AP Chemistry 2.2 Intramolecular Force and Potential Energy - Exam Style questions - FRQs- New Syllabus
Question
| Reaction Number | Equation | \( \Delta H^\circ_{\mathrm{rxn}} \) \( \mathrm{(kJ/mol}_{\mathrm{rxn}}\mathrm{)} \) |
|---|---|---|
| \( 2 \) | \( \mathrm{Al}(s) + \dfrac{3}{2}\,\mathrm{Cl}_{2}(g) \rightarrow \mathrm{AlCl}_{3}(g) \) | \( -583 \) |
| \( 3 \) | \( \mathrm{Al}(s) \rightarrow \mathrm{Al}(g) \) | \( +326 \) |
| \( 4 \) | \( \mathrm{Cl}_{2}(g) \rightarrow 2\,\mathrm{Cl}(g) \) | \( +243 \) |
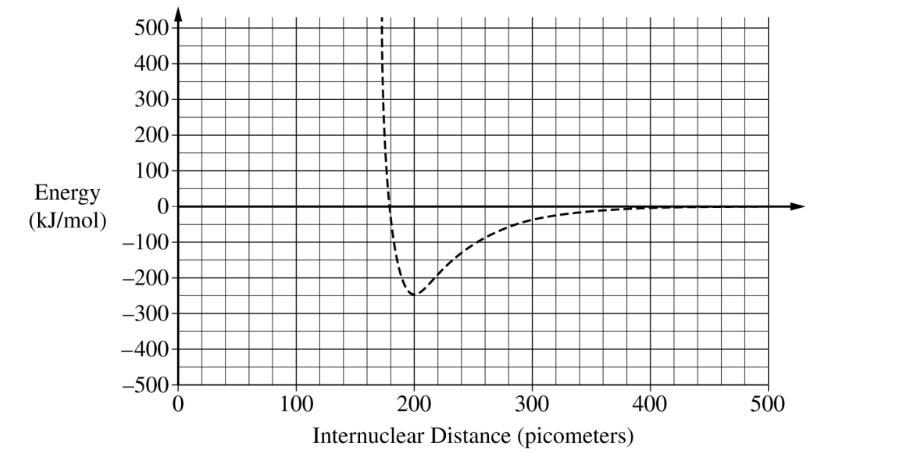
(ii) A student finds that the average \( \mathrm{Al-Cl} \) bond length is \( 220\ \mathrm{pm} \) and the average bond energy is \( 425\ \mathrm{kJ/mol} \). Draw the potential energy curve for the average \( \mathrm{Al-Cl} \) bond on the preceding graph.
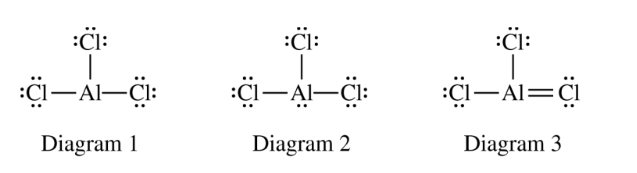
(ii) Which of the three diagrams is the best representation for the bonding in \( \mathrm{AlCl}_{3} \)? Justify your choice based on formal charges.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 6.9 \) — Hess’s Law (Part \( \mathrm{(b)} \))
• Topic \( 2.2 \) — Intramolecular Force and Potential Energy (Parts \( \mathrm{(c)(i)} \), \( \mathrm{(c)(ii)} \))
• Topic \( 2.7 \) — VSEPR and Hybridization (Part \( \mathrm{(d)(i)} \))
• Topic \( 2.6 \) — Resonance and Formal Charge (Part \( \mathrm{(d)(ii)} \))
• Topic \( 7.3 \) — Reaction Quotient and Equilibrium Constant (Part \( \mathrm{(e)} \))
• Topic \( 7.4 \) — Calculating the Equilibrium Constant (Part \( \mathrm{(f)} \))
• Topic \( 7.8 \) — Representations of Equilibrium (Part \( \mathrm{(f)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value reported with the correct number of significant figures:
\( 1.25\ \mathrm{mol\ AlCl}_{3} \times \dfrac{3\ \mathrm{mol\ Cl}}{1\ \mathrm{mol\ AlCl}_{3}} \times \dfrac{35.45\ \mathrm{g\ Cl}}{1\ \mathrm{mol\ Cl}} = 133\ \mathrm{g\ Cl} \)
First use the mole ratio from reaction \( 1 \), then convert moles of \( \mathrm{Cl} \) to grams using its molar mass.
(b)
For the correct algebraic manipulation of either \( \Delta H^\circ_{2} \) or \( \Delta H^\circ_{4} \) (may be implicit):
Accept one of the following:
• Reversing reaction \( 2 \):
\( \mathrm{AlCl}_{3}(g) \rightarrow \mathrm{Al}(s) + \dfrac{3}{2}\,\mathrm{Cl}_{2}(g) \qquad \Delta H^\circ_{\mathrm{rxn}} = -(-583) = +583\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
• Multiplying reaction \( 4 \) by \( \dfrac{3}{2} \):
\( \dfrac{3}{2}\,\mathrm{Cl}_{2}(g) \rightarrow 3\,\mathrm{Cl}(g) \qquad \Delta H^\circ_{\mathrm{rxn}} = \dfrac{3}{2}(243) = +365\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
For the correct calculated value:
\( \Delta H^\circ_{1} = -\Delta H^\circ_{2} + \Delta H^\circ_{3} + 1.5(\Delta H^\circ_{4}) = -(-583) + 326 + 1.5(243) = 1274\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
This is a Hess’s law problem: reverse reaction \( 2 \), keep reaction \( 3 \), and multiply reaction \( 4 \) by \( \dfrac{3}{2} \), then add.
(c)(i)
For the correct answer:
\( 200\ \mathrm{pm} \) \((\pm 10\ \mathrm{pm})\)
The bond length is the internuclear distance at the minimum point of the potential energy curve.
(c)(ii)
For a curve with a minimum at an internuclear distance of \( 220 \pm 10\ \mathrm{pm} \):
See sample curve below.
For a curve with a minimum energy value of \( -425 \pm 20\ \mathrm{kJ/mol} \) that approaches zero as the internuclear distance approaches \( 500\ \mathrm{pm} \):
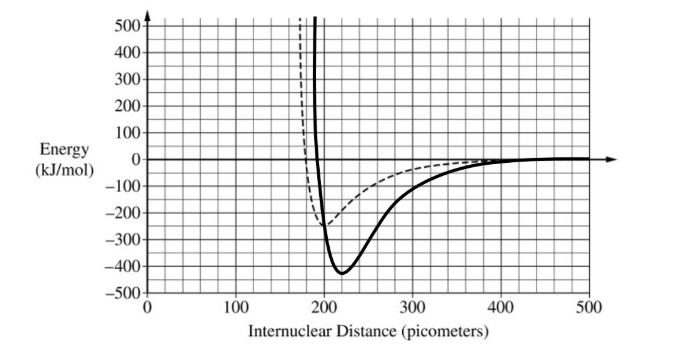
Because the \( \mathrm{Al-Cl} \) bond is longer than the \( \mathrm{Cl-Cl} \) bond, its minimum is shifted slightly to the right. Because its bond energy is greater, the well is deeper.
(d)(i)
For the correct answer and a valid justification:
Diagram \( 2 \). Aluminum has four electron domains in Diagram \( 2 \), which would be trigonal pyramidal, not trigonal planar.
A trigonal planar molecule has \( 3 \) electron domains around the central atom, not \( 4 \).
(d)(ii)
For the correct answer and a valid justification:
Diagram \( 1 \). All atoms in diagram \( 1 \) have a formal charge of zero, whereas atoms in diagrams \( 2 \) and \( 3 \) have nonzero formal charges.
The best Lewis structure is usually the one with the smallest magnitude of formal charges, especially when all formal charges can be zero.
(e)
For the correct answer:
\( K_p = \dfrac{P_{\mathrm{Al}_{2}\mathrm{Cl}_{6}}}{\left(P_{\mathrm{AlCl}_{3}}\right)^2} \)
The exponent \( 2 \) on \( P_{\mathrm{AlCl}_{3}} \) comes directly from the coefficient \( 2 \) in the balanced equilibrium equation.
(f)
For the correct calculated value, consistent with part \( \mathrm{(e)} \):
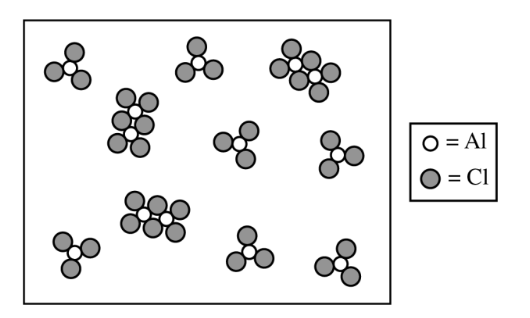
From the particulate diagram, there are \( 3 \) molecules of \( \mathrm{Al}_{2}\mathrm{Cl}_{6} \) and \( 7 \) molecules of \( \mathrm{AlCl}_{3} \).
Therefore, \( \chi_{\mathrm{Al}_{2}\mathrm{Cl}_{6}} = \dfrac{3}{10} \) and \( \chi_{\mathrm{AlCl}_{3}} = \dfrac{7}{10} \)
So, \( P_{\mathrm{Al}_{2}\mathrm{Cl}_{6}} = \dfrac{3}{10}(22.1) \) and \( P_{\mathrm{AlCl}_{3}} = \dfrac{7}{10}(22.1) \)
\( K_p = \dfrac{\chi_{\mathrm{Al}_{2}\mathrm{Cl}_{6}}(P_{\mathrm{total}})}{\left(\chi_{\mathrm{AlCl}_{3}}(P_{\mathrm{total}})\right)^2} = \dfrac{\dfrac{3}{10}(22.1)}{\left(\dfrac{7}{10}(22.1)\right)^2} = 0.0277 \)
Thus, \( \boxed{K_p = 0.0277} \).
