CIE AS/A Level Chemistry 37.4 Proton 1H NMR spectroscopy Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 37.4 Proton 1H NMR spectroscopy Study Notes – New Syllabus
CIE AS/A Level Chemistry 37.4 Proton 1H NMR spectroscopy Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
analyse and interpret a proton (¹H) NMR spectrum of a simple molecule to deduce:
(a) the different environments of proton present using chemical shift values
(b) the relative numbers of each type of proton present from relative peak areas
(c) the number of equivalent protons on the carbon atom adjacent to the one to which the given
proton is attached from the splitting pattern, using the n + 1 rule (limited to singlet, doublet,
triplet, quartet and multiplet)
(d) the possible structures for the moleculepredict the chemical shifts and splitting patterns of the protons in a given molecule
describe the use of tetramethylsilane, TMS, as the standard for chemical shift measurements
state the need for deuterated solvents, e.g. CDCl₃, when obtaining a proton NMR spectrum
describe the identification of O–H and N–H protons by proton exchange using D₂O
Interpreting a Proton (¹H) NMR Spectrum
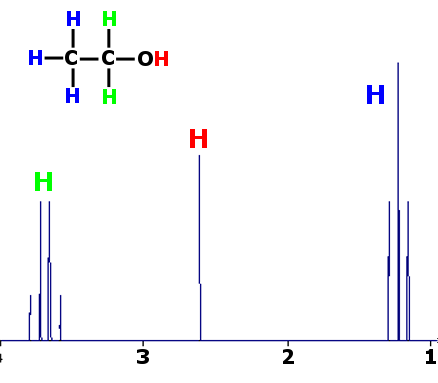
Proton (¹H) NMR spectroscopy is used to analyse and interpret the structure of simple organic molecules. From a ¹H NMR spectrum, you must be able to deduce:
- the different proton environments using chemical shift values
- the relative numbers of each type of proton from peak areas
- the number of equivalent neighbouring protons using splitting patterns and the n + 1 rule
- the possible structures for the molecule
(a) Proton Environments and Chemical Shift
A proton environment is a set of hydrogen atoms experiencing the same electronic surroundings.
In a proton NMR spectrum:
- each peak corresponds to a different proton environment
- the chemical shift (δ, in ppm) indicates the type of proton present
Typical chemical shift ranges:
- 0–1.5 ppm → alkane protons
- 1.5–3.0 ppm → protons next to electronegative atoms

- 3.0–5.0 ppm → protons bonded to C–O or C–N
- 5.0–7.0 ppm → alkene protons
- 7.0–8.0 ppm → aromatic protons
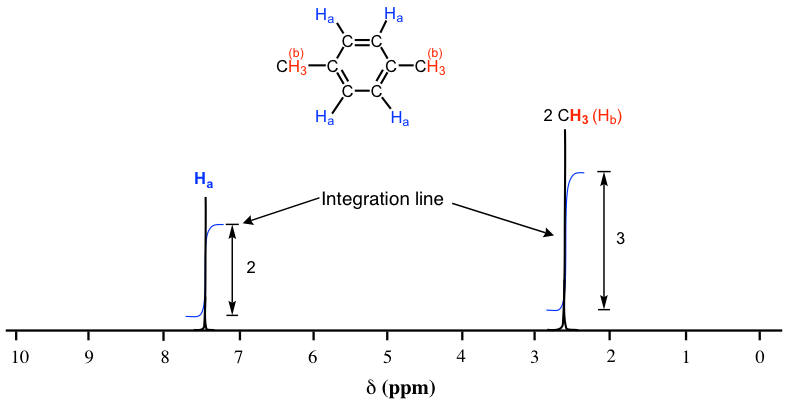
(b) Relative Peak Areas
The area under each peak is proportional to the number of protons in that environment.
Peak areas are shown as ratios (e.g. 3 : 2 : 1) and allow the number of each type of proton to be deduced.
(c) Splitting Patterns and the n + 1 Rule
Splitting occurs due to interaction between non-equivalent protons on adjacent carbon atoms.
The number of peaks in a signal is given by the n + 1 rule, where n is the number of equivalent neighbouring protons.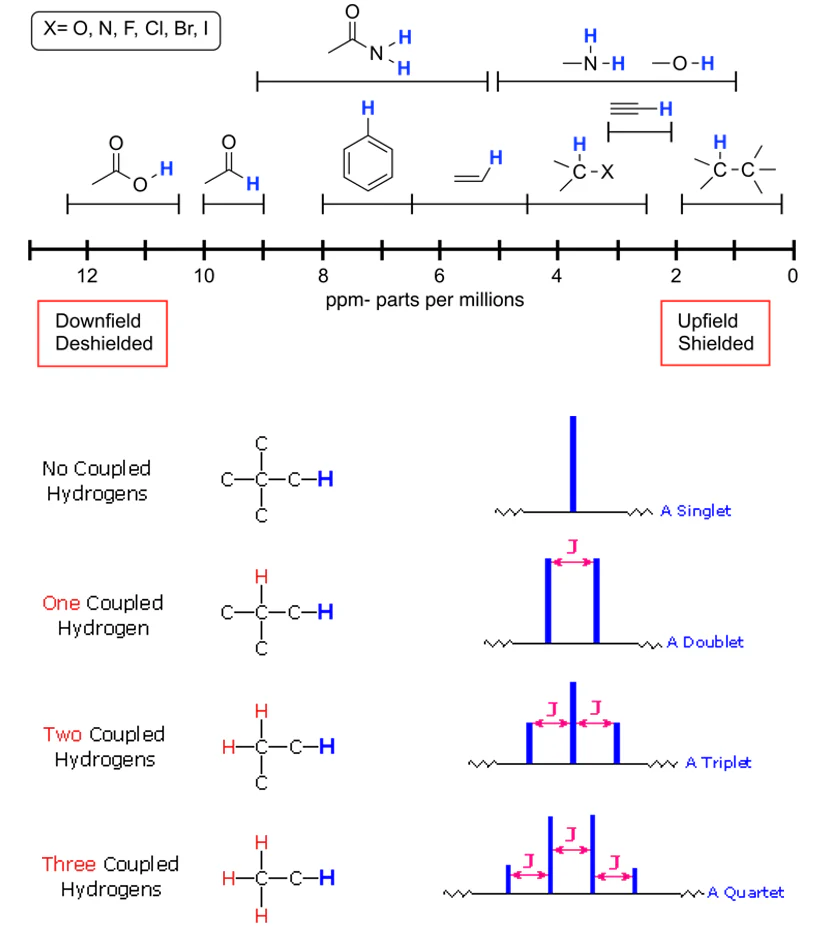
- 0 neighbours → singlet
- 1 neighbour → doublet
- 2 neighbours → triplet
- 3 neighbours → quartet
- 4 or more neighbours → multiplet
Only splitting up to quartets and multiplets is required at this level.
(d) Deducing Possible Structures
To deduce possible structures from a proton NMR spectrum:
- count the number of peaks to find proton environments
- use chemical shifts to identify functional groups
- use peak area ratios to determine proton numbers
- use splitting patterns to identify neighbouring protons
- combine all information with the molecular formula
Example
A compound has a proton NMR spectrum with:
- a triplet at 1.2 ppm with area 3
- a quartet at 3.6 ppm with area 2
Deduce the environments and suggest a structure.
▶️ Answer / Explanation
The triplet indicates 2 neighbouring protons and corresponds to a \( \mathrm{CH_3} \) group.
The quartet indicates 3 neighbouring protons and corresponds to a \( \mathrm{CH_2} \) group.
The shift at 3.6 ppm suggests a \( \mathrm{CH_2} \) attached to oxygen.
The structure is ethanol, \( \mathrm{CH_3CH_2OH} \).
Example
A compound with molecular formula \( \mathrm{C_4H_{10}O} \) has a proton NMR spectrum showing:
- a singlet at 1.2 ppm (area 9)
- a singlet at 2.0 ppm (area 1)
Deduce the structure.
▶️ Answer / Explanation
A singlet with area 9 indicates three equivalent \( \mathrm{CH_3} \) groups attached to the same carbon.
The singlet at 2.0 ppm with area 1 corresponds to an \( \mathrm{OH} \) proton.
The lack of splitting shows no neighbouring non-equivalent protons.
The structure is 2-methylpropan-2-ol.
Predicting Chemical Shifts and Splitting Patterns in ¹H NMR
You must be able to predict the chemical shifts and splitting patterns of protons in a given molecule using knowledge of the local chemical environment and neighbouring protons.
Predicting Chemical Shifts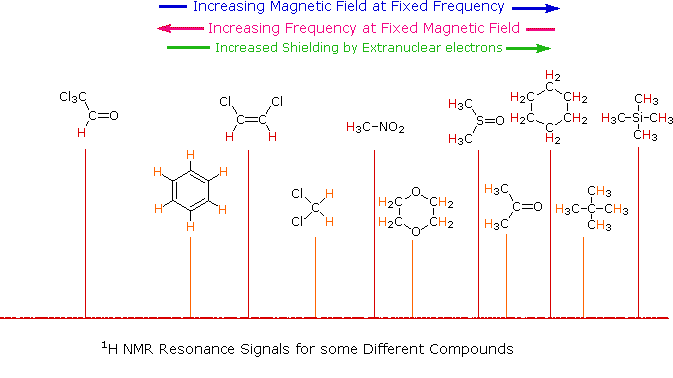
The chemical shift (δ, in ppm) depends on how shielded or deshielded a proton is by nearby electrons.
Protons near electronegative atoms or π systems are more deshielded and appear at higher ppm values.
Typical chemical shift ranges:
- 0.8–1.5 ppm → alkyl \( \mathrm{CH_3} \) and \( \mathrm{CH_2} \)
- 1.5–3.0 ppm → protons next to C=O or aromatic ring
- 3.0–4.5 ppm → protons bonded to carbon attached to O or N
- 5.0–6.5 ppm → alkene protons
- 7.0–8.0 ppm → aromatic protons
- 10–13 ppm → carboxylic acid proton
Predicting Splitting Patterns (n + 1 Rule)
Splitting occurs due to interaction with non-equivalent neighbouring protons on adjacent carbon atoms.
The number of peaks in a signal is given by: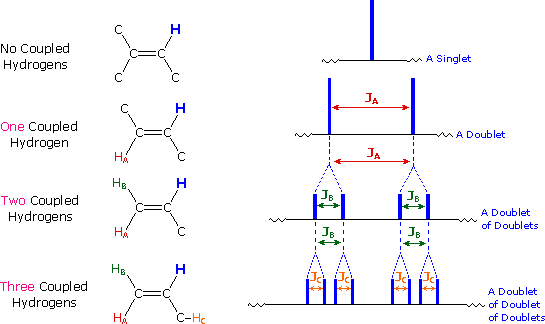
\( \mathrm{number\ of\ peaks = n + 1} \)
where n is the number of equivalent neighbouring protons.
- 0 neighbours → singlet
- 1 neighbour → doublet
- 2 neighbours → triplet
- 3 neighbours → quartet
- 4+ neighbours → multiplet
Exchangeable protons such as –OH usually appear as singlets and do not follow the n + 1 rule.
Step-by-Step Prediction Method
- identify each unique proton environment
- estimate the chemical shift from nearby functional groups
- count neighbouring protons on adjacent carbons
- apply the n + 1 rule to predict splitting
Example
Predict the chemical shifts and splitting patterns of the protons in propan-1-ol, \( \mathrm{CH_3CH_2CH_2OH} \).
▶️ Answer / Explanation
The \( \mathrm{CH_3} \) group is next to a \( \mathrm{CH_2} \) group (2 neighbours), so it appears as a triplet at about 1.0 ppm.
The middle \( \mathrm{CH_2} \) group has five neighbouring protons in total, so it appears as a multiplet at about 1.4–1.8 ppm.
The \( \mathrm{CH_2} \) group next to oxygen appears at about 3.5–4.0 ppm as a triplet.
The \( \mathrm{OH} \) proton appears as a broad singlet.
Example
Predict the chemical shifts and splitting patterns of the protons in ethyl ethanoate, \( \mathrm{CH_3COOCH_2CH_3} \).
▶️ Answer / Explanation
The \( \mathrm{CH_3CO–} \) group appears as a singlet at about 2.0 ppm because there are no neighbouring protons.
The \( \mathrm{–OCH_2–} \) protons appear as a quartet at about 4.0 ppm due to three neighbouring \( \mathrm{CH_3} \) protons.
The terminal \( \mathrm{CH_3} \) group appears as a triplet at about 1.2 ppm due to two neighbouring \( \mathrm{CH_2} \) protons.
Tetramethylsilane (TMS) as the Chemical Shift Standard
In NMR spectroscopy, chemical shifts are measured relative to a standard reference compound. The standard used in both ¹H NMR and ¹³C NMR spectroscopy is tetramethylsilane (TMS).
What is TMS?
Tetramethylsilane has the formula \( \mathrm{Si(CH_3)_4} \).
It contains twelve equivalent hydrogen atoms and four equivalent carbon atoms, so it produces a single sharp peak in both ¹H and ¹³C NMR spectra.
Chemical Shift Reference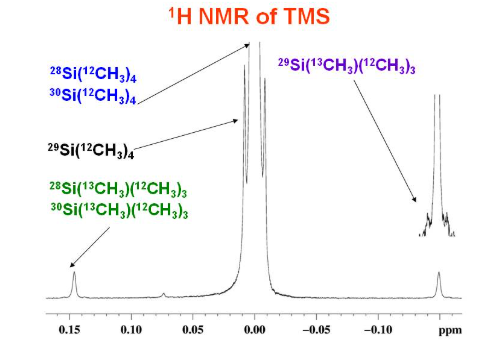
The NMR peak of TMS is assigned a chemical shift of:
\( \mathrm{\delta = 0.0\ ppm} \)
All other chemical shifts are measured relative to this peak.
Why TMS is an Ideal Standard
- All protons are in identical environments, giving one peak
- The TMS peak occurs at a very low chemical shift, outside most organic signals
- TMS is chemically inert and does not react with samples
- TMS is volatile, making it easy to remove after analysis
Silicon is less electronegative than carbon, so the electrons around the hydrogen atoms provide strong shielding, resulting in a very low chemical shift.
How TMS is Used in Practice
A small amount of TMS is added to the sample before the NMR spectrum is recorded.
The position of other peaks is then measured relative to the TMS peak and reported as chemical shift values in ppm.
Example
Explain why TMS produces only one peak in a proton NMR spectrum.
▶️ Answer / Explanation
All twelve hydrogen atoms in TMS are in identical chemical environments.
Therefore, they absorb at the same energy and give a single NMR peak.
Example
TMS is assigned a chemical shift of 0 ppm. Explain why its peak appears at a lower ppm value than most organic protons.
▶️ Answer / Explanation
Silicon is less electronegative than carbon.
As a result, the protons in TMS are highly shielded by surrounding electrons.
This causes them to absorb at a lower frequency and appear at a lower chemical shift than most organic protons.
Use of Deuterated Solvents in ¹H NMR Spectroscopy
When obtaining a proton (¹H) NMR spectrum, the sample must be dissolved in a solvent. This solvent is required to be deuterated, for example CDCl₃.
Why a Solvent is Needed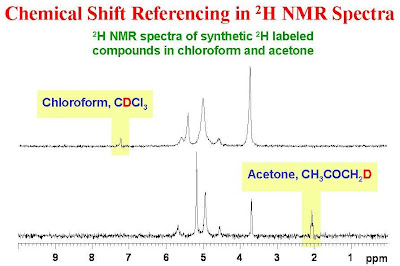
The sample must be in solution so that molecules can move freely, giving a clear and well-resolved NMR spectrum.
Need for Deuterated Solvents
A deuterated solvent is one in which the hydrogen atoms have been replaced by deuterium (²H).
Deuterium does not produce a signal in a proton NMR spectrum.
Key Reasons for Using Deuterated Solvents
- to prevent the solvent from producing a large ¹H NMR peak
- to ensure peaks observed come only from the sample
- to allow accurate measurement of chemical shifts
If a non-deuterated solvent were used, its many hydrogen atoms would produce a very strong signal that would mask the sample’s peaks.
Additional Benefit
Deuterated solvents also provide a deuterium signal that is used by the spectrometer to maintain a stable magnetic field (field locking).
Example
Explain why CDCl₃ is suitable for use as a solvent in proton NMR spectroscopy.
▶️ Answer / Explanation
CDCl₃ contains deuterium instead of hydrogen.
Deuterium does not give a signal in a proton NMR spectrum.
Therefore, the solvent does not interfere with or obscure the sample’s proton peaks.
Example
A student accidentally uses chloroform, CHCl₃, instead of CDCl₃ as the solvent for a proton NMR experiment. Explain the effect this would have on the spectrum.
▶️ Answer / Explanation
CHCl₃ contains hydrogen atoms that absorb in proton NMR.
The solvent would produce a strong peak in the spectrum.
This peak could overlap with or mask peaks from the sample, making interpretation difficult or unreliable.
Identification of O–H and N–H Protons Using D₂O Exchange
In ¹H NMR spectroscopy, protons attached to oxygen or nitrogen can be identified using proton exchange with deuterium oxide (D₂O). This method is known as the D₂O shake test.
Principle of Proton Exchange
Protons in O–H and N–H bonds are exchangeable. When D₂O is added, these protons are replaced by deuterium (²H).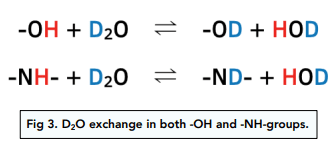
Deuterium does not produce a signal in a proton NMR spectrum.
What Happens in the Spectrum
After shaking the sample with D₂O:
- the O–H or N–H peak disappears
- all other proton peaks remain unchanged
This confirms that the missing peak was due to an O–H or N–H proton.
Why Only O–H and N–H Protons Exchange
Hydrogen atoms bonded to oxygen or nitrogen are relatively labile and can readily exchange with deuterium.
Protons bonded directly to carbon (C–H) do not exchange under these conditions, so their peaks remain unchanged.
Uses of the D₂O Shake Test
- to confirm the presence of an –OH group (alcohols, phenols, carboxylic acids)
- to confirm the presence of an –NH group (amines, amides)
- to distinguish exchangeable protons from alkyl protons
Example
A proton NMR spectrum shows a broad singlet at 2.5 ppm. After adding D₂O, this peak disappears.
Identify the type of proton responsible for this peak.
▶️ Answer / Explanation
The disappearance of the peak after D₂O addition shows it is an exchangeable proton.
Therefore, the peak corresponds to an O–H or N–H proton.
Example
A compound with formula \( \mathrm{C_3H_7NO} \) gives a proton NMR spectrum containing:
- a triplet (3H)
- a quartet (2H)
- a broad singlet (1H)
The broad singlet disappears after shaking with D₂O. Deduce the functional group present.
▶️ Answer / Explanation
The disappearing singlet corresponds to an N–H or O–H proton.
The remaining triplet and quartet indicate a \( \mathrm{CH_3CH_2–} \) fragment.
The presence of nitrogen in the formula and an exchangeable proton indicates an amine.