CIE AS/A Level Chemistry 28.2 General characteristic chemical properties of the first set of transition elements Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 28.2 General characteristic chemical properties of the first set of transition elements Study Notes – New Syllabus
CIE AS/A Level Chemistry 28.2 General characteristic chemical properties of the first set of transition elements Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
describe and explain the reactions of transition elements with ligands to form complexes, including the complexes of copper(II) and cobalt(II) ions with water and ammonia molecules and hydroxide and chloride ions
define the term ligand as a species that contains a lone pair of electrons that forms a dative covalent bond to a central metal atom/ion
understand and use the terms:
(a) monodentate ligand including as examples H₂O, NH₃, Cl⁻ and CN⁻
(b) bidentate ligand including as examples 1,2-diaminoethane, en, H₂NCH₂CH₂NH₂ and the ethanedioate ion, C₂O₄²⁻
(c) polydentate ligand including as an example EDTA⁴⁻define the term complex as a molecule or ion formed by a central metal atom/ion surrounded by one or more ligands
describe the geometry (shape and bond angles) of transition element complexes which are linear, square planar, tetrahedral or octahedral
(a) state what is meant by coordination number
(b) predict the formula and charge of a complex ion, given the metal ion, its charge or oxidation state, the ligand and its coordination number or geometryexplain qualitatively that ligand exchange can occur, including the complexes of copper(II) ions and cobalt(II) ions with water and ammonia molecules and hydroxide and chloride ions
predict, using E⦵ values, the feasibility of redox reactions involving transition elements and their ions
describe the reactions of, and perform calculations involving:
(a) MnO₄⁻/C₂O₄²⁻ in acid solution given suitable data
(b) MnO₄⁻/Fe²⁺ in acid solution given suitable data
(c) Cu²⁺/I⁻ given suitable dataperform calculations involving other redox systems given suitable data
Reactions of Transition Metal Ions with Ligands
Transition metal ions react readily with ligands to form complex ions. These reactions occur because transition metal ions have vacant, energetically accessible d orbitals that can accept lone pairs from ligands.
Ligands and Complex Formation
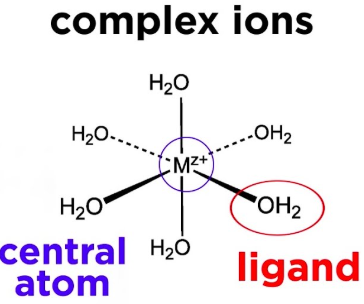
A ligand is a species that donates a lone pair of electrons to a central metal ion to form a dative (coordinate) bond.
Common ligands include:
- \( \mathrm{H_2O} \) (neutral ligand)
- \( \mathrm{NH_3} \) (neutral ligand)
- \( \mathrm{OH^-} \) and \( \mathrm{Cl^-} \) (negative ligands)
Ligand substitution reactions occur when one ligand is replaced by another.
Copper(II) Complexes
Copper(II) with Water
In aqueous solution, copper(II) ions form a pale blue complex:

\( \mathrm{[Cu(H_2O)_6]^{2+}} \)
Water molecules donate lone pairs to the copper(II) ion, forming six coordinate bonds in an octahedral arrangement.
Copper(II) with Hydroxide Ions
When hydroxide ions are added to the aqueous copper(II) complex, a pale blue precipitate of copper(II) hydroxide forms:
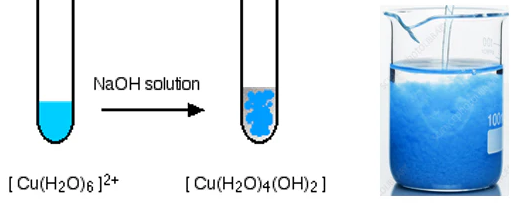
\( \mathrm{[Cu(H_2O)_6]^{2+} + 2OH^- \rightarrow Cu(OH)_2(s) + 6H_2O} \)
This occurs because hydroxide ions remove protons from coordinated water molecules.
Copper(II) with Ammonia
With a small amount of ammonia, a pale blue precipitate of \( \mathrm{Cu(OH)_2} \) is first formed. In excess ammonia, this precipitate dissolves to form a deep blue complex:
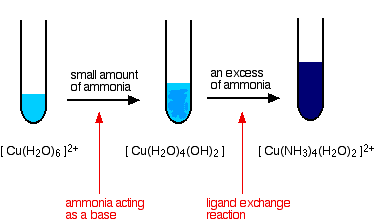
\( \mathrm{[Cu(H_2O)_6]^{2+} + 4NH_3 \rightarrow [Cu(NH_3)_4(H_2O)_2]^{2+} + 4H_2O} \)
Ammonia molecules replace water ligands because they form stronger dative bonds with copper(II).
Cobalt(II) Complexes
Cobalt(II) with Water
Cobalt(II) ions form a pink complex in aqueous solution:
\( \mathrm{[Co(H_2O)_6]^{2+}} \)
Cobalt(II) with Hydroxide Ions
Addition of hydroxide ions produces a blue precipitate of cobalt(II) hydroxide:
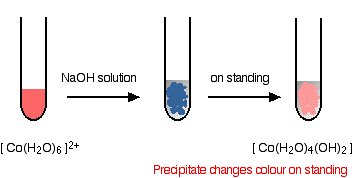
\( \mathrm{[Co(H_2O)_6]^{2+} + 2OH^- \rightarrow Co(OH)_2(s) + 6H_2O} \)
Cobalt(II) with Ammonia
Ammonia initially acts as a base, forming \( \mathrm{Co(OH)_2} \). In excess ammonia, a yellow-brown ammine complex forms (in air):

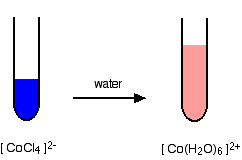
Cobalt(II) with Chloride Ions
In high concentrations of chloride ions, ligand substitution occurs:
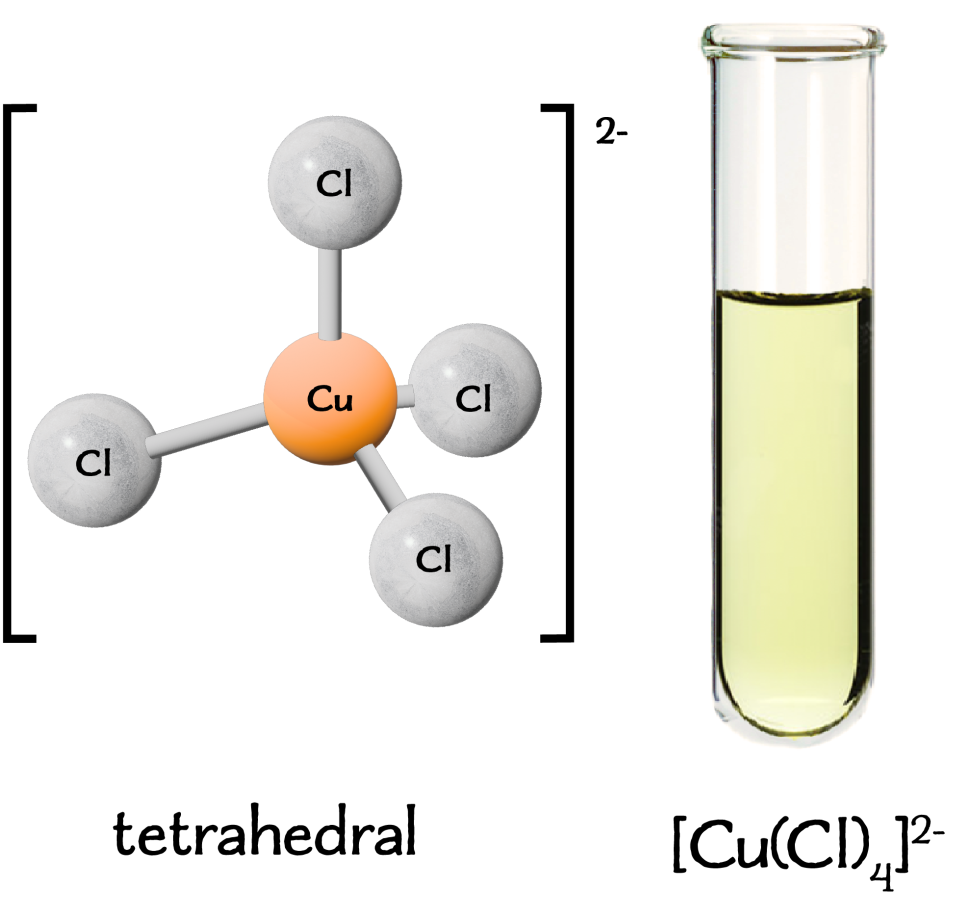
\( \mathrm{[Co(H_2O)_6]^{2+} + 4Cl^- \rightleftharpoons [CoCl_4]^{2-} + 6H_2O} \)
The equilibrium shifts to the right at high chloride concentration, producing a blue complex.
Explanation of Complex Formation
These reactions occur because transition metal ions:
- Have vacant, low-energy d orbitals
- Accept lone pairs from ligands to form dative bonds
- Undergo ligand substitution when stronger ligands are present
Example
Describe the reaction of aqueous copper(II) ions with excess ammonia.
▶️ Answer / Explanation
A pale blue precipitate of \( \mathrm{Cu(OH)_2} \) forms initially.
In excess ammonia, the precipitate dissolves to form a deep blue complex, \( \mathrm{[Cu(NH_3)_4(H_2O)_2]^{2+}} \).
This occurs because ammonia replaces water ligands by forming stronger dative bonds.
Example
Describe and explain the colour change when concentrated hydrochloric acid is added to aqueous cobalt(II) chloride.
▶️ Answer / Explanation
The pink \( \mathrm{[Co(H_2O)_6]^{2+}} \) complex is converted into the blue \( \mathrm{[CoCl_4]^{2-}} \) complex.
Chloride ions replace water ligands in a ligand substitution reaction.
The high concentration of chloride ions shifts the equilibrium to favour the chloro complex.
Ligands
A ligand is a species that contains a lone pair of electrons which it donates to a central metal atom or ion to form a dative (coordinate) covalent bond.
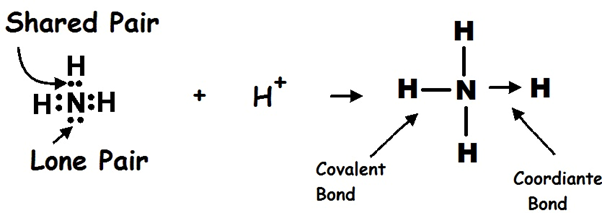
- In complex ions, the metal atom or ion acts as a Lewis acid by accepting electron pairs, while the ligand acts as a Lewis base by donating a lone pair.
- The bond formed is called a dative covalent bond because both electrons in the bond come from the ligand.
Examples of Ligands
- \( \mathrm{H_2O} \)
- \( \mathrm{NH_3} \)
- \( \mathrm{Cl^-} \)
- \( \mathrm{OH^-} \)
Example
Explain why ammonia can act as a ligand.
▶️ Answer / Explanation
Ammonia has a lone pair of electrons on the nitrogen atom.
This lone pair can be donated to a central metal ion to form a dative covalent bond, so ammonia acts as a ligand.
Example
State and explain the role of ligands in the formation of complex ions.
▶️ Answer / Explanation
Ligands donate lone pairs of electrons to a central metal atom or ion.
These lone pairs form dative covalent bonds, allowing complex ions to form around the metal centre.
Types of Ligands: Denticity
Ligands can be classified according to the number of dative covalent bonds they form with a central metal atom or ion. This is described using the terms monodentate, bidentate and polydentate.
(a) Monodentate Ligands
A monodentate ligand is a ligand that donates one lone pair of electrons to the central metal ion, forming one dative covalent bond.
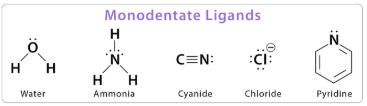
Examples of monodentate ligands include:
- \( \mathrm{H_2O} \)
- \( \mathrm{NH_3} \)
- \( \mathrm{Cl^-} \)
- \( \mathrm{CN^-} \)
Each of these ligands contains a single atom with a lone pair that can be donated to the metal ion.
(b) Bidentate Ligands
A bidentate ligand is a ligand that donates two lone pairs of electrons to a central metal ion, forming two dative covalent bonds.
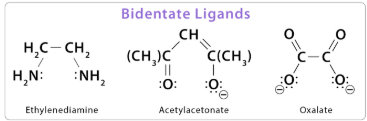
The two donor atoms in a bidentate ligand are usually separated by a short chain, allowing both to bond to the same metal ion.
Examples include:
- 1,2-diaminoethane (en), \( \mathrm{H_2NCH_2CH_2NH_2} \)
- Ethanedioate ion, \( \mathrm{C_2O_4^{2-}} \)
Bidentate ligands form chelate rings, which increase the stability of the complex.
(c) Polydentate Ligands
A polydentate ligand is a ligand that donates more than two lone pairs of electrons to a central metal ion, forming several dative covalent bonds.
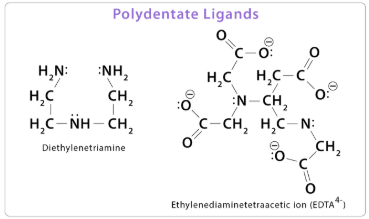
An important example is:
EDTA, \( \mathrm{EDTA^{4-}} \)
EDTA is a hexadentate ligand that can form six coordinate bonds to a single metal ion, producing very stable complexes.
Example
Classify each of the following ligands as monodentate or bidentate: \( \mathrm{NH_3} \) and \( \mathrm{H_2NCH_2CH_2NH_2} \).
▶️ Answer / Explanation
\( \mathrm{NH_3} \) is monodentate because it donates one lone pair.
\( \mathrm{H_2NCH_2CH_2NH_2} \) is bidentate because it donates two lone pairs.
Example
Explain why EDTA forms more stable complexes than ammonia.
▶️ Answer / Explanation
EDTA is a polydentate ligand that forms several dative covalent bonds with a single metal ion.
This creates multiple chelate rings, making the complex more stable than complexes formed with monodentate ligands such as ammonia.
Complexes
A complex is a molecule or ion formed by a central metal atom or ion surrounded by one or more ligands.

- In a complex, each ligand donates a lone pair of electrons to the central metal to form a dative (coordinate) covalent bond.
- The metal atom or ion acts as a Lewis acid, while the ligands act as Lewis bases.
Examples of Complexes
- \( \mathrm{[Cu(H_2O)_6]^{2+}} \)
- \( \mathrm{[Co(NH_3)_6]^{3+}} \)
- \( \mathrm{[Fe(CN)_6]^{4-}} \)
Example
Define the term complex.
▶️ Answer / Explanation
A complex is a molecule or ion formed by a central metal atom or ion surrounded by one or more ligands.
Example
Explain the roles of the metal ion and ligands in a complex ion.
▶️ Answer / Explanation
The metal ion acts as a Lewis acid by accepting lone pairs of electrons.
The ligands act as Lewis bases by donating lone pairs to form dative covalent bonds around the metal centre.
Geometry of Transition Element Complexes
Transition element complexes have specific geometries (shapes and bond angles) that depend mainly on the number of ligands surrounding the central metal ion.
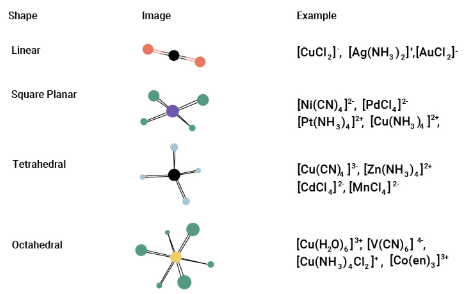
Linear Complexes
A linear complex has two ligands bonded to the central metal ion in a straight line.
- Coordination number: 2
- Bond angle: 180°
Example: complexes of \( \mathrm{Ag^+} \) such as \( \mathrm{[Ag(NH_3)_2]^+} \).
Square Planar Complexes
A square planar complex has four ligands arranged in one plane around the metal ion.
- Coordination number: 4
- Bond angles: 90°
Square planar geometry is common for some transition metals such as platinum(II).
Tetrahedral Complexes
A tetrahedral complex has four ligands arranged towards the corners of a tetrahedron.
- Coordination number: 4
- Bond angle: 109.5°
Example: \( \mathrm{[CoCl_4]^{2-}} \).
Octahedral Complexes
An octahedral complex has six ligands arranged symmetrically around the metal ion.
- Coordination number: 6
- Bond angles: 90° (and 180° between opposite ligands)
Example: \( \mathrm{[Cu(H_2O)_6]^{2+}} \).
Example
State the geometry and bond angles of a complex ion with coordination number 6.
▶️ Answer / Explanation
The geometry is octahedral.
The bond angles are 90° (and 180° between opposite ligands).
Example
A transition metal complex has the formula \( \mathrm{[MCl_4]^{2-}} \). Suggest two possible geometries and give the bond angles for each.
▶️ Answer / Explanation
The complex could be tetrahedral with bond angles of 109.5°.
Alternatively, it could be square planar with bond angles of 90°.
Coordination Number and Formulae of Complex Ions
Transition metal complexes can be described and predicted using the ideas of coordination number, ligand type and metal ion charge.
(a) Coordination Number
The coordination number is the number of coordinate (dative) bonds formed between a central metal atom or ion and surrounding ligands.
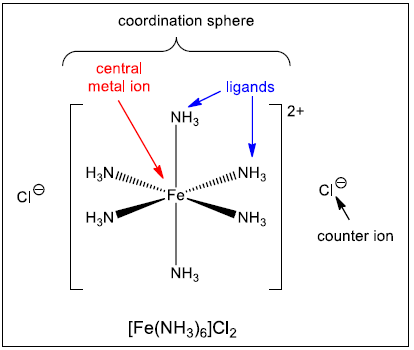
Each dative bond corresponds to one lone pair donated by a ligand.
For example:
- \( \mathrm{[Cu(H_2O)_6]^{2+}} \) has a coordination number of 6
- \( \mathrm{[Ag(NH_3)_2]^+} \) has a coordination number of 2
- \( \mathrm{[CoCl_4]^{2-}} \) has a coordination number of 4
(b) Predicting the Formula and Charge of a Complex Ion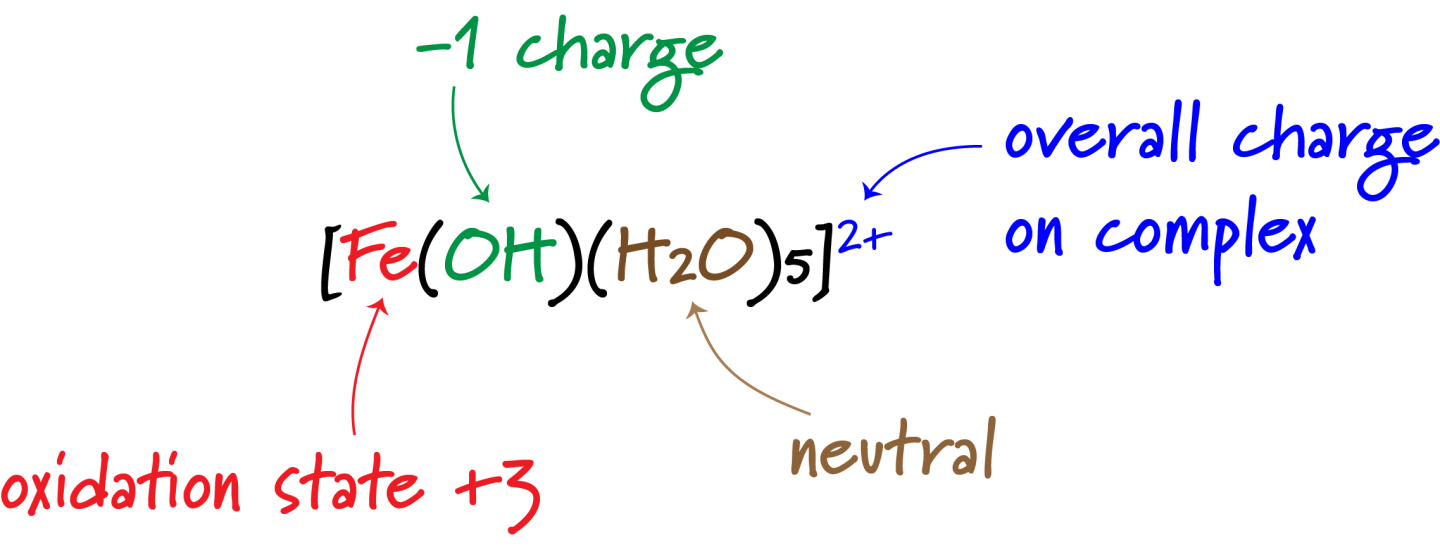
To predict the formula and charge of a complex ion:
- Identify the metal ion and its charge (or oxidation state)
- Determine the coordination number (from the ligand or geometry)
- Determine the charge on each ligand
- Calculate the overall charge of the complex
Neutral Ligands
Neutral ligands such as \( \mathrm{H_2O} \) and \( \mathrm{NH_3} \) do not affect the charge of the complex.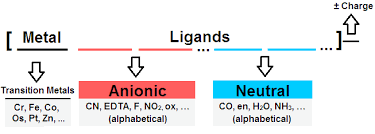
Example:
Copper(II), coordination number 6, ligand \( \mathrm{H_2O} \):
\( \mathrm{[Cu(H_2O)_6]^{2+}} \)
Negative Ligands
Negatively charged ligands such as \( \mathrm{Cl^-} \), \( \mathrm{OH^-} \) and \( \mathrm{CN^-} \) reduce the overall charge of the complex.
Example:
Cobalt(II), coordination number 4, ligand \( \mathrm{Cl^-} \):
Metal charge: \( \mathrm{+2} \)
Ligand charge: \( \mathrm{4 \times (-1) = -4} \)
Overall charge:
\( \mathrm{[CoCl_4]^{2-}} \)
Using Geometry to Predict Formula
The geometry often indicates the coordination number:
- Linear → coordination number 2
- Tetrahedral or square planar → coordination number 4
- Octahedral → coordination number 6
Example
Predict the formula and charge of the complex formed when \( \mathrm{Cu^{2+}} \) forms an octahedral complex with ammonia.
▶️ Answer / Explanation
Octahedral geometry means a coordination number of 6.
Ammonia is a neutral ligand, so it does not affect the charge.
The complex formed is:
\( \mathrm{[Cu(NH_3)_6]^{2+}} \)
Example
A nickel ion forms a square planar complex with cyanide ions. The oxidation state of nickel is +2. Predict the formula and charge of the complex ion.
▶️ Answer / Explanation
Square planar geometry corresponds to a coordination number of 4.
Each cyanide ion has a charge of \( \mathrm{-1} \).
Total ligand charge = \( \mathrm{4 \times (-1) = -4} \).
Metal charge = \( \mathrm{+2} \).
Overall charge = \( \mathrm{+2 – 4 = -2} \).
The complex ion is:
\( \mathrm{[Ni(CN)_4]^{2-}} \)
Ligand Exchange in Transition Metal Complexes
Ligand exchange occurs when one or more ligands in a complex ion are replaced by different ligands.
- This happens readily for transition metal complexes because the metal ions have vacant, energetically accessible d orbitals that can accept lone pairs from different ligands.
- In aqueous solution, transition metal ions are usually surrounded by water molecules. If another ligand is added that forms stronger dative bonds, it can replace one or more water ligands.
- Ligand exchange is usually observed as a colour change or formation/dissolution of a precipitate.
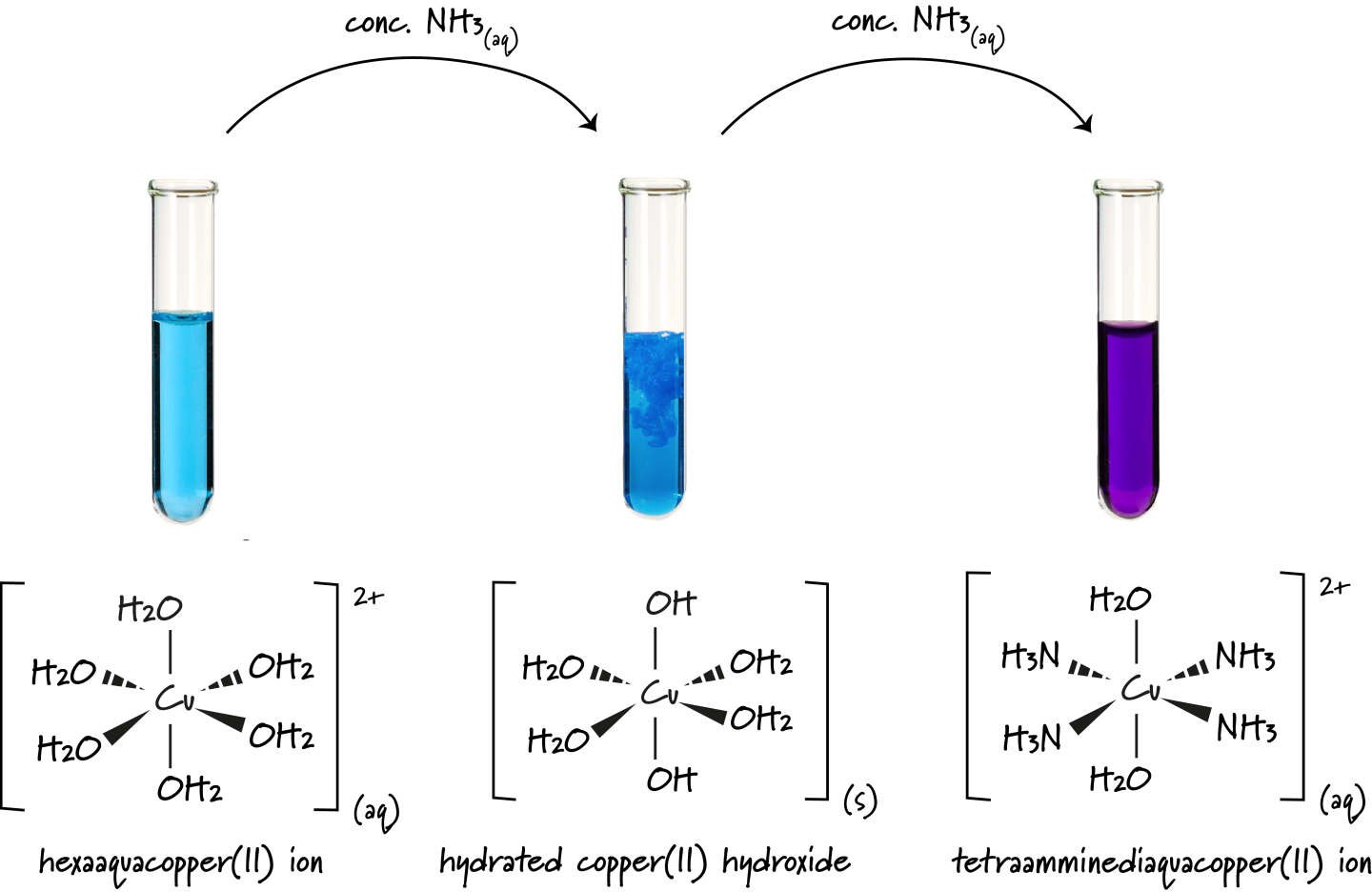
Copper(II) Ion Complexes
Copper(II) with Water
In water, copper(II) ions form a pale blue octahedral complex:
\( \mathrm{[Cu(H_2O)_6]^{2+}} \)
Copper(II) with Hydroxide Ions
When hydroxide ions are added, ligand exchange accompanied by deprotonation occurs, forming a pale blue precipitate:
\( \mathrm{[Cu(H_2O)_6]^{2+} + 2OH^- \rightarrow Cu(OH)_2(s) + 6H_2O} \)
Copper(II) with Ammonia
With a small amount of ammonia, the same pale blue precipitate of \( \mathrm{Cu(OH)_2} \) forms.
In excess ammonia, ligand exchange occurs and the precipitate dissolves, forming a deep blue complex:
\( \mathrm{[Cu(H_2O)_6]^{2+} + 4NH_3 \rightarrow [Cu(NH_3)_4(H_2O)_2]^{2+} + 4H_2O} \)
Ammonia replaces water because it forms stronger coordinate bonds with copper(II).
Cobalt(II) Ion Complexes
Cobalt(II) with Water
In aqueous solution, cobalt(II) ions form a pink complex:
\( \mathrm{[Co(H_2O)_6]^{2+}} \)
Cobalt(II) with Hydroxide Ions
Addition of hydroxide ions produces a blue precipitate of cobalt(II) hydroxide:
\( \mathrm{[Co(H_2O)_6]^{2+} + 2OH^- \rightarrow Co(OH)_2(s) + 6H_2O} \)
Cobalt(II) with Ammonia
Ammonia initially acts as a base, forming \( \mathrm{Co(OH)_2} \). In excess ammonia, ligand exchange occurs to form ammine complexes (which are readily oxidised in air).
Cobalt(II) with Chloride Ions
At high chloride ion concentration, ligand exchange occurs:
\( \mathrm{[Co(H_2O)_6]^{2+} + 4Cl^- \rightleftharpoons [CoCl_4]^{2-} + 6H_2O} \)
The colour changes from pink to blue as chloride ions replace water ligands.
Why Ligand Exchange Occurs
- Transition metal ions have vacant d orbitals
- Different ligands form bonds of different strengths
- Stronger ligands can replace weaker ligands
Example
Describe the changes observed when excess ammonia is added to an aqueous copper(II) solution.
▶️ Answer / Explanation
A pale blue precipitate of \( \mathrm{Cu(OH)_2} \) forms initially.
In excess ammonia, the precipitate dissolves to give a deep blue solution.
This is due to ligand exchange, where ammonia replaces water ligands to form \( \mathrm{[Cu(NH_3)_4(H_2O)_2]^{2+}} \).
Example
Explain the colour change observed when concentrated hydrochloric acid is added to aqueous cobalt(II) chloride.
▶️ Answer / Explanation
The pink \( \mathrm{[Co(H_2O)_6]^{2+}} \) complex is converted to the blue \( \mathrm{[CoCl_4]^{2-}} \) complex.
This occurs because chloride ions replace water ligands in a ligand exchange reaction.
A high concentration of chloride ions shifts the equilibrium towards the chloro complex.
Predicting the Feasibility of Redox Reactions Using Standard Electrode Potentials
- Redox reactions involving transition elements and their ions can be assessed quantitatively using standard electrode potentials, \( \mathrm{E^\circ} \).
- These values indicate the tendency of a species to be reduced.
Standard Electrode Potential, \( \mathrm{E^\circ} \)
The standard electrode potential, \( \mathrm{E^\circ} \), is the potential difference measured under standard conditions when a half-cell is connected to the standard hydrogen electrode.
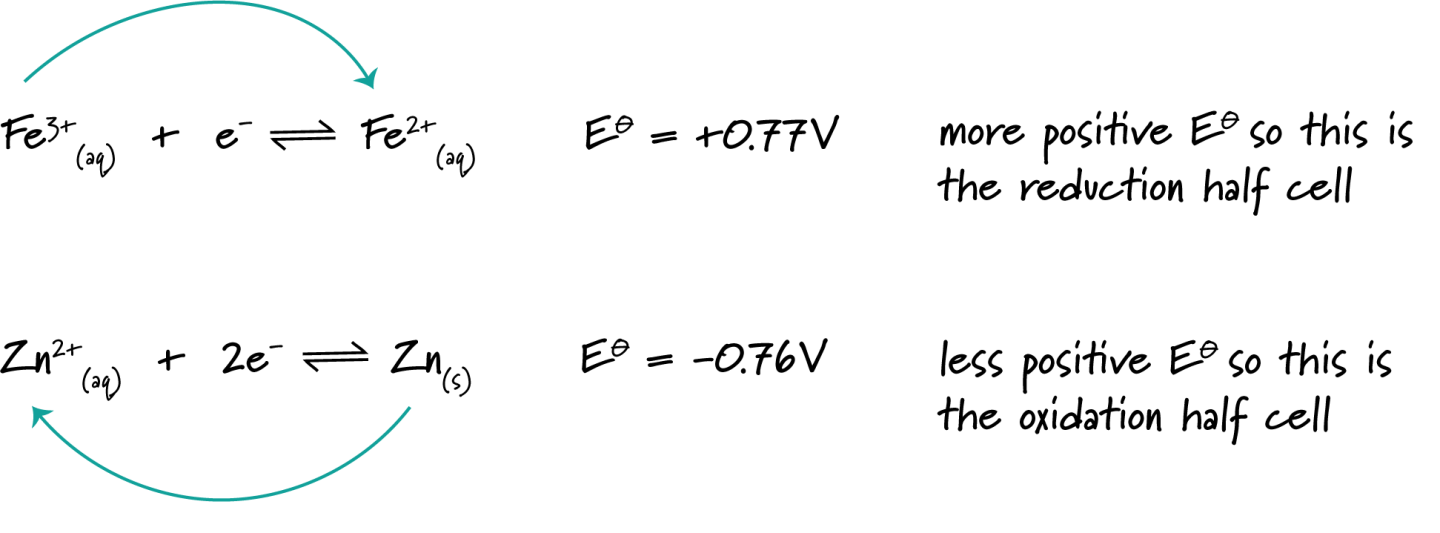
- All \( \mathrm{E^\circ} \) values are written as reduction potentials.
Predicting Feasibility of a Redox Reaction
A redox reaction is feasible if the overall cell potential, \( \mathrm{E^\circ_{cell}} \), is positive.
\( \mathrm{E^\circ_{cell} = E^\circ_{reduction} – E^\circ_{oxidation}} \)
In practice:
- The half-equation with the more positive \( \mathrm{E^\circ} \) occurs as reduction
- The half-equation with the more negative \( \mathrm{E^\circ} \) is reversed and occurs as oxidation
Key Rule for Feasibility
A redox reaction is feasible if electrons flow from the half-cell with the more negative \( \mathrm{E^\circ} \) to the half-cell with the more positive \( \mathrm{E^\circ} \).
Transition Elements and Variable Oxidation States
Transition elements often have several \( \mathrm{E^\circ} \) values corresponding to different oxidation states. This allows predictions about which oxidation state is favoured under given conditions.
For example, if:
\( \mathrm{Fe^{3+} + e^- \rightleftharpoons Fe^{2+}} \quad E^\circ = +0.77\ V \)
Iron(III) ions readily act as oxidising agents, as they are easily reduced.
Using \( \mathrm{E^\circ} \) Values Step-by-Step
- Write both half-equations as reductions
- Identify which species is reduced (more positive \( \mathrm{E^\circ} \))
- Reverse the other half-equation to show oxidation
- Calculate \( \mathrm{E^\circ_{cell}} \)
- If \( \mathrm{E^\circ_{cell} > 0} \), the reaction is feasible
Example
Predict whether \( \mathrm{Fe^{2+}} \) ions can reduce \( \mathrm{Cu^{2+}} \) ions, given the following data:
\( \mathrm{Fe^{3+} + e^- \rightleftharpoons Fe^{2+}} \quad E^\circ = +0.77\ V \)
\( \mathrm{Cu^{2+} + 2e^- \rightleftharpoons Cu} \quad E^\circ = +0.34\ V \)
▶️ Answer / Explanation
\( \mathrm{Fe^{3+}/Fe^{2+}} \) has a more positive \( \mathrm{E^\circ} \), so iron(III) is preferentially reduced.
Therefore, \( \mathrm{Fe^{2+}} \) will not reduce \( \mathrm{Cu^{2+}} \).
The reaction is not feasible.
Example
Determine whether \( \mathrm{Zn(s)} \) will react with \( \mathrm{Fe^{2+}(aq)} \), given:
\( \mathrm{Zn^{2+} + 2e^- \rightleftharpoons Zn} \quad E^\circ = -0.76\ V \)
\( \mathrm{Fe^{2+} + 2e^- \rightleftharpoons Fe} \quad E^\circ = -0.44\ V \)
▶️ Answer / Explanation
The \( \mathrm{Fe^{2+}/Fe} \) couple has the more positive \( \mathrm{E^\circ} \), so iron(II) ions are reduced.
Zinc is oxidised.
\( \mathrm{E^\circ_{cell} = (-0.44) – (-0.76) = +0.32\ V} \)
Since \( \mathrm{E^\circ_{cell}} \) is positive, the reaction is feasible.
Redox Reactions Involving Transition Metal Ions
Transition metal ions commonly take part in redox reactions. These reactions can be described using ionic equations and used in stoichiometric calculations such as titrations.
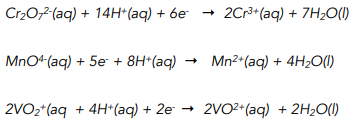
(a) \( \mathrm{MnO_4^- / C_2O_4^{2-}} \) in Acid Solution
In acidic conditions, the permanganate ion acts as a strong oxidising agent and is reduced from manganese(VII) to manganese(II).
Oxidation half-equation:
\( \mathrm{C_2O_4^{2-} \rightarrow 2CO_2 + 2e^-} \)
Reduction half-equation:
\( \mathrm{MnO_4^- + 8H^+ + 5e^- \rightarrow Mn^{2+} + 4H_2O} \)
Overall ionic equation:
\( \mathrm{2MnO_4^- + 5C_2O_4^{2-} + 16H^+ \rightarrow 2Mn^{2+} + 10CO_2 + 8H_2O} \)
This reaction is slow at room temperature and is usually warmed. The purple permanganate solution is self-indicating.
(b) \( \mathrm{MnO_4^- / Fe^{2+}} \) in Acid Solution
In acidic solution, permanganate ions oxidise iron(II) ions to iron(III).
Oxidation half-equation:
\( \mathrm{Fe^{2+} \rightarrow Fe^{3+} + e^-} \)
Reduction half-equation:
\( \mathrm{MnO_4^- + 8H^+ + 5e^- \rightarrow Mn^{2+} + 4H_2O} \)
Overall ionic equation:
\( \mathrm{MnO_4^- + 5Fe^{2+} + 8H^+ \rightarrow Mn^{2+} + 5Fe^{3+} + 4H_2O} \)
The end point is the first permanent pale purple colour of excess permanganate.
(c) \( \mathrm{Cu^{2+} / I^-} \)
Copper(II) ions oxidise iodide ions to iodine and are themselves reduced to copper(I) iodide.
Overall ionic equation:
\( \mathrm{2Cu^{2+} + 4I^- \rightarrow 2CuI(s) + I_2} \)
Observations:
- A white precipitate of \( \mathrm{CuI} \)
- A brown solution due to iodine
Example
Write the ionic equation for the reaction between \( \mathrm{MnO_4^-} \) and \( \mathrm{Fe^{2+}} \) in acidic solution.
▶️ Answer / Explanation
\( \mathrm{MnO_4^- + 5Fe^{2+} + 8H^+ \rightarrow Mn^{2+} + 5Fe^{3+} + 4H_2O} \)
Example
25.0 cm3 of \( \mathrm{Fe^{2+}} \) solution required 20.0 cm3 of 0.0200 mol dm–3 \( \mathrm{KMnO_4} \) for complete reaction in acidic solution. Calculate the concentration of the \( \mathrm{Fe^{2+}} \) solution.
▶️ Answer / Explanation
Moles of \( \mathrm{MnO_4^-} \):
\( \mathrm{0.0200 \times \dfrac{20.0}{1000} = 4.00 \times 10^{-4}\ mol} \)
From the equation, 1 mole of \( \mathrm{MnO_4^-} \) reacts with 5 moles of \( \mathrm{Fe^{2+}} \).
Moles of \( \mathrm{Fe^{2+}} \):
\( \mathrm{5 \times 4.00 \times 10^{-4} = 2.00 \times 10^{-3}\ mol} \)
Concentration of \( \mathrm{Fe^{2+}} \):
\( \mathrm{\dfrac{2.00 \times 10^{-3}}{25.0/1000} = 0.0800\ mol\,dm^{-3}} \)
Calculations Involving Other Redox Systems
Redox calculations for unfamiliar systems can be performed using the same general approach, provided suitable data are given (e.g. equations, oxidation states, or \( \mathrm{E^\circ} \) values).
General Method for Redox Calculations![]()
- Identify what is oxidised and what is reduced
- Write or use the balanced ionic equation
- Use the equation to determine the mole ratio
- Convert given data into moles
- Use stoichiometry to find the required quantity
Example Redox Systems
The method above applies to all redox systems, including:
- \( \mathrm{Cr_2O_7^{2-} / Fe^{2+}} \) in acid
- \( \mathrm{I_2 / S_2O_3^{2-}} \)
- \( \mathrm{Cl_2 / Br^-} \)
- \( \mathrm{Zn / Cu^{2+}} \)
Worked Approach Using an Unknown System
If the ionic equation is given: Use it directly for mole ratios.
If only half-equations or oxidation states are given: Balance electrons first, then combine the equations.
Example
Acidified potassium dichromate reacts with iron(II) ions according to the equation:
\( \mathrm{Cr_2O_7^{2-} + 6Fe^{2+} + 14H^+ \rightarrow 2Cr^{3+} + 6Fe^{3+} + 7H_2O} \)
Calculate the amount (in moles) of \( \mathrm{Fe^{2+}} \) required to react completely with 0.0100 mol of \( \mathrm{Cr_2O_7^{2-}} \).
▶️ Answer / Explanation
From the equation:
\( \mathrm{1\ mol\ Cr_2O_7^{2-} : 6\ mol\ Fe^{2+}} \)
Moles of \( \mathrm{Fe^{2+}} \):
\( \mathrm{6 \times 0.0100 = 0.0600\ mol} \)
Example
Chlorine gas reacts with bromide ions according to the equation:
\( \mathrm{Cl_2 + 2Br^- \rightarrow 2Cl^- + Br_2} \)
100.0 cm3 of bromide ion solution reacts exactly with 25.0 cm3 of chlorine gas at room temperature and pressure (molar gas volume = 24.0 dm3 mol–1). Calculate the concentration of the bromide ion solution.
▶️ Answer / Explanation
Moles of chlorine gas:
\( \mathrm{\dfrac{25.0}{24000} = 1.04 \times 10^{-3}\ mol} \)
From the equation:
\( \mathrm{1\ mol\ Cl_2 : 2\ mol\ Br^-} \)
Moles of \( \mathrm{Br^-} \):
\( \mathrm{2 \times 1.04 \times 10^{-3} = 2.08 \times 10^{-3}\ mol} \)
Concentration of \( \mathrm{Br^-} \):
\( \mathrm{\dfrac{2.08 \times 10^{-3}}{0.100} = 0.0208\ mol\,dm^{-3}} \)
