CIE AS/A Level Chemistry 26.2 Homogeneous and heterogeneous catalysts Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 26.2 Homogeneous and heterogeneous catalysts Study Notes – New Syllabus
CIE AS/A Level Chemistry 26.2 Homogeneous and heterogeneous catalysts Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- explain that catalysts can be homogeneous or heterogeneous
- describe the mode of action of a heterogeneous catalyst to include adsorption of reactants, bond weakening and desorption of products, for example:
(a) iron in the Haber process
(b) palladium, platinum and rhodium in the catalytic removal of oxides of nitrogen from the exhaust gases of car engines - describe the mode of action of a homogeneous catalyst by being used in one step and reformed in a later step, for example:
(a) atmospheric oxides of nitrogen in the oxidation of atmospheric sulfur dioxide
(b) Fe²⁺ or Fe³⁺ in the I⁻/S₂O₈²⁻ reaction
Catalysts: Homogeneous and Heterogeneous
A catalyst is a substance that increases the rate of a reaction by providing an alternative pathway with a lower activation energy. Catalysts are not used up and remain chemically unchanged at the end of the reaction.
Types of Catalysts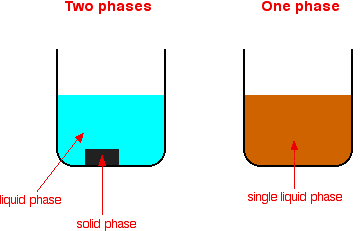
Homogeneous Catalysts
A homogeneous catalyst is in the same physical state as the reactants.
- Usually all species are in the aqueous or gaseous state
- The catalyst forms temporary intermediates with reactants
- The catalyst is regenerated during the reaction
Heterogeneous Catalysts
A heterogeneous catalyst is in a different physical state from the reactants.
- Commonly a solid catalyst with gaseous or liquid reactants
- Reaction occurs at the surface of the catalyst
- The catalyst provides active sites for adsorption
Key Differences
| Homogeneous catalyst | Heterogeneous catalyst |
|---|---|
| Same phase as reactants | Different phase from reactants |
| Forms intermediates | Acts at catalyst surface |
| Harder to separate from products | Easy to separate and reuse |
Example
Classify the catalyst in the reaction between iodide ions and hydrogen peroxide in acidic solution.
▶️ Answer / Explanation
The iodide ions and reactants are all in aqueous solution.
Therefore, the catalyst is a homogeneous catalyst.
Example
The Haber process uses an iron catalyst to produce ammonia from nitrogen and hydrogen. Classify the catalyst and explain your answer.
▶️ Answer / Explanation
Nitrogen and hydrogen are gases, while iron is a solid.
Because the catalyst is in a different physical state from the reactants, it is a heterogeneous catalyst.
The reaction occurs on the surface of the iron catalyst.
Mode of Action of a Heterogeneous Catalyst
A heterogeneous catalyst is in a different physical state from the reactants. It increases the rate of reaction by providing an alternative pathway with a lower activation energy, usually at the surface of the catalyst.
Key Steps in Heterogeneous Catalysis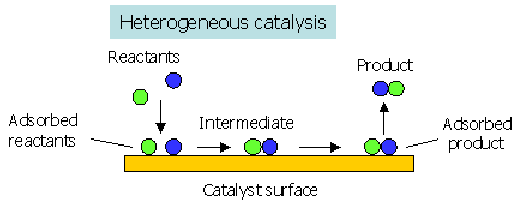
The action of a heterogeneous catalyst involves three main stages:
- Adsorption of reactant molecules onto the catalyst surface
- Bond weakening and reaction at the surface
- Desorption of product molecules from the surface
Adsorption
Reactant molecules are adsorbed onto the surface of the solid catalyst.
- Adsorption occurs at active sites
- Bonds within the reactant molecules are weakened
- This lowers the activation energy for the reaction
Bond Weakening and Reaction
When reactants are adsorbed, their bonds are stretched or weakened.
This makes it easier for bonds to break and new bonds to form, so the reaction occurs more rapidly at the catalyst surface.
Desorption
Product molecules are desorbed from the catalyst surface.
- The catalyst is left unchanged
- Active sites are freed for further reactions
(a) Iron in the Haber Process
In the Haber process, nitrogen and hydrogen react to form ammonia using an iron catalyst.
- \( \mathrm{N_2} \) and \( \mathrm{H_2} \) molecules are adsorbed onto the iron surface
- The strong \( \mathrm{N \equiv N} \) bond is weakened on the surface
- Nitrogen and hydrogen atoms react to form \( \mathrm{NH_3} \)
- Ammonia molecules desorb from the surface
This significantly increases the rate of ammonia formation.
(b) Catalytic Converters in Car Exhausts
Car catalytic converters use metals such as platinum, palladium and rhodium to remove harmful gases.
These metals catalyse reactions involving oxides of nitrogen, carbon monoxide and unburnt hydrocarbons.
- Oxides of nitrogen and carbon monoxide are adsorbed onto the metal surface
- Bonds in the molecules are weakened
- \( \mathrm{NO} \) is reduced to \( \mathrm{N_2} \)
- \( \mathrm{CO} \) is oxidised to \( \mathrm{CO_2} \)
- Products desorb from the catalyst surface
Example
Describe how a heterogeneous catalyst increases the rate of reaction.
▶️ Answer / Explanation
Reactant molecules are adsorbed onto the surface of the catalyst at active sites.
Adsorption weakens bonds within the reactant molecules, lowering the activation energy.
Products then desorb from the surface, leaving the catalyst unchanged.
Example
Explain how iron acts as a heterogeneous catalyst in the Haber process.
▶️ Answer / Explanation
Nitrogen and hydrogen molecules are adsorbed onto the surface of the iron catalyst.
Adsorption weakens the strong \( \mathrm{N \equiv N} \) bond, reducing the activation energy.
Nitrogen and hydrogen atoms react on the surface to form ammonia molecules.
Ammonia then desorbs from the surface, freeing active sites for further reactions.
Mode of Action of a Homogeneous Catalyst
A homogeneous catalyst is in the same physical state as the reactants. It increases the rate of reaction by providing an alternative pathway with a lower activation energy.
A key feature of homogeneous catalysis is that the catalyst is: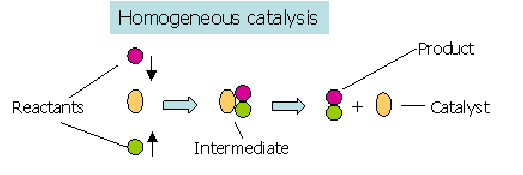
- Used up in one step of the reaction mechanism
- Reformed in a later step
As a result, the catalyst does not appear in the overall equation.
General Catalytic Cycle
A homogeneous catalytic mechanism typically involves:
- Reaction between the catalyst and a reactant to form an intermediate
- Reaction of the intermediate to regenerate the catalyst
(a) Oxides of Nitrogen in the Oxidation of Sulfur Dioxide
In the atmosphere, sulfur dioxide is oxidised to sulfur trioxide. This reaction is catalysed by oxides of nitrogen, such as \( \mathrm{NO} \) and \( \mathrm{NO_2} \).
The catalytic cycle is:
Step 1: \( \mathrm{SO_2 + NO_2 \rightarrow SO_3 + NO} \)
Step 2: \( \mathrm{NO + \tfrac{1}{2}O_2 \rightarrow NO_2} \)
In this mechanism:
- \( \mathrm{NO_2} \) is used up in Step 1
- \( \mathrm{NO_2} \) is reformed in Step 2
- \( \mathrm{NO} \) is an intermediate
The overall reaction is:
\( \mathrm{SO_2 + \tfrac{1}{2}O_2 \rightarrow SO_3} \)
(b) Fe²⁺ / Fe³⁺ in the I⁻ / S₂O₈²⁻ Reaction
The reaction between iodide ions and peroxodisulfate ions is slow, but can be catalysed by iron(II) or iron(III) ions in solution.
The catalytic cycle is:
Step 1: \( \mathrm{S_2O_8^{2-} + 2Fe^{2+} \rightarrow 2SO_4^{2-} + 2Fe^{3+}} \)
Step 2: \( \mathrm{2Fe^{3+} + 2I^- \rightarrow I_2 + 2Fe^{2+}} \)
In this mechanism:
- \( \mathrm{Fe^{2+}} \) is used up in Step 1
- \( \mathrm{Fe^{2+}} \) is regenerated in Step 2
- \( \mathrm{Fe^{3+}} \) is an intermediate
The overall reaction is:
\( \mathrm{S_2O_8^{2-} + 2I^- \rightarrow I_2 + 2SO_4^{2-}} \)
Example
Explain how oxides of nitrogen act as homogeneous catalysts in the oxidation of sulfur dioxide.
▶️ Answer / Explanation
\( \mathrm{NO_2} \) reacts with sulfur dioxide to form sulfur trioxide and \( \mathrm{NO} \).
The \( \mathrm{NO} \) formed then reacts with oxygen to regenerate \( \mathrm{NO_2} \).
The catalyst is used in one step and reformed in a later step, so it does not appear in the overall equation.
Example
The reaction between iodide ions and peroxodisulfate ions is catalysed by iron ions. Identify the catalyst, the intermediate, and explain how the catalyst increases the rate.
▶️ Answer / Explanation
The catalyst is \( \mathrm{Fe^{2+}} \), which is used up in the first step.
\( \mathrm{Fe^{3+}} \) is an intermediate because it is formed in one step and used up in a later step.
The catalyst provides an alternative reaction pathway with a lower activation energy and is regenerated, increasing the rate of reaction.
