CIE AS/A Level Chemistry 3.5 Shapes of molecules Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 3.5 Shapes of molecules Study Notes – New Syllabus
CIE AS/A Level Chemistry 3.5 Shapes of molecules Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:6
- state and explain the shapes of, and bond angles in, molecules by using VSEPR theory, including as simple examples:
• BF3 (trigonal planar, 120°)
• CO2 (linear, 180°)
• CH4 (tetrahedral, 109.5°)
• NH3 (pyramidal, 107°)
• H2O (non-linear, 104.5°)
• SF6 (octahedral, 90°)
• PF5 (trigonal bipyramidal, 120° and 90°) - predict the shapes of, and bond angles in, molecules and ions analogous to those specified in 3.5.1
VSEPR Theory — Molecular Shapes and Bond Angles
VSEPR (Valence Shell Electron Pair Repulsion) theory states that electron pairs around a central atom repel each other and arrange themselves as far apart as possible. This determines the shape and bond angles of molecules.
Key Principles of VSEPR
![]()
- Electron pairs (bonding and lone pairs) repel each other.
- Lone pair–lone pair repulsion > lone pair–bond pair repulsion > bond pair–bond pair repulsion.
- More repulsion → smaller bond angle.
Required Molecular Shapes
1. \( \mathrm{BF_3} \) — Trigonal Planar — \( 120^\circ \)

- Boron has 3 bonding pairs and no lone pairs.
- Bond pairs spread out evenly in a plane → 120° apart.
- Shape: trigonal planar
2. \( \mathrm{CO_2}\ ) — Linear — \( 180^\circ \)
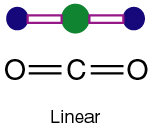
- Carbon has 2 bonding regions (double bonds count as 1 region each).
- No lone pairs on carbon.
- Electron pairs arrange opposite each other → 180°.
- Shape: linear
3. \( \mathrm{CH_4} \) — Tetrahedral — \( 109.5^\circ \)
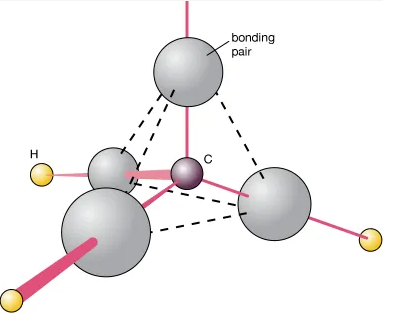
- Four bonding pairs around carbon.
- No lone pairs.
- Electron pairs repel symmetrically → tetrahedron.
- Bond angle: \( 109.5^\circ \)
4. \( \mathrm{NH_3} \) — Pyramidal — \( 107^\circ \)

- 3 bonding pairs, 1 lone pair.
- Lone pair repels more strongly → pushes bonds slightly closer.
- Shape: pyramidal
- Bond angle reduced to \( 107^\circ \)
5. \( \mathrm{H_2O} \) — Non-linear (Bent) — \( 104.5^\circ \)
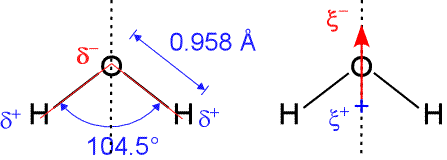
- 2 bonding pairs, 2 lone pairs.
- Two lone pairs create strong repulsion → angle decreases.
- Shape: bent / V-shaped
- Bond angle: \( 104.5^\circ \)
6. \( \mathrm{SF_6} \) — Octahedral — \( 90^\circ \)

- 6 bonding pairs around sulfur.
- No lone pairs.
- Electron pairs arrange at right angles → octahedral.
- Bond angle: \( 90^\circ \)
7. \( \mathrm{PF_5} \) — Trigonal Bipyramidal — \( 120^\circ \) and \( 90^\circ \)

- 5 bonding pairs and no lone pairs.
- Three equatorial positions → \( 120^\circ \)
- Two axial positions → \( 90^\circ \)
- Shape: trigonal bipyramidal
Example
What is the shape and bond angle of \( \mathrm{CH_4} \)?
▶️ Answer / Explanation
4 bonding pairs, no lone pairs → tetrahedral.
Bond angle = \( 109.5^\circ \).
Example
Explain why the bond angle in \( \mathrm{NH_3} \) is smaller than in \( \mathrm{CH_4} \).
▶️ Answer / Explanation
\( \mathrm{NH_3} \) has one lone pair, while \( \mathrm{CH_4} \) has none.
Lone pair–bond pair repulsion is greater than bond pair–bond pair repulsion.
This pushes the N–H bonds closer → reducing angle from \( 109.5^\circ \) to \( 107^\circ \).
Example
Explain the difference in bond angles between \( \mathrm{NH_3} \) (107°) and \( \mathrm{H_2O} \) (104.5°).
▶️ Answer / Explanation
\( \mathrm{H_2O} \) has 2 lone pairs, whereas \( \mathrm{NH_3} \) has only 1.
Lone pairs repel more strongly than bonding pairs. More lone pairs → more repulsion → smaller bond angle.
Thus, \( \mathrm{H_2O} \) (2 lone pairs) has a smaller angle (\( 104.5^\circ \)) than \( \mathrm{NH_3} \) (\( 107^\circ \)).
Predicting Shapes and Bond Angles of Analogous Molecules and Ions (VSEPR Theory)
Molecules and ions that have the same number of bonding pairs and lone pairs around the central atom have the same shape and similar bond angles. These are called analogous species. VSEPR theory allows us to predict their geometry by counting electron pairs around the central atom.
Steps for Predicting Shape
![]()
- Determine the central atom.
- Count bonding pairs (each single, double, or triple bond = 1 bonding region).
- Count lone pairs on the central atom.
- Total electron pairs = bonding + lone pairs.
- Match this to a known VSEPR shape.
Lone pairs reduce bond angles due to stronger repulsion.
Common Analogous Shapes

| Electron Pair Arrangement | Bonding / Lone Pairs | Shape | Bond Angle | Example |
|---|---|---|---|---|
| Tetrahedral | 4 BP, 0 LP | Tetrahedral | \( 109.5^\circ \) | \( \mathrm{CH_4} \) |
| Tetrahedral | 3 BP, 1 LP | Pyramidal | \( 107^\circ \) | \( \mathrm{NH_3} \) |
| Tetrahedral | 2 BP, 2 LP | Bent / V-shaped | \( 104.5^\circ \) | \( \mathrm{H_2O} \) |
| Trigonal Planar | 3 BP, 0 LP | Trigonal planar | \( 120^\circ \) | \( \mathrm{BF_3} \) |
| Linear | 2 BP, 0 LP | Linear | \( 180^\circ \) | \( \mathrm{CO_2} \) |
| Trigonal bipyramidal | 5 BP, 0 LP | Trigonal bipyramidal | \( 120^\circ \) & \( 90^\circ \) | \( \mathrm{PF_5} \) |
| Octahedral | 6 BP, 0 LP | Octahedral | \( 90^\circ \) | \( \mathrm{SF_6} \) |
Examples of Analogous Molecules & Ions
1. Trigonal Planar Analogues (like \( \mathrm{BF_3} \))
- Examples: \( \mathrm{NO_3^-} \), \( \mathrm{CO_3^{2-}} \), \( \mathrm{SO_3} \)
- Shape: Trigonal planar
- Bond angle: \( 120^\circ \)
2. Linear Analogues (like \( \mathrm{CO_2} \))
- Examples: \( \mathrm{BeCl_2} \), \( \mathrm{CS_2} \), \( \mathrm{HCN} \)
- Shape: Linear
- Bond angle: \( 180^\circ \)
3. Tetrahedral Analogues (like \( \mathrm{CH_4} \))
- Examples: \( \mathrm{SiH_4} \), \( \mathrm{NH_4^+} \), \( \mathrm{ClO_4^-} \)
- Shape: Tetrahedral
- Bond angle: \( 109.5^\circ \)
4. Pyramidal Analogues (like \( \mathrm{NH_3} \))
- Examples: \( \mathrm{PH_3} \), \( \mathrm{AsH_3} \)
- Shape: Pyramidal
- Bond angle: \( 107^\circ \)
5. Bent Analogues (like \( \mathrm{H_2O} \))
- Examples: \( \mathrm{OF_2} \), \( \mathrm{SCl_2} \)
- Shape: Bent / V-shaped
- Bond angle: \( 104.5^\circ \)
6. Trigonal Bipyramidal Analogues (like \( \mathrm{PF_5} \))
- Examples: \( \mathrm{PCl_5} \), \( \mathrm{AsF_5} \)
- Shape: Trigonal bipyramidal
- Bond angles: \( 120^\circ \) (equatorial), \( 90^\circ \) (axial)
7. Octahedral Analogues (like \( \mathrm{SF_6} \))
- Examples: \( \mathrm{SiF_6^{2-}} \), \( \mathrm{PF_6^-} \)
- Shape: Octahedral
- Bond angle: \( 90^\circ \)
Example
Predict the shape of \( \mathrm{NH_4^+} \).
▶️ Answer / Explanation
\( \mathrm{NH_4^+} \) has 4 bonding pairs, 0 lone pairs → tetrahedral.
Bond angle = \( 109.5^\circ \).
Example
Predict the shape and bond angle of \( \mathrm{NO_3^-} \).
▶️ Answer / Explanation
3 bonding regions around N, 0 lone pairs → trigonal planar.
Bond angle = \( 120^\circ \).
Example
Predict and explain the shape and bond angle of \( \mathrm{ClO_2^-} \).
▶️ Answer / Explanation
Central Cl has 2 bonding pairs and 2 lone pairs → total 4 electron pairs.
Shape = bent (like \( \mathrm{H_2O} \)).
Lone pairs compress bond angle → approx. \( 104^\circ \).
