CIE AS/A Level Chemistry 37.3 Carbon-13 NMR spectroscopy Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 37.3 Carbon-13 NMR spectroscopy Study Notes – New Syllabus
CIE AS/A Level Chemistry 37.3 Carbon-13 NMR spectroscopy Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
analyse and interpret a carbon-13 NMR spectrum of a simple molecule to deduce:
(a) the different environments of the carbon atoms present
(b) the possible structures for the moleculepredict or explain the number of peaks in a carbon-13 NMR spectrum for a given molecule
Interpreting a Carbon-13 NMR Spectrum
Carbon-13 NMR spectroscopy is used to analyse and interpret the structure of organic molecules. You must be able to use a spectrum to deduce:
- the number of different carbon environments present
- the possible structures for the molecule
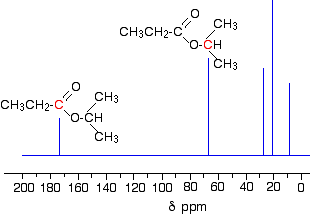
Carbon Environments
A carbon environment refers to a carbon atom that experiences a unique electronic environment.
In a carbon-13 NMR spectrum:
- each peak represents a different carbon environment
- the number of peaks equals the number of unique carbon environments
Carbon atoms in identical environments (e.g. due to symmetry) produce one peak.
Chemical Shift and Carbon Type
The chemical shift (δ value, in ppm) indicates the type of carbon present.
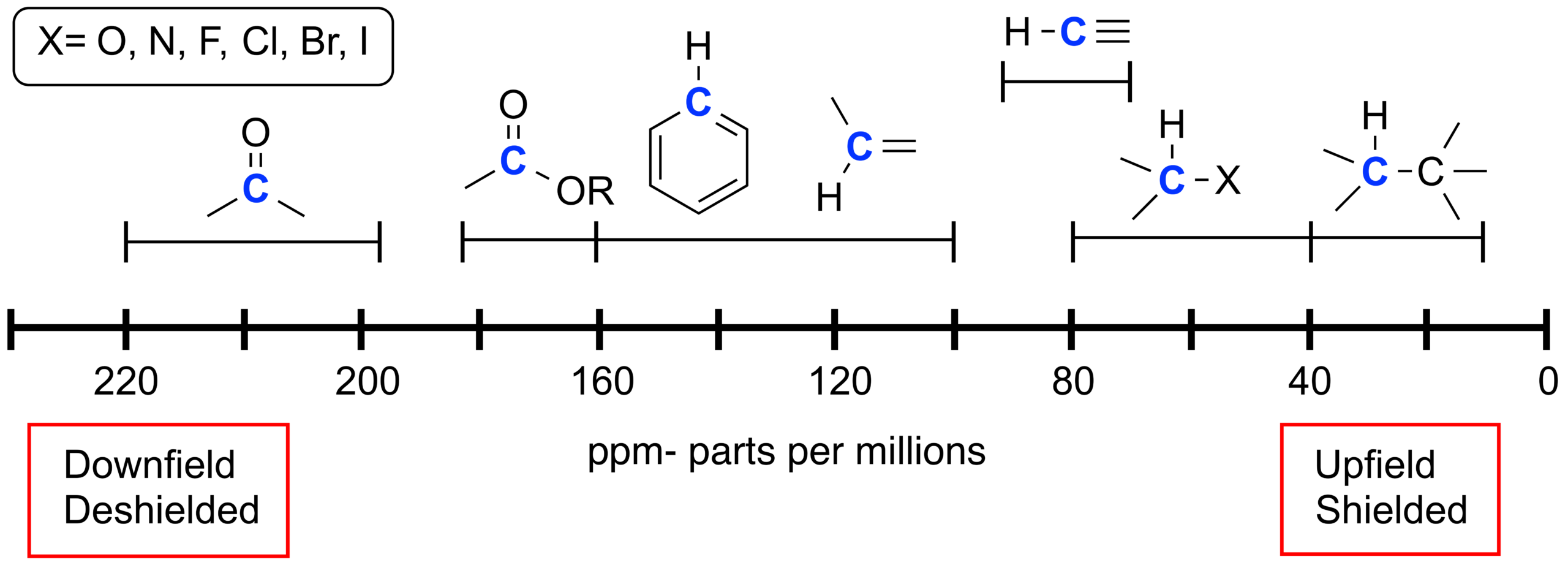
Deducing Possible Structures
To deduce possible structures from a carbon-13 NMR spectrum:
- count the number of peaks to determine carbon environments
- use the chemical shift values to identify functional groups
- consider symmetry to reduce the number of environments
- match findings to the molecular formula
More than one structure may fit the same spectrum, giving several possible isomers.
Example
A compound has the molecular formula \( \mathrm{C_3H_6O} \). Its carbon-13 NMR spectrum shows two peaks, one at 210 ppm and one at 30 ppm.
▶️ Answer / Explanation
A peak at 210 ppm indicates a carbonyl carbon.
The peak at 30 ppm corresponds to an alkane carbon.
Two peaks mean two different carbon environments.
A structure consistent with this data is propanone, where the two methyl carbons are equivalent due to symmetry.
Example
A molecule has the molecular formula \( \mathrm{C_4H_{10}O} \). Its carbon-13 NMR spectrum shows three peaks at approximately 18, 32 and 65 ppm.
Deduce the carbon environments and suggest a possible structure.
▶️ Answer / Explanation
The peak at 65 ppm suggests a carbon bonded to oxygen.
The other peaks are alkane carbons in different environments.
Three peaks indicate three different carbon environments.
A suitable structure is butan-1-ol, which contains three unique carbon environments due to lack of symmetry.
Predicting the Number of Peaks in a Carbon-13 NMR Spectrum
In a carbon-13 NMR spectrum, the number of peaks corresponds to the number of different carbon environments in the molecule. You must be able to predict or explain the number of peaks for a given structure.
Key Principle
Each unique carbon environment gives rise to one peak in a carbon-13 NMR spectrum.
Carbon atoms that are in identical environments (for example due to symmetry) produce a single shared peak.
What Makes Carbon Environments the Same?
- carbon atoms bonded to the same atoms
- carbon atoms in symmetrical positions within the molecule
- carbon atoms experiencing the same electronic environment
If rotating or reflecting the molecule makes two carbons interchangeable, they are in the same environment.
Effect of Symmetry
Symmetry reduces the number of peaks because equivalent carbons are counted once.
- High symmetry → fewer peaks
- Low or no symmetry → more peaks
How to Predict the Number of Peaks
To predict the number of carbon-13 NMR peaks:
- draw the full structure of the molecule
- circle each carbon atom
- identify carbons in identical positions
- count the number of different environments
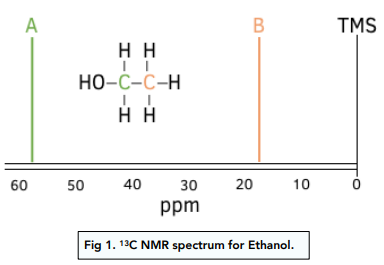
Example
Predict the number of carbon-13 NMR peaks for ethanol, \( \mathrm{CH_3CH_2OH} \).
▶️ Answer / Explanation
Ethanol contains two carbon atoms.
The \( \mathrm{CH_3} \) carbon and the \( \mathrm{CH_2} \) carbon are in different environments.
Therefore, ethanol produces two carbon-13 NMR peaks.
Example
Predict the number of carbon-13 NMR peaks for 2-methylpropane, \( \mathrm{(CH_3)_3CH} \), and explain your reasoning.
▶️ Answer / Explanation
2-methylpropane contains four carbon atoms in total.
The three \( \mathrm{CH_3} \) groups are arranged symmetrically and are in identical environments.
These three carbons give one peak.
The central \( \mathrm{CH} \) carbon is in a different environment and gives a second peak.
Therefore, the molecule produces two carbon-13 NMR peaks.
