AP Chemistry 1.6 Photoelectron Spectroscopy - Exam Style questions - FRQs- New Syllabus
Question
(ii) Write the ground-state electron configuration of \( \mathrm{Si} \).
| Substance | \( S^\circ \) \( \mathrm{(J/(mol \cdot K))} \) |
|---|---|
| \( \mathrm{H_2}(g) \) | \( 131 \) |
| \( \mathrm{Si}(s) \) | \( 18 \) |
| \( \mathrm{SiH_4}(g) \) | \( 205 \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 1.5 \) — Atomic Structure and Electron Configuration (Part \( \mathrm{(a)(ii)} \))
• Topic \( 3.1 \) — Intermolecular and Interparticle Forces (Part \( \mathrm{(b)} \))
• Topic \( 9.2 \) — Absolute Entropy and Entropy Change (Parts \( \mathrm{(d)} \), \( \mathrm{(e)} \))
• Topic \( 5.11 \) — Catalysis (Part \( \mathrm{(f)} \))
• Topic \( 1.6 \) — Photoelectron Spectroscopy (Part \( \mathrm{(g)} \))
• Topic \( 1.7 \) — Periodic Trends (Part \( \mathrm{(h)} \))
• Topic \( 3.12 \) — Properties of Photons (Part \( \mathrm{(i)} \))
▶️ Answer/Explanation
(a)(i)
For the correct answer:
\( 14 \) protons and \( 14 \) neutrons
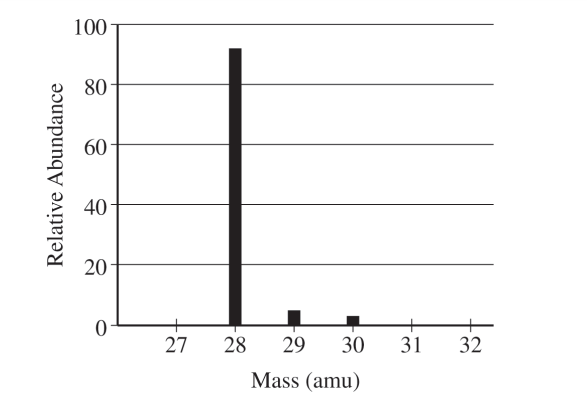
The tallest peak is at mass number \( 28 \), so the most abundant isotope is \( \mathrm{Si}\text{-}28 \). Silicon has atomic number \( 14 \), so neutrons \( = 28 – 14 = 14 \).
(a)(ii)
For the correct answer:
Accept one of the following:
• \( 1s^2\,2s^2\,2p^6\,3s^2\,3p^2 \)
• \( [\mathrm{Ne}]\,3s^2\,3p^2 \)
Silicon has \( 14 \) electrons, so after the neon core, the remaining four electrons go into the \( 3s \) and \( 3p \) sublevels.
(b)
For a correct explanation:
\( \mathrm{SiH_4} \) is composed of molecules, for which the only intermolecular forces are London dispersion forces. \( \mathrm{SiO_2} \) is a network covalent compound with covalent bonds between silicon and oxygen atoms. London dispersion forces are much weaker than covalent bonds, so \( \mathrm{SiH_4} \) boils at a much lower temperature than \( \mathrm{SiO_2} \).
In short, boiling \( \mathrm{SiH_4} \) only requires overcoming weak intermolecular attractions, whereas changing \( \mathrm{SiO_2} \) requires breaking part of a giant covalent network.
(c)
For the correct balanced equation (state symbols not required):
\( \mathrm{SiH_4}(g) \rightarrow \mathrm{Si}(s) + 2\,\mathrm{H_2}(g) \)
A quick atom check shows \( 1 \) silicon atom and \( 4 \) hydrogen atoms on each side.
(d)
For a correct explanation:
The \( \mathrm{H_2}(g) \) molecules are more highly dispersed than the \( \mathrm{Si}(s) \) atoms and, therefore, have a higher absolute molar entropy. Silicon is a solid; therefore, its atoms are in fixed positions, are less dispersed, and have a lower absolute molar entropy.
Gases have many more possible arrangements and much greater freedom of motion than solids, so they generally have higher entropy.
(e)
For the correct calculated value:
\( \Delta S^\circ_{\mathrm{rxn}} = (18 + 2(131)) – 205 = +75\ \mathrm{J/(mol_{rxn} \cdot K)} \)
The positive sign makes sense because the reaction forms more gas particles than it starts with.
(f)
For a correct explanation:
High temperature is required for the reactant particles to have sufficient thermal energy to overcome the activation energy of the reaction.
So even though the reaction is thermodynamically favorable, it is still kinetically slow unless enough energy is supplied.
(g)
For the correct peak height and location:
The peak should be drawn to the right of the other peaks, and it should reach the second line above the horizontal axis.
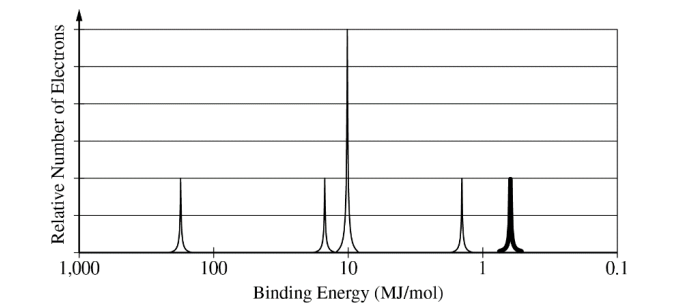
The \( 3p \) electrons have lower binding energy than the \( 3s \) electrons, so their peak is farther to the right. Since there are \( 2 \) electrons in \( 3p \), the peak height corresponds to \( 2 \) electrons.
(h)
For a correct explanation:
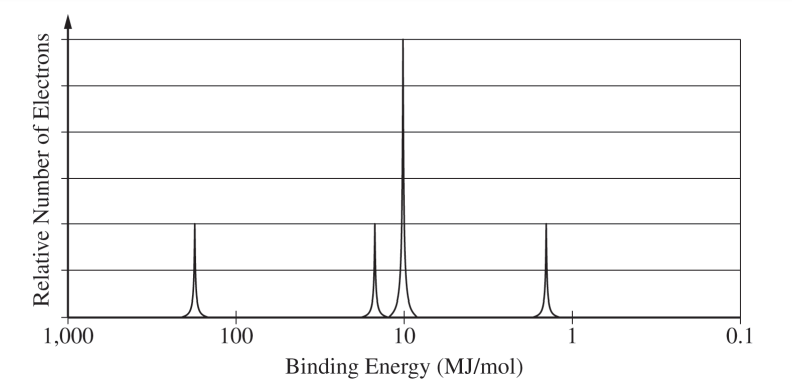
The valence electrons of a \( \mathrm{Ge} \) atom occupy a higher shell \( (n = 4) \) than those of a \( \mathrm{Si} \) atom \( (n = 3) \), so the average distance between the nucleus and the valence electrons is greater in \( \mathrm{Ge} \) than in \( \mathrm{Si} \). This greater separation results in weaker Coulombic attractions between the \( \mathrm{Ge} \) nucleus and its valence electrons, making them less tightly bound and, therefore, easier to remove compared to those in \( \mathrm{Si} \).
More shielding and a larger atomic radius in germanium both help lower the first ionization energy.
(i)
For the correct calculated value:
\( E = h\nu = h\left(\dfrac{c}{\lambda}\right) = (6.626 \times 10^{-34}\ \mathrm{J \cdot s}) \left( \dfrac{2.998 \times 10^8\ \mathrm{m \cdot s^{-1}}}{4.00 \times 10^{-7}\ \mathrm{m}} \right) = 4.97 \times 10^{-19}\ \mathrm{J} \)
The energy is inversely proportional to wavelength, so a shorter wavelength would have given an even higher-energy photon.
