CIE AS/A Level Chemistry 3.2 Ionic bonding Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 3.2 Ionic bonding Study Notes – New Syllabus
CIE AS/A Level Chemistry 3.2 Ionic bonding Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- define ionic bonding as the electrostatic attraction between oppositely charged ions (positively charged cations and negatively charged anions)
- describe ionic bonding including the examples of sodium chloride, magnesium oxide and calcium fluoride
Ionic Bonding
Ionic bonding occurs when electrons are transferred from one atom to another, forming charged ions. These oppositely charged ions attract each other strongly.
Definition of Ionic Bonding
Ionic bonding is the electrostatic attraction between oppositely charged ions (positively charged cations and negatively charged anions).
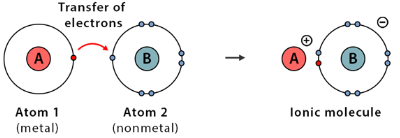
- Cations form when atoms lose electrons (usually metals).
- Anions form when atoms gain electrons (usually non-metals).
- The ionic bond is the strong electrostatic force holding these ions together.
Key Features of Ionic Bonding
- Occurs between metals and non-metals.
- Involves the transfer of electrons.
- Produces giant ionic lattices with strong attractions in all directions.
- Strength of ionic bond depends on charge and ionic radius.
Example
Define ionic bonding.
▶️ Answer / Explanation
Ionic bonding is the electrostatic attraction between oppositely charged ions (cations and anions).
Example
Explain how ionic bonding forms between sodium and chlorine.
▶️ Answer / Explanation
Sodium loses one electron to form \( \mathrm{Na^+} \).
Chlorine gains one electron to form \( \mathrm{Cl^-} \).
The opposite charges attract strongly → ionic bond.
Bond = electrostatic attraction between \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \)
Example
Explain why the ionic bond in \( \mathrm{MgO} \) is stronger than in \( \mathrm{NaCl} \).
▶️ Answer / Explanation
\( \mathrm{MgO} \): ions are \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \) \( \mathrm{NaCl} \): ions are \( \mathrm{Na^{+}} \) and \( \mathrm{Cl^{-}} \)
The charges in \( \mathrm{MgO} \) are larger (\( 2^+ \) and \( 2^- \)) than in \( \mathrm{NaCl} \) (\( 1^+ \) and \( 1^- \)).
Stronger electrostatic attraction exists between ions with higher charges.
Therefore, \( \mathrm{MgO} \) has stronger ionic bonding than \( \mathrm{NaCl} \).
Describing Ionic Bonding (with NaCl, MgO, CaF₂)
Ionic bonding forms when electrons are transferred from metal atoms to non-metal atoms, producing oppositely charged ions. These ions attract each other strongly in all directions, forming a giant ionic lattice.
How Ionic Bonds Form
- A metal atom loses electrons → forms a positively charged cation.
- A non-metal atom gains electrons → forms a negatively charged anion.
- These ions attract through strong electrostatic forces.
- This attraction is the ionic bond.
Ionic bond = electrostatic attraction between \( \mathrm{cations} \) and \( \mathrm{anions} \)
Examples of Ionic Bonding
1. Sodium Chloride — \( \mathrm{NaCl} \)
- Sodium atom loses 1 electron → \( \mathrm{Na^+} \)
- Chlorine atom gains 1 electron → \( \mathrm{Cl^-} \)
- Attraction between \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) forms the ionic bond.

\( \mathrm{Na \rightarrow Na^+ + e^-} \)
\( \mathrm{Cl + e^- \rightarrow Cl^-} \)
2. Magnesium Oxide — \( \mathrm{MgO} \)
- Magnesium loses 2 electrons → \( \mathrm{Mg^{2+}} \)
- Oxygen gains 2 electrons → \( \mathrm{O^{2-}} \)
- Greater charges result in very strong ionic bonding.
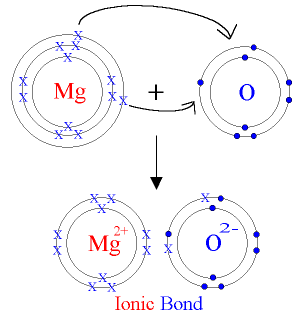
\( \mathrm{Mg \rightarrow Mg^{2+} + 2e^-} \)
\( \mathrm{O + 2e^- \rightarrow O^{2-}} \)
3. Calcium Fluoride — \( \mathrm{CaF_2} \)
- Calcium loses 2 electrons → \( \mathrm{Ca^{2+}} \)
- Each fluorine atom gains 1 electron → \( \mathrm{F^-} \)
- Two fluoride ions are needed to balance the \( \mathrm{Ca^{2+}} \) charge.
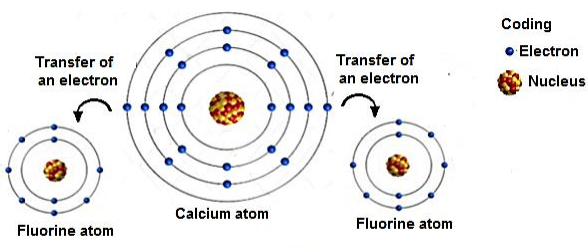
\( \mathrm{Ca \rightarrow Ca^{2+} + 2e^-} \)
\( \mathrm{F + e^- \rightarrow F^-} \)
Overall: \( \mathrm{Ca^{2+} + 2F^- \rightarrow CaF_2} \)
Example
Describe how ionic bonding forms in sodium chloride.
▶️ Answer / Explanation
Sodium loses one electron to form \( \mathrm{Na^+} \). Chlorine gains one electron to form \( \mathrm{Cl^-} \). The electrostatic attraction between \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) is the ionic bond.
Example
Explain why the formula of calcium fluoride is \( \mathrm{CaF_2} \) and how the ionic bond forms.
▶️ Answer / Explanation
Calcium forms \( \mathrm{Ca^{2+}} \) by losing 2 electrons.
Each fluorine forms \( \mathrm{F^-} \) by gaining 1 electron.
Two fluoride ions are needed to balance the \( \mathrm{Ca^{2+}} \) charge.
The ionic bond is the attraction between \( \mathrm{Ca^{2+}} \) and two \( \mathrm{F^-} \) ions.
Example
Explain why the ionic bonding in \( \mathrm{MgO} \) is stronger than in \( \mathrm{CaF_2} \).
▶️ Answer / Explanation
Magnesium oxide: ions are \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \). Calcium fluoride: ions are \( \mathrm{Ca^{2+}} \) and \( \mathrm{F^-} \).
In \( \mathrm{MgO} \), both ions carry a \( 2^+ \) and \( 2^- \) charge → stronger electrostatic attraction.
In \( \mathrm{CaF_2} \), the attraction is between \( \mathrm{Ca^{2+}} \) and \( \mathrm{F^-} \) (only single negative charge).
Thus, \( \mathrm{MgO} \) has stronger ionic bonding than \( \mathrm{CaF_2} \).
