CIE AS/A Level Chemistry 3.1 Electronegativity and bonding Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 3.1 Electronegativity and bonding Study Notes – New Syllabus
CIE AS/A Level Chemistry 3.1 Electronegativity and bonding Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- define electronegativity as the power of an atom to attract electrons to itself
- explain the factors influencing the electronegativities of the elements in terms of nuclear charge, atomic radius and shielding by inner shells and sub-shells
- state and explain the trends in electronegativity across a period and down a group of the Periodic Table
- use the differences in Pauling electronegativity values to predict the formation of ionic and covalent bonds (the presence of covalent character in some ionic compounds will not be assessed) (Pauling electronegativity values will be given where necessary)
Electronegativity
Electronegativity is a property of atoms that describes how strongly they attract shared electrons in a covalent bond. It helps explain bond polarity, molecule polarity, and patterns in reactivity.
Definition of Electronegativity

Electronegativity is defined as: “The power of an atom to attract electrons to itself in a covalent bond.”
- Electronegativity values are dimensionless (no units).
- They are based on the Pauling scale (most commonly used).
- Higher values mean stronger attraction for bonding electrons.
Key Points
- Non-metals generally have high electronegativity.
- Metals generally have low electronegativity.
- Electronegativity affects bond polarity: – If difference = 0 → non-polar covalent – If difference is moderate → polar covalent – If difference is large → ionic bond
Example
Define electronegativity.
▶️ Answer / Explanation
Electronegativity is the power of an atom to attract electrons to itself in a covalent bond.
Example
Which atom is more electronegative: oxygen or sulfur? Explain your answer.
▶️ Answer / Explanation
Oxygen is more electronegative because it is smaller and the bonding electrons are closer to the nucleus, so attraction is stronger. Electronegativity decreases down a group.
Example
Predict whether the bond in \( \mathrm{HCl} \) is polar, and explain using electronegativity.
▶️ Answer / Explanation
Chlorine is more electronegative than hydrogen.
This means the shared pair of electrons is pulled closer to chlorine.
As a result, the \( \mathrm{H–Cl} \) bond is polar, with:
\( \delta^+ \mathrm{H} – \mathrm{Cl} \, \delta^- \)
Electronegativity difference causes separation of charge across the bond.
Factors Affecting Electronegativity
Electronegativity varies across the Periodic Table because atoms differ in their nuclear charge, atomic radius, and the amount of shielding from inner electrons.
1. Nuclear Charge
![]()
- A higher nuclear charge (more protons) means a stronger attraction for bonding electrons.
- As proton number increases across a period, electronegativity increases.
- Greater positive charge pulls the bonding electrons more strongly toward the nucleus.
Higher nuclear charge → stronger attraction → higher electronegativity
2. Atomic Radius
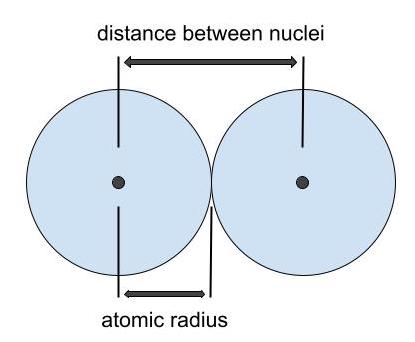
- Electrons in a larger atom are farther from the nucleus.
- The attraction between nucleus and bonding electrons decreases with distance.
- As atomic radius increases down a group, electronegativity decreases.
Smaller atom → electrons closer → stronger attraction → higher electronegativity
3. Shielding (Electron Repulsion by Inner Shells)
![]()
- Inner-shell electrons repel outer electrons and reduce the effective nuclear attraction.
- More inner shells or filled sub-shells = greater shielding = weaker pull on bonding electrons.
- Down a group, shielding increases because more shells are added, reducing electronegativity.
More shielding → weaker effective nuclear charge → lower electronegativity
Summary Table
| Factor | Increase in factor → Effect on Electronegativity |
|---|---|
| Nuclear charge | ↑ nuclear charge → ↑ electronegativity |
| Atomic radius | ↑ radius → ↓ electronegativity |
| Shielding | ↑ shielding → ↓ electronegativity |
Example
Why is fluorine the most electronegative element?
▶️ Answer / Explanation
Fluorine has a very small atomic radius and high nuclear charge relative to its size.
Its bonding electrons are close to the nucleus and experience little shielding.
Therefore, fluorine attracts electrons very strongly.
Example
Explain why electronegativity decreases from nitrogen to phosphorus.
▶️ Answer / Explanation
Going from nitrogen to phosphorus:
- Atomic radius increases → bonding electrons are farther from the nucleus.
- Shielding increases because phosphorus has an additional energy shell.
These effects outweigh the increase in nuclear charge.
Therefore electronegativity decreases down the group.
Example
Explain why oxygen is more electronegative than sulfur but less electronegative than fluorine.
▶️ Answer / Explanation
Oxygen vs sulfur:
- Oxygen has a smaller atomic radius.
- Less shielding in oxygen.
- Bonding electrons are closer and more strongly attracted.
→ Oxygen is more electronegative than sulfur.
Oxygen vs fluorine:
- Fluorine has a smaller atomic radius than oxygen.
- Fluorine has greater effective nuclear charge.
- Less shielding in fluorine’s 2p electrons.
→ Fluorine is more electronegative than oxygen.
Overall: Atomic radius + shielding + effective nuclear charge explain the trend.
Trends in Electronegativity Across a Period and Down a Group
Electronegativity varies in a predictable way across the Periodic Table due to changes in nuclear charge, atomic radius, and shielding.
![]()
1. Trend Across a Period (Left → Right)
- Electronegativity increases across a period.
- Nuclear charge increases (more protons).
- Atomic radius decreases (nucleus pulls electrons closer).
- Shielding remains approximately constant because electrons are added to the same shell.
Across a period: ↑ nuclear charge, ↓ radius, ≈ constant shielding → ↑ electronegativity
Example: In Period 2, electronegativity increases from Li → Be → B → C → N → O → F.
2. Trend Down a Group (Top → Bottom)
- Electronegativity decreases down a group.
- Atomic radius increases (more shells).
- Shielding increases significantly due to more inner electrons.
- Increased distance and shielding outweigh the increase in nuclear charge.
Down a group: ↑ radius, ↑ shielding, ↑ nuclear charge but less effective → ↓ electronegativity
Example: In Group 17, electronegativity decreases from F → Cl → Br → I.
3. Explanation Summary
| Direction in Periodic Table | Trend in Electronegativity | Main Reasons |
|---|---|---|
| Across a period | Increases | ↑ nuclear charge, ↓ atomic radius, constant shielding |
| Down a group | Decreases | ↑ atomic radius, ↑ shielding, weaker effective attraction |
Example
State the trend in electronegativity across a period.
▶️ Answer / Explanation
Electronegativity increases across a period because nuclear charge increases and atomic radius decreases.
Example
Explain why electronegativity decreases down Group 2.
▶️ Answer / Explanation
Down Group 2:
- Atomic radius increases (more shells).
- Shielding increases (more inner electrons).
These reduce the effective nuclear attraction for bonding electrons.
Therefore electronegativity decreases down the group.
Example
Explain why chlorine is more electronegative than sulfur but less electronegative than fluorine.
▶️ Answer / Explanation
Chlorine vs sulfur (across a period):
- Chlorine is to the right of sulfur in the same period.
- It has a higher nuclear charge and smaller atomic radius.
- Shielding is similar.
→ Chlorine is more electronegative.
Chlorine vs fluorine (down a group):
- Fluorine is above chlorine.
- Fluorine has a much smaller atomic radius and less shielding.
- Bonding electrons feel a stronger effective nuclear attraction.
→ Fluorine is more electronegative than chlorine.
Predicting Ionic and Covalent Bonding Using Electronegativity Differences
The type of bond formed between two atoms depends on the difference in their Pauling electronegativity values. A larger difference means electrons are transferred (ionic), while a smaller difference means electrons are shared (covalent).
Electronegativity Difference (\( \Delta EN \))
- Pauling electronegativity values measure how strongly an atom attracts electrons.
- Subtract the electronegativity of one atom from the other: \( \Delta EN = |EN_1 – EN_2| \)
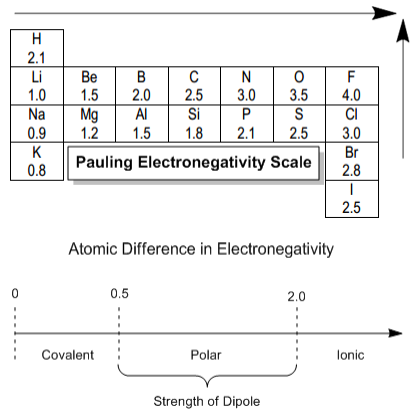
General Rules (Pauling Scale)
| Electronegativity Difference \( \Delta EN \) | Likely Bond Type |
|---|---|
| \( 0 \leq \Delta EN \lesssim 0.4 \) | Non-polar covalent |
| \( 0.4 \lesssim \Delta EN \lesssim 1.7 \) | Polar covalent |
| \( \Delta EN \gtrsim 1.7 \) | Ionic bond |
(Values vary slightly depending on source; exam questions will provide electronegativity values where needed.)
Understanding the Trend
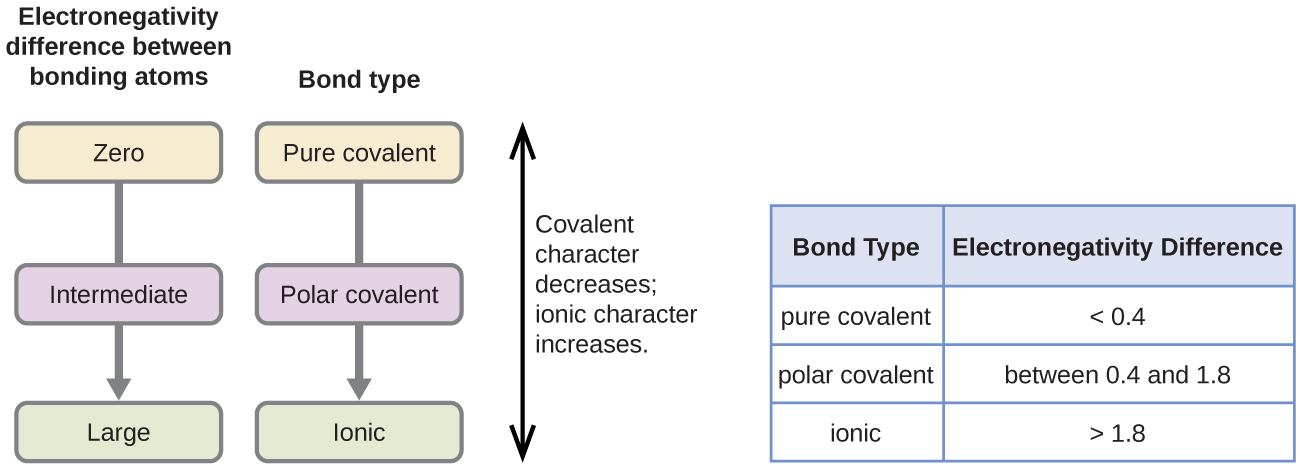
- Large \( \Delta EN \) → one atom pulls electrons strongly → electron transfer → ionic bond
- Small \( \Delta EN \) → atoms share electrons → covalent bond
- Zero \( \Delta EN \) → equal sharing → non-polar covalent
Example
The electronegativities of sodium and chlorine are 0.9 and 3.0. Predict the type of bond formed in \( \mathrm{NaCl} \).
▶️ Answer / Explanation
\( \Delta EN = |3.0 – 0.9| = 2.1 \)
This is greater than 1.7 → ionic bond.
Electrons are transferred from sodium to chlorine.
Example
Predict the bond type in hydrogen chloride, given electronegativities: H = 2.1, Cl = 3.0.
▶️ Answer / Explanation
\( \Delta EN = |3.0 – 2.1| = 0.9 \)
\( 0.4 \lesssim 0.9 \lesssim 1.7 \) → polar covalent bond.
Electrons are shared but unequally → dipole forms.
Example
Using the electronegativities below, arrange the bonds \( \mathrm{C–H} \), \( \mathrm{C–O} \), and \( \mathrm{K–O} \) in order of increasing ionic character.
C = 2.5, H = 2.1, O = 3.5, K = 0.8.
▶️ Answer / Explanation
Step 1: Calculate \( \Delta EN \)
\( \mathrm{C–H}: |2.5 – 2.1| = 0.4 \) (non-polar covalent)
\( \mathrm{C–O}: |3.5 – 2.5| = 1.0 \) (polar covalent)
\( \mathrm{K–O}: |3.5 – 0.8| = 2.7 \) (ionic)
Step 2: Rank in increasing ionic character
\( \mathrm{C–H} < \mathrm{C–O} < \mathrm{K–O} \)
Reason: larger electronegativity difference → more ionic character.
