}} \)
Substitute the values:
\( \mathrm{k = \dfrac{2.4 \times 10^{-4}}{0.30}} \)
\( \mathrm{k = 8.0 \times 10^{-4}\ s^{-1}} \)
Example
The half-life of a first-order reaction is 250 s. Calculate the rate constant and state its units.
▶️ Answer / Explanation
Use the half-life equation for a first-order reaction:
\( \mathrm{k = \dfrac{0.693}{t_{1/2}}} \)
Substitute the value:
\( \mathrm{k = \dfrac{0.693}{250}} \)
\( \mathrm{k = 2.77 \times 10^{-3}\ s^{-1}} \)
The units are \( \mathrm{s^{-1}} \), which are the units of a first-order rate constant.
Multi-Step Reactions: Mechanisms and Rate Equations
Many reactions occur by a sequence of elementary steps rather than a single collision. For multi-step reactions, the rate equation is linked to the reaction mechanism and in particular to the rate-determining step.
Reaction Mechanisms
A reaction mechanism is a series of elementary steps that show how reactants are converted into products.
![]()
Each step involves a small number of particles and may be fast or slow.
(a) Suggesting a Reaction Mechanism
A proposed mechanism must be consistent with:
- The overall equation (all steps must add up correctly)
- The rate equation (reactants in the rate-determining step must match the rate law)
Species that appear in the mechanism but not in the overall equation must cancel out.
(b) Predicting the Order from a Mechanism
The rate-determining step is the slowest step and controls the overall rate.
The order of reaction is determined by the reactants involved in the rate-determining step.
(c) Deducing a Rate Equation
To deduce a rate equation from a mechanism:
- Identify the rate-determining step
- Write the rate equation using the reactants in that step
- If an intermediate appears, use an earlier fast step to substitute for it
(d) Intermediates and Catalysts
From a reaction mechanism:
- An intermediate is formed in one step and used up in a later step
- A catalyst is used up in an early step and re-formed in a later step
Neither intermediates nor catalysts appear in the overall equation.
(e) Identifying the Rate-Determining Step
If the rate equation is known, the rate-determining step must involve the same reactants (and powers) as the rate equation.
![]()
Any step containing species not present in the rate equation cannot be the rate-determining step.
Example
The overall reaction is:
\( \mathrm{A + B \rightarrow C} \)
The rate equation is:
\( \mathrm{rate = k[A][B]} \)
Suggest a possible two-step reaction mechanism.
▶️ Answer / Explanation
One possible mechanism is:
Step 1 (slow): \( \mathrm{A + B \rightarrow D} \)
Step 2 (fast): \( \mathrm{D \rightarrow C} \)
\( \mathrm{D} \) is an intermediate as it is formed and then used up.
The slow step contains A and B, matching the rate equation.
Example
A reaction occurs by the following mechanism:
Step 1 (fast): \( \mathrm{A + B \rightleftharpoons C} \)
Step 2 (slow): \( \mathrm{C + D \rightarrow E} \)
(i) Identify the rate-determining step
(ii) Identify the intermediate
(iii) Deduce the rate equation
▶️ Answer / Explanation
The rate-determining step is Step 2 because it is slow.
\( \mathrm{C} \) is an intermediate, as it is formed in Step 1 and used up in Step 2.
The rate equation depends on the reactants in the slow step:
\( \mathrm{rate = k[C][D]} \)
If required, \( \mathrm{[C]} \) could be substituted using the fast equilibrium in Step 1.
Effect of Temperature on Rate Constant and Reaction Rate
Changing the temperature has a significant effect on both the rate constant, \( \mathrm{k} \), and therefore the rate of a reaction. This effect can be explained qualitatively using collision theory and activation energy.
Effect on the Rate Constant
As temperature increases, the rate constant, \( \mathrm{k} \), increases.
Increasing temperature gives reacting particles more kinetic energy. As a result:
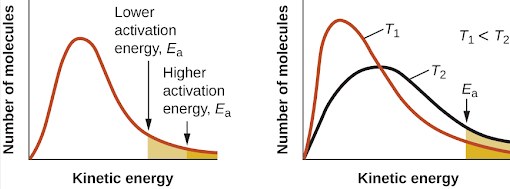
- A greater proportion of particles have energy ≥ activation energy
- More successful collisions occur per unit time
Because the rate constant reflects the frequency of successful collisions, \( \mathrm{k} \) increases rapidly with temperature.
Effect on the Rate of Reaction
The rate of reaction depends on the rate equation:
\( \mathrm{rate = k[A]^m[B]^n} \)
For a given set of concentrations, increasing temperature increases \( \mathrm{k} \), so the rate of reaction increases.
![]()
Conversely, lowering the temperature decreases \( \mathrm{k} \), so the reaction rate decreases.
Key Points
- Temperature changes do not change the orders in the rate equation
- Only the value of the rate constant \( \mathrm{k} \) is affected
- A small temperature increase can cause a large increase in reaction rate
Example
Explain why increasing the temperature increases the rate of a chemical reaction.
▶️ Answer / Explanation
Increasing the temperature increases the kinetic energy of reacting particles.
A greater proportion of particles have energy equal to or greater than the activation energy.
This leads to more successful collisions per second, increasing the rate constant and therefore the rate of reaction.
Example
A reaction has the rate equation \( \mathrm{rate = k[A]^2} \). The temperature is increased while the concentration of A remains constant. Explain the effect on the rate.
▶️ Answer / Explanation
Increasing the temperature increases the rate constant \( \mathrm{k} \).
The order of reaction and the concentration of A remain unchanged.
Since rate depends directly on \( \mathrm{k} \), the rate of reaction increases.
