CIE AS/A Level Chemistry 34.2 Phenylamine and azo compounds Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 34.2 Phenylamine and azo compounds Study Notes – New Syllabus
CIE AS/A Level Chemistry 34.2 Phenylamine and azo compounds Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
describe the preparation of phenylamine via the nitration of benzene to form nitrobenzene followed
by reduction with hot Sn/concentrated HCl followed by NaOH(aq)describe:
(a) the reaction of phenylamine with Br₂(aq) at room temperature
(b) the reaction of phenylamine with HNO₂ or NaNO₂ and dilute acid below 10°C to produce the
diazonium salt; further warming of the diazonium salt with H₂O to give phenoldescribe and explain the relative basicities of aqueous ammonia, ethylamine and phenylamine
recall the following about azo compounds:
(a) describe the coupling of benzenediazonium chloride with phenol in NaOH(aq) to form an azo
compound
(b) identify the azo group
(c) state that azo compounds are often used as dyes
(d) that other azo dyes can be formed via a similar route
Preparation of Phenylamine from Benzene
Phenylamine can be prepared from benzene in a two-step synthesis involving nitration followed by reduction. You must be able to describe the reagents and conditions for each step and explain the purpose of the final treatment with sodium hydroxide.
Step 1: Nitration of Benzene → Nitrobenzene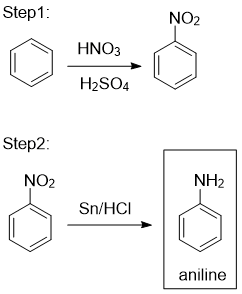
Benzene is nitrated by an electrophilic substitution reaction.
Reagents and conditions:
- concentrated \( \mathrm{HNO_3} \)
- concentrated \( \mathrm{H_2SO_4} \)
- temperature between 50–60°C
\( \mathrm{C_6H_6 + HNO_3 \xrightarrow{H_2SO_4} C_6H_5NO_2 + H_2O} \)
The product formed is nitrobenzene. Sulfuric acid generates the nitronium ion, which acts as the electrophile.
Step 2: Reduction of Nitrobenzene → Phenylamine
Nitrobenzene is reduced to phenylamine using a metal and acid.
Reagents and conditions:
- tin (Sn)
- concentrated HCl
- heat under reflux
During this step, the nitro group is reduced to an ammonium salt:
\( \mathrm{C_6H_5NO_2 \rightarrow C_6H_5NH_3^+Cl^-} \)
Final Step: Treatment with NaOH(aq)
The reaction mixture is then treated with aqueous sodium hydroxide.
\( \mathrm{C_6H_5NH_3^+Cl^- + NaOH \rightarrow C_6H_5NH_2 + NaCl + H_2O} \)
This neutralises the acid and liberates free phenylamine from its ammonium salt.
Overall Conversion
Benzene → nitrobenzene → phenylamine
Key Points
- nitration is an electrophilic substitution
- reduction uses Sn / concentrated HCl and heat
- NaOH is required to release the free amine
- phenylamine is initially formed as an ammonium salt
Example
State the reagents needed to convert nitrobenzene into phenylamine.
▶️ Answer / Explanation
Tin and concentrated hydrochloric acid, followed by aqueous sodium hydroxide.
Example
Describe how benzene can be converted into phenylamine, including reagents and conditions.
▶️ Answer / Explanation
Benzene is first nitrated using concentrated nitric acid and concentrated sulfuric acid at 50–60°C to form nitrobenzene.
Nitrobenzene is then reduced by heating with tin and concentrated hydrochloric acid under reflux, forming an ammonium salt.
The mixture is finally treated with aqueous sodium hydroxide to produce phenylamine.
Chemistry of Phenylamine
Phenylamine (aniline) shows distinctive reactions due to the presence of the –NH₂ group directly attached to the benzene ring. The nitrogen lone pair interacts strongly with the aromatic π system, making phenylamine highly activating towards electrophilic substitution.
(a) Reaction of Phenylamine with Br₂(aq)
Phenylamine reacts rapidly with bromine water at room temperature.
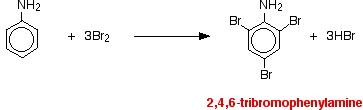
\( \mathrm{C_6H_5NH_2 + 3Br_2 \rightarrow C_6H_2Br_3NH_2 + 3HBr} \)
The product formed is 2,4,6-tribromophenylamine, which appears as a white precipitate.
No halogen carrier catalyst is required.
Explanation
The lone pair on the nitrogen atom overlaps with the delocalised π system of the ring.
This strongly increases electron density at the 2-, 4- and 6-positions, so multiple substitution occurs very easily.
(b) Reaction of Phenylamine with Nitrous Acid (Diazotisation)
Phenylamine reacts with nitrous acid at low temperature to form a diazonium salt.
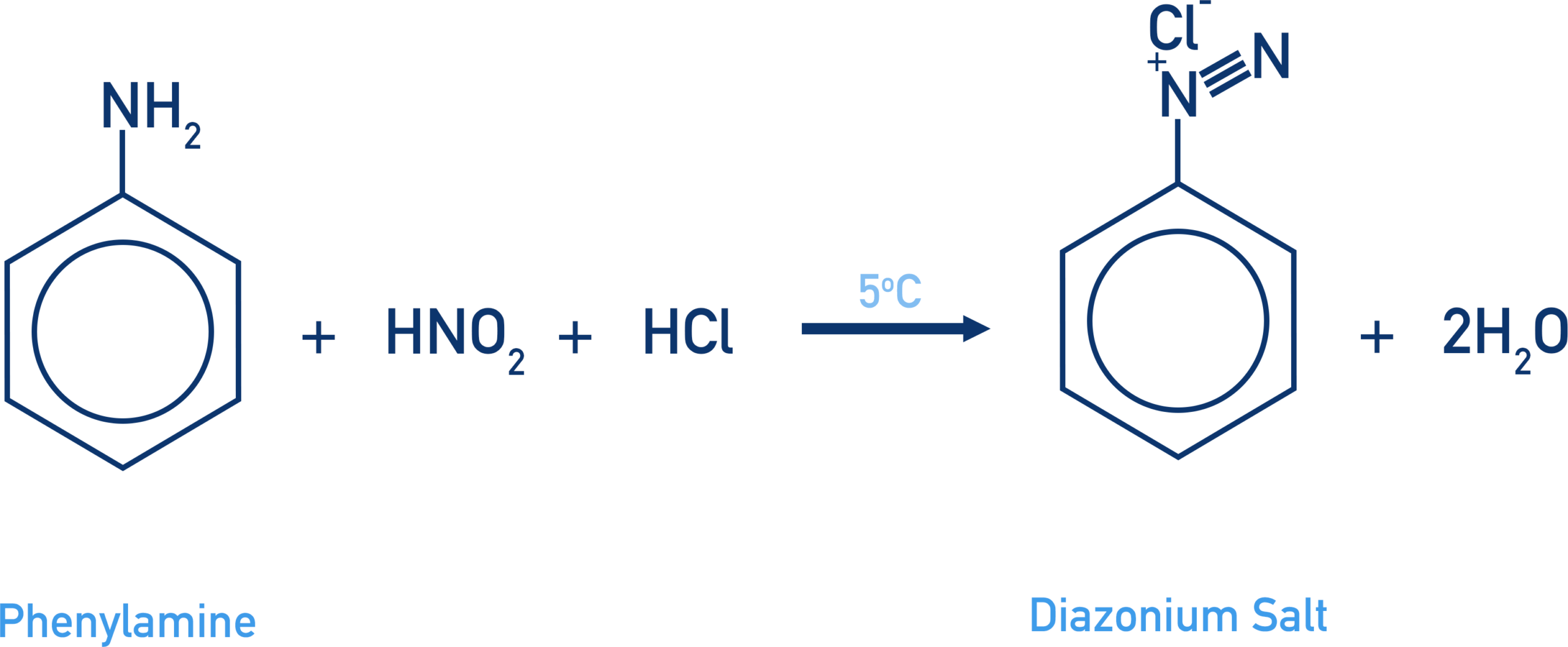
Nitrous acid is generated in situ using sodium nitrite and a dilute acid (usually dilute HCl).
Reagents and conditions:
- \( \mathrm{NaNO_2} \) and dilute \( \mathrm{HCl} \)
- temperature below 10°C
\( \mathrm{C_6H_5NH_2 + HNO_2 + HCl \rightarrow C_6H_5N_2^+Cl^- + 2H_2O} \)
The product is a benzenediazonium chloride, which is only stable at low temperatures.
Further Reaction: Formation of Phenol
On warming the diazonium salt with water, it decomposes to form phenol.
\( \mathrm{C_6H_5N_2^+Cl^- + H_2O \rightarrow C_6H_5OH + N_2 + HCl} \)
Nitrogen gas is released, which provides a strong driving force for the reaction.
Example
Describe what is observed when phenylamine reacts with bromine water.
▶️ Answer / Explanation
Bromine water is rapidly decolourised and a white precipitate of 2,4,6-tribromophenylamine is formed.
Example
Describe how phenylamine can be converted into phenol, stating reagents and conditions.
▶️ Answer / Explanation
Phenylamine is first reacted with sodium nitrite and dilute hydrochloric acid below 10°C to form a diazonium salt.
The diazonium salt is then warmed with water, producing phenol, nitrogen gas and hydrochloric acid.
Relative Basicities of Aqueous Ammonia, Ethylamine and Phenylamine
Ammonia, ethylamine and phenylamine are all bases in aqueous solution. You must be able to describe and explain their relative basicities in terms of the availability of the lone pair on the nitrogen atom.
Order of Basicity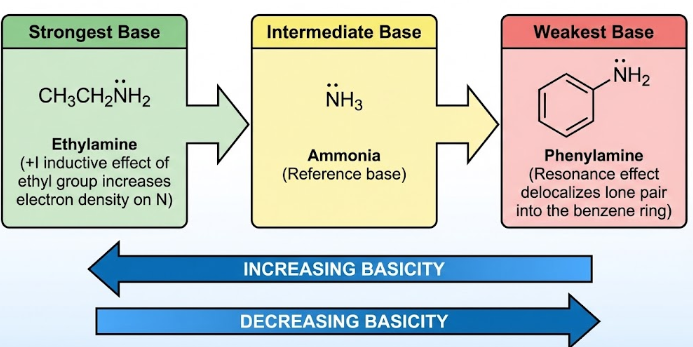
The relative basicities in aqueous solution are:
ethylamine > ammonia > phenylamine
General Principle
A base is stronger when the lone pair on nitrogen is more readily available to accept a proton.
The more available the lone pair, the stronger the base.
Ethylamine
Ethylamine contains an alkyl group attached to the nitrogen atom.
The ethyl group has a +I (electron-donating) effect, which pushes electron density towards the nitrogen atom.
This makes the lone pair more available to accept a proton.
As a result, ethylamine is the most basic of the three.
Ammonia
Ammonia has a lone pair on nitrogen but no alkyl groups.
There is no electron-donating inductive effect to increase electron density on the nitrogen atom.
Therefore, ammonia is less basic than ethylamine, but more basic than phenylamine.
Phenylamine
In phenylamine, the nitrogen atom is attached directly to a benzene ring.
The lone pair on nitrogen is partially delocalised into the aromatic π system.
This reduces the availability of the lone pair to accept a proton.
As a result, phenylamine is the least basic of the three.
Overall Explanation
- Ethylamine is most basic due to electron donation from the alkyl group.
- Ammonia has no electron-donating or withdrawing groups, giving intermediate basicity.
- Phenylamine is least basic because the lone pair on nitrogen is delocalised into the benzene ring.
Example
Arrange ammonia, ethylamine and phenylamine in order of increasing basicity.
▶️ Answer / Explanation
Phenylamine < ammonia < ethylamine.
Example
Explain why phenylamine is less basic than ethylamine in aqueous solution.
▶️ Answer / Explanation
In phenylamine, the lone pair on nitrogen is delocalised into the benzene ring, making it less available to accept a proton.
In ethylamine, the alkyl group donates electron density to the nitrogen atom, increasing the availability of the lone pair.
Azo Compounds
Azo compounds are an important class of aromatic compounds formed by azo coupling reactions. You must be able to recall how azo compounds are formed, identify the azo group, and state their uses.
(a) Coupling of Benzenediazonium Chloride with Phenol
Benzenediazonium chloride reacts with phenol in alkaline conditions to form an azo compound.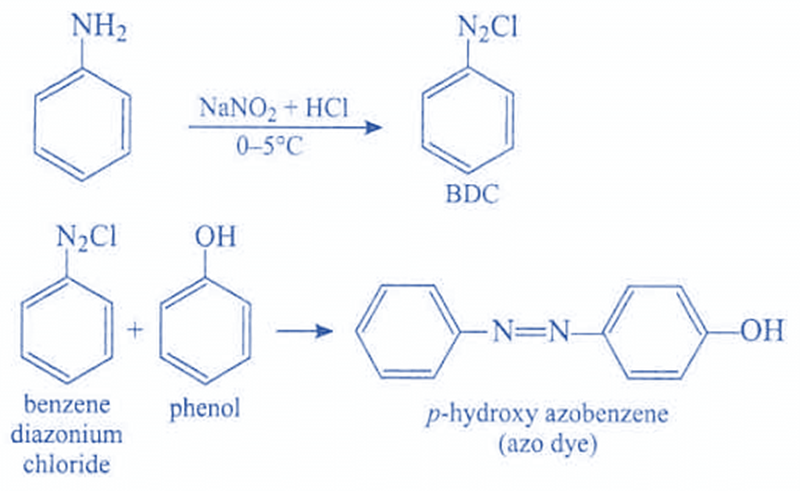
Reagents and conditions:
- benzenediazonium chloride (cold solution)
- phenol dissolved in NaOH(aq)
- temperature kept below 10°C
In alkaline solution, phenol forms the phenoxide ion, which is more reactive towards electrophilic substitution.
The coupling reaction occurs mainly at the 4-position of phenol, producing a brightly coloured azo compound.
(b) The Azo Group
The azo group is:
–N=N–
This group links two aromatic rings together and is responsible for many of the properties of azo compounds.
(c) Uses of Azo Compounds
Azo compounds are often strongly coloured because the –N=N– group is part of an extended delocalised π system.
As a result:
- azo compounds are widely used as dyes
- they are used in textiles, inks and food colourings
(d) Formation of Other Azo Dyes
Other azo dyes can be formed by a similar coupling reaction.
For example:
- using different diazonium salts
- using different activated aromatic compounds (e.g. substituted phenols or amines)
Changing the aromatic compounds alters the colour and properties of the dye produced.
Azo compounds are formed by coupling reactions between diazonium salts and activated aromatic rings and contain the azo group, –N=N–.
Example
State the conditions needed for the coupling of benzenediazonium chloride with phenol.
▶️ Answer / Explanation
The reaction is carried out in aqueous sodium hydroxide solution at a temperature below 10°C.
Example
Explain why azo compounds are often brightly coloured and used as dyes.
▶️ Answer / Explanation
Azo compounds contain the –N=N– group linking aromatic rings.
This creates an extended delocalised π system that absorbs visible light.
As a result, azo compounds are strongly coloured and suitable for use as dyes.
