AP Chemistry 2.3 Structure of Ionic Solids - Exam Style questions - FRQs- New Syllabus
Question
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.10 \) — Solubility (Part \( \mathrm{(b)} \), Part \( \mathrm{(d)} \))
• Topic \( 3.8 \) — Representations of Solutions (Part \( \mathrm{(c)} \))
• Topic \( 7.12 \) — Common-Ion Effect (Part \( \mathrm{(d)} \))
▶️ Answer/Explanation
(a)
For a correct description:
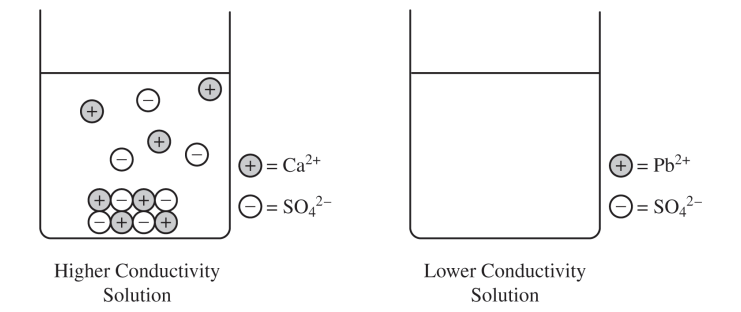
Ionic solids do not have free-moving ions that are required to carry an electric current. Therefore, there is no conduction of electricity.
In both solids, the ions are locked into fixed positions in a crystal lattice, so they cannot move from place to place and carry charge.
(b)
For the correct answer and a valid justification:
\( \mathrm{CaSO_4} \). The greater electrical conductivity of the \( \mathrm{CaSO_4} \) solution relative to the \( \mathrm{PbSO_4} \) solution implies a higher concentration of ions, which comes from the dissolution \( (\text{dissociation}) \) of \( \mathrm{CaSO_4} \) to a greater extent.
More dissolved ions in solution means better conductivity, so the more conductive beaker must contain the more soluble salt.
(c)
For a correct drawing that shows an equal number of cations and anions:
The drawing shows solid \( \mathrm{PbSO_4} \) at the bottom of the beaker \( (\text{similar to the solid shown for } \mathrm{CaSO_4}) \) and fewer dissolved \( \mathrm{Pb^{2+}} \) and \( \mathrm{SO_4^{2-}} \) ions in the solution.
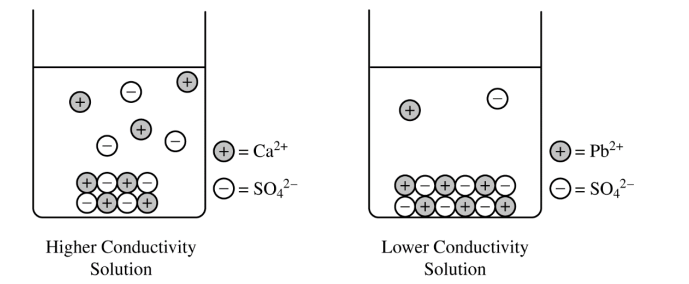
The number of dissolved \( \mathrm{Pb^{2+}} \) ions must equal the number of dissolved \( \mathrm{SO_4^{2-}} \) ions, because the dissolved salt separates in a \( 1:1 \) ratio.
(d)
For a correct explanation:
The additional precipitate is \( \mathrm{CaSO_4} \) that forms in response to the increased \( [\mathrm{SO_4^{2-}}] \) in solution. According to Le Châtelier’s principle \( (Q > K_{sp}) \), the introduction of \( \mathrm{SO_4^{2-}} \) as a common ion shifts the equilibrium towards the formation of more \( \mathrm{CaSO_4}(s) \).
Adding \( \mathrm{H_2SO_4} \) increases sulfate ion concentration, so the dissolution equilibrium \( \mathrm{CaSO_4}(s) \rightleftharpoons \mathrm{Ca^{2+}}(aq) + \mathrm{SO_4^{2-}}(aq) \) shifts left and more solid forms.
