CIE AS/A Level Chemistry 4.2 Bonding and structure Study Notes- 2025-2027 Syllabus
CIE AS/A Level Chemistry 4.2 Bonding and structure Study Notes – New Syllabus
CIE AS/A Level Chemistry 4.2 Bonding and structure Study Notes at IITian Academy focus on specific topic and type of questions asked in actual exam. Study Notes focus on AS/A Level Chemistry latest syllabus with Candidates should be able to:
- describe, in simple terms, the lattice structure of a crystalline solid which is:
(a) giant ionic, including sodium chloride and magnesium oxide
(b) simple molecular, including iodine, buckminsterfullerene C60 and ice
(c) giant molecular, including silicon(IV) oxide, graphite and diamond
(d) giant metallic, including copper - describe, interpret and predict the effect of different types of structure and bonding on the physical properties of substances, including melting point, boiling point, electrical conductivity and solubility
- deduce the type of structure and bonding present in a substance from given information
Lattice Structures of Crystalline Solids
Crystalline solids have particles arranged in a highly ordered repeating structure called a lattice. The type of lattice depends on the nature of the bonding between particles.
(a) Giant Ionic Lattices
Giant ionic lattices consist of oppositely charged ions arranged in a regular 3D structure. Each cation is surrounded by anions and vice versa, maximising electrostatic attraction.
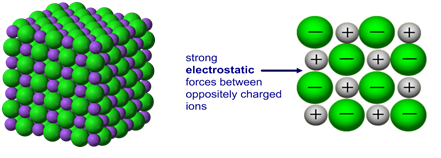
- Ions are held by strong ionic bonds.
- High melting and boiling points.
- Conduct electricity only when molten or in aqueous solution (ions are mobile).
- Brittle — shifting layers cause repulsion.
Sodium chloride (\( \mathrm{NaCl} \))
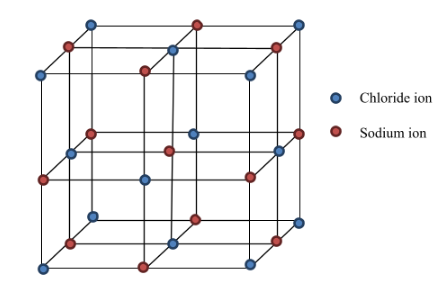
- Each \( \mathrm{Na^+} \) is surrounded by six \( \mathrm{Cl^-} \) ions.
- Cubic lattice structure.
Magnesium oxide (\( \mathrm{MgO} \))
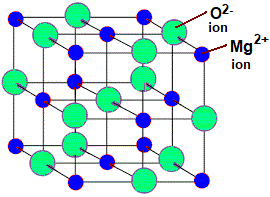
- Stronger ionic bonding than NaCl due to \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \).
- Very high melting point.
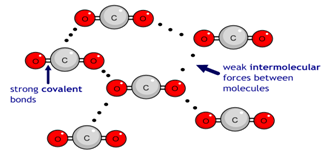
(b) Simple Molecular Lattices
Simple molecular crystals contain individual molecules held together by weak intermolecular forces (van der Waals’ forces). The molecules themselves contain covalent bonds.
- Low melting and boiling points.
- Do not conduct electricity.
- Soft and brittle.
Iodine (\( \mathrm{I_2} \))
- \( \mathrm{I_2} \) molecules held by London forces.
- Shiny grey–black solid.
Buckminsterfullerene (\( \mathrm{C_{60}} \))
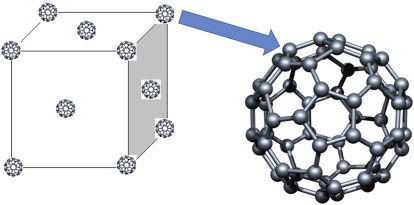
- Spherical molecules of 60 carbon atoms.
- Molecules pack together via weak van der Waals forces.
Ice (\( \mathrm{H_2O} \))
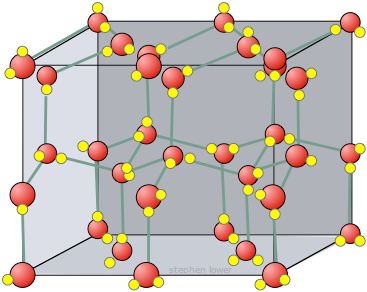
- Hydrogen bonds between water molecules.
- Open lattice structure → ice less dense than liquid water.
(c) Giant Molecular Lattices (Covalent Networks)
Giant molecular lattices consist of atoms joined by strong covalent bonds throughout the whole structure.
- Very high melting points.
- Hard (except graphite).
- Do not conduct electricity (except graphite).
- Insoluble.
Silicon(IV) oxide (\( \mathrm{SiO_2} \))
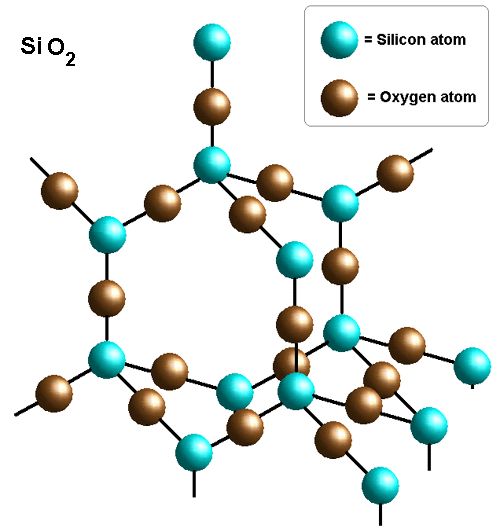
- Each Si bonded to 4 O atoms.
- Network similar to diamond.
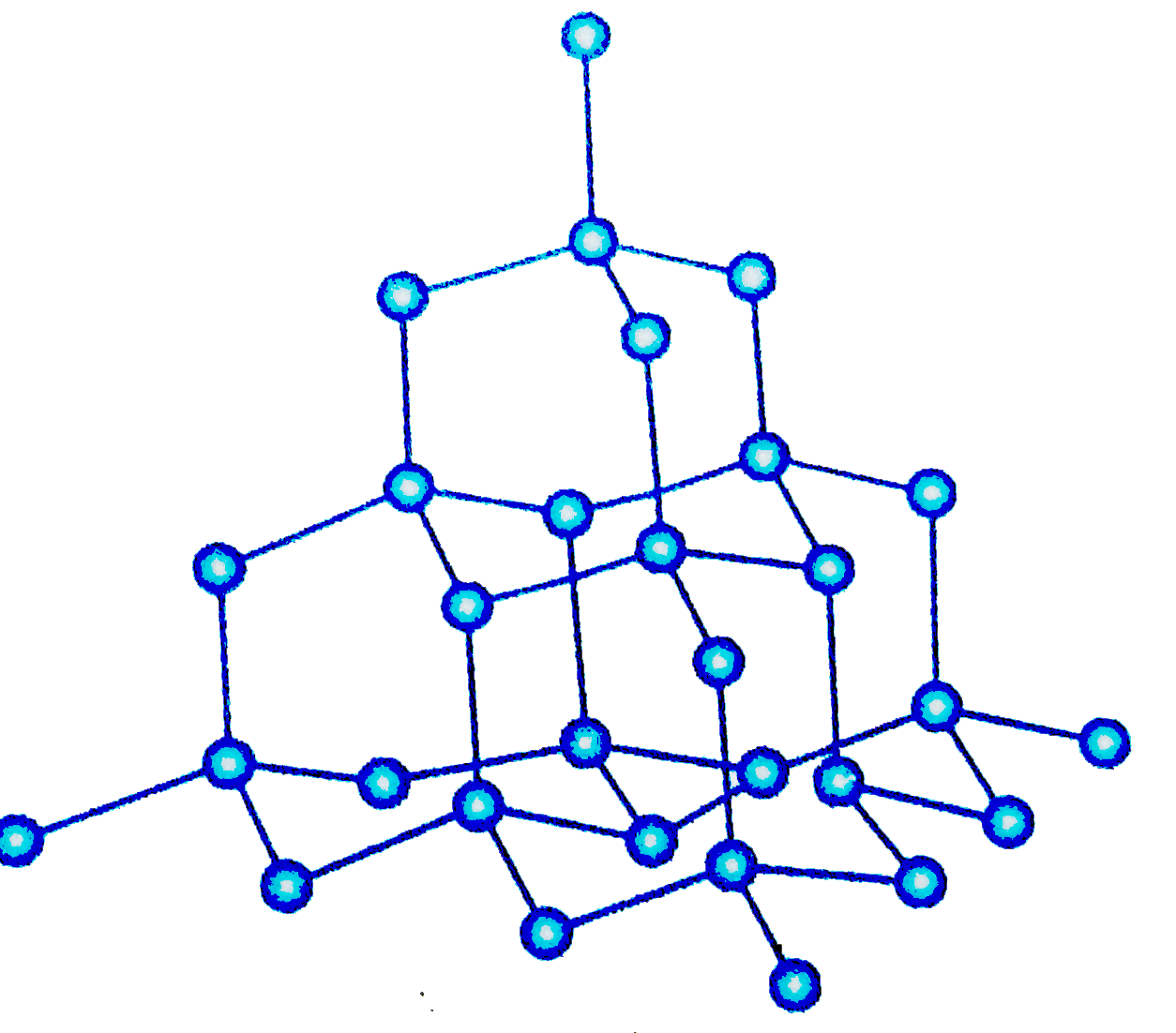
Diamond
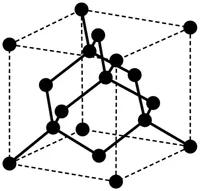
- Each carbon forms 4 covalent bonds (tetrahedral).
- Extremely hard; non-conductive.
Graphite
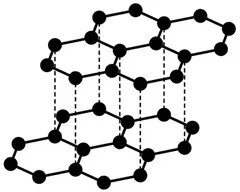
- Each carbon bonded to 3 others → layered structure.
- Delocalised electrons between layers → conducts electricity.
- Layers slide due to weak forces → soft/lubricant.
(d) Giant Metallic Lattices
Metallic lattices consist of positive metal ions in a sea of delocalised electrons.
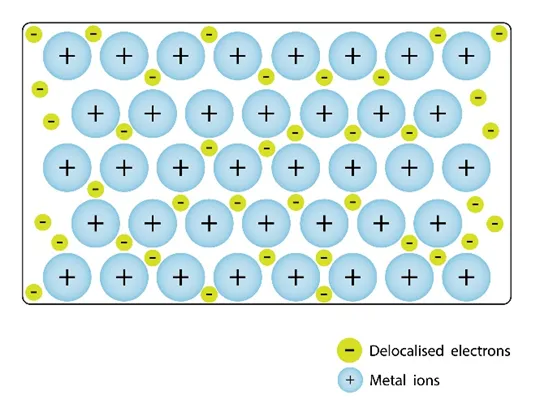
- Strong metallic bonding.
- High melting and boiling points.
- Good electrical and thermal conductivity.
- Malleable and ductile (layers slide).
Copper (\( \mathrm{Cu} \))
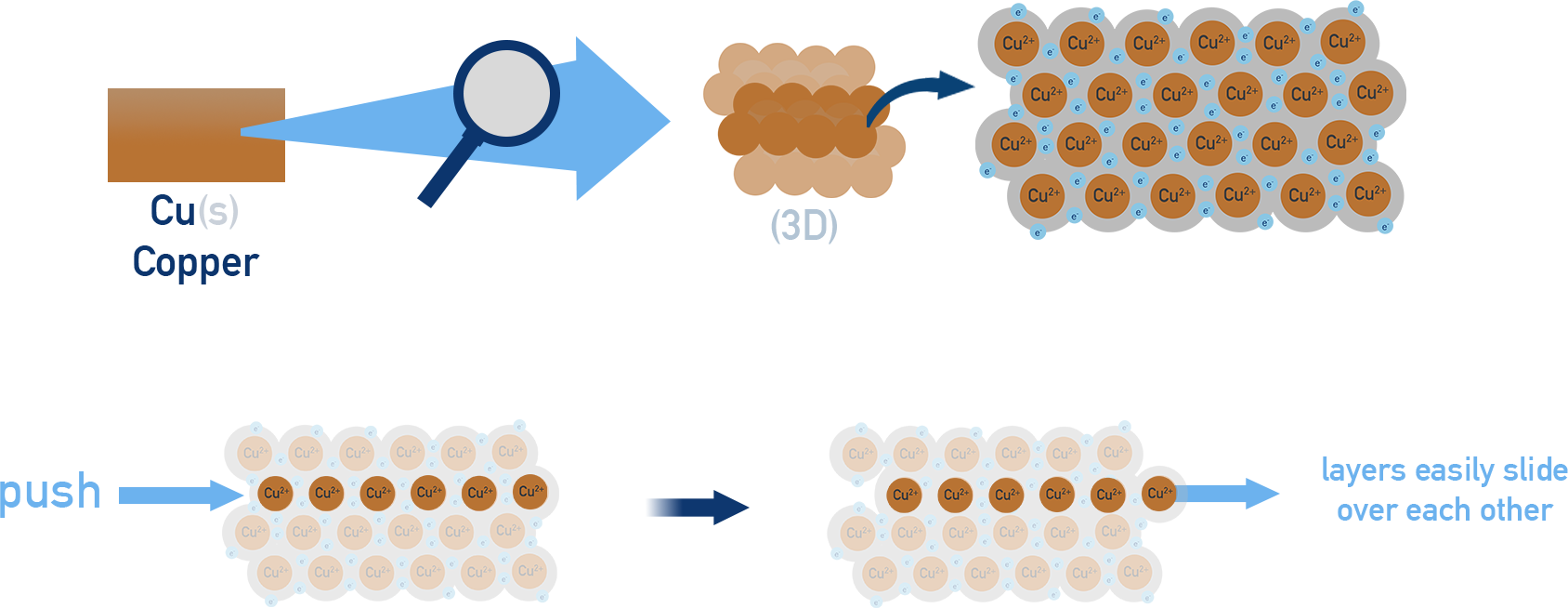
- Close-packed lattice of \( \mathrm{Cu^+} \) ions.
- Delocalised electrons allow efficient conductivity.
- Ductile and malleable.
Example
State the type of lattice found in sodium chloride.
▶️ Answer / Explanation
Sodium chloride has a giant ionic lattice.
Example
Explain why graphite conducts electricity but diamond does not.
▶️ Answer / Explanation
In graphite, each carbon forms 3 covalent bonds, leaving 1 delocalised electron that moves freely → electrical conduction.
In diamond, all 4 electrons form covalent bonds → no delocalised electrons → no conduction.
Example
Explain why magnesium oxide has a much higher melting point than iodine.
▶️ Answer / Explanation
\( \mathrm{MgO} \) is a giant ionic lattice with strong electrostatic forces between \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \).
Iodine is a simple molecular lattice with only weak London forces between \( \mathrm{I_2} \) molecules.
Therefore, much more energy is needed to break the ionic lattice in \( \mathrm{MgO} \).
Effect of Structure and Bonding on Physical Properties
The physical properties of a substance melting point, boiling point, electrical conductivity, and solubility are determined by its type of bonding and the structure of its lattice.
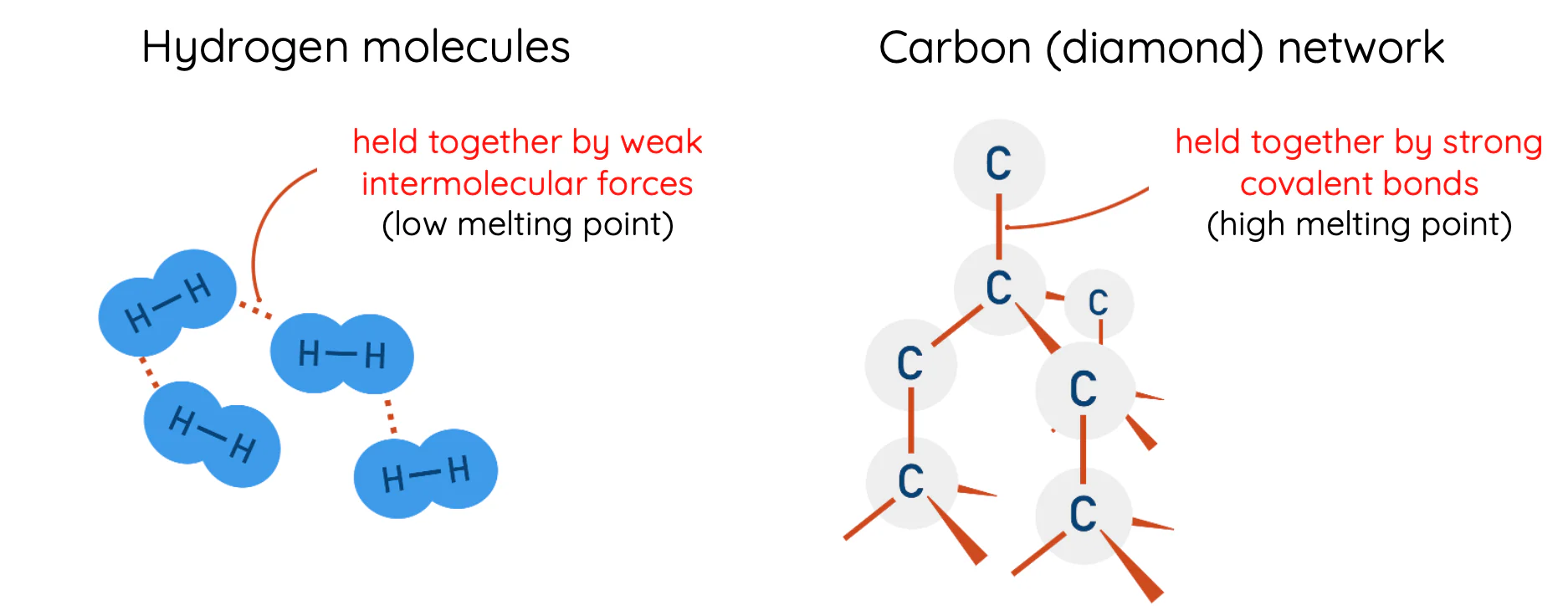
Melting and Boiling Points
- Giant ionic lattices (e.g., \( \mathrm{NaCl} \), \( \mathrm{MgO} \)) High melting and boiling points due to strong electrostatic forces between oppositely charged ions.
- Giant covalent lattices (e.g., diamond, \( \mathrm{SiO_2} \)) Very high melting points — many strong covalent bonds throughout the structure.
- Giant metallic lattices (e.g., Cu) High melting points — strong attraction between metal ions and delocalised electrons.
- Simple molecular lattices (e.g., \( \mathrm{I_2} \), \( \mathrm{C_{60}} \), ice) Low melting and boiling points — weak intermolecular forces (London forces or hydrogen bonding).
Electrical Conductivity
- Ionic compounds Conduct when molten or dissolved in water — ions become mobile.
- Metals Good conductors — delocalised electrons move freely.
- Graphite Conducts electricity — delocalised electrons between layers.
- Diamond, \( \mathrm{SiO_2} \) Do not conduct — no mobile charged particles.
- Simple molecular substances Do not conduct — no ions or delocalised electrons.
Solubility
- Ionic substances Generally soluble in water — water molecules surround ions (ion–dipole interactions).
- Simple molecular substances Generally soluble in organic solvents (non-polar dissolves non-polar).
- Giant covalent substances Insoluble — covalent bonds too strong to break.
- Metals Insoluble in all solvents — metallic lattice not disrupted by solvent interactions.
Linking Structure to Physical Properties
- Strength of bonding determines melting/boiling point.
- Presence of mobile charged particles determines electrical conductivity.
- Compatibility between solute and solvent determines solubility.
- Intermolecular forces vs. lattice forces explain differences between simple molecular and giant structures.
Example
Why does sodium chloride have a high melting point?
▶️ Answer / Explanation
Sodium chloride has a giant ionic lattice with strong electrostatic forces between \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \) ions. A large amount of energy is required to break these forces.
Example
Explain why graphite conducts electricity but diamond does not.
▶️ Answer / Explanation
Graphite contains delocalised electrons that move between layers → conducts electricity.
Diamond has all electrons fixed in covalent bonds → no mobile electrons → non-conductor.
Example
Predict and explain the melting point and electrical conductivity of magnesium oxide.
▶️ Answer / Explanation
\( \mathrm{MgO} \) has a giant ionic lattice with highly charged ions \( \mathrm{Mg^{2+}} \) and \( \mathrm{O^{2-}} \). This produces very strong electrostatic attraction → extremely high melting point.
Solid \( \mathrm{MgO} \) does not conduct electricity because ions are fixed. When molten, ions are mobile and the substance conducts electricity.
Deducing Structure and Bonding from Given Information
The physical properties of a substance (melting point, boiling point, electrical conductivity, solubility, hardness) allow us to determine the type of bonding and structure present. Each structure has characteristic features that can be identified from data.
How to Deduce Structure from Properties
- Very high melting/boiling point → Either giant ionic, giant covalent, or metallic.
- Low melting/boiling point → Likely simple molecular (weak intermolecular forces).
- Conducts electricity when molten or in aqueous solution → Ionic lattice (mobile ions when molten/aqueous).
- Conducts electricity as a solid → Metallic (delocalised electrons) → Or graphite (delocalised electrons between layers).
- Hard and insoluble in all solvents → Giant covalent (diamond, \( \mathrm{SiO_2} \)).
- Brittle → Ionic (shifting layers cause repulsion).
- Soft, low density, weak forces → Simple molecular (London forces).
- Malleable + ductile → Metallic (layers slide under sea of electrons).
- Soluble in water → Often ionic or polar simple molecules.
- Soluble in organic solvents → Simple molecular non-polar substances.
Summary Table: Deduction Rules
| Observed Property | Likely Structure |
|---|---|
| High melting point, conducts when molten | Ionic lattice |
| High melting point, hard, non-conductor | Giant covalent (diamond, \( \mathrm{SiO_2} \)) |
| Conducts as solid, malleable | Metallic |
| Low melting point, does not conduct | Simple molecular |
| Conducts as solid, slippery layers | Graphite (giant covalent with delocalised electrons) |
Example
A substance has a low melting point and does not conduct electricity. What type of structure does it have?
▶️ Answer / Explanation
Low melting point + no conductivity → simple molecular lattice (weak intermolecular forces).
Example
A solid has a very high melting point, is extremely hard, and does not conduct electricity. Deduce its structure.
▶️ Answer / Explanation
Properties match a giant covalent lattice (diamond or \( \mathrm{SiO_2} \)).
High melting point → strong covalent bonds. Hard + non-conductor → typical of giant covalent structures.
Example
A substance is a solid that conducts electricity, is shiny, malleable, and has a high melting point. Predict and explain its bonding and structure.
▶️ Answer / Explanation
Metallic lattice.
Conducts electricity → delocalised electrons.
Malleable + ductile → layers of positive ions slide over each other.
High melting point → strong attraction between ions and delocalised electrons.
