AP Chemistry 2.5 Lewis Diagrams - Exam Style questions - FRQs- New Syllabus
Question
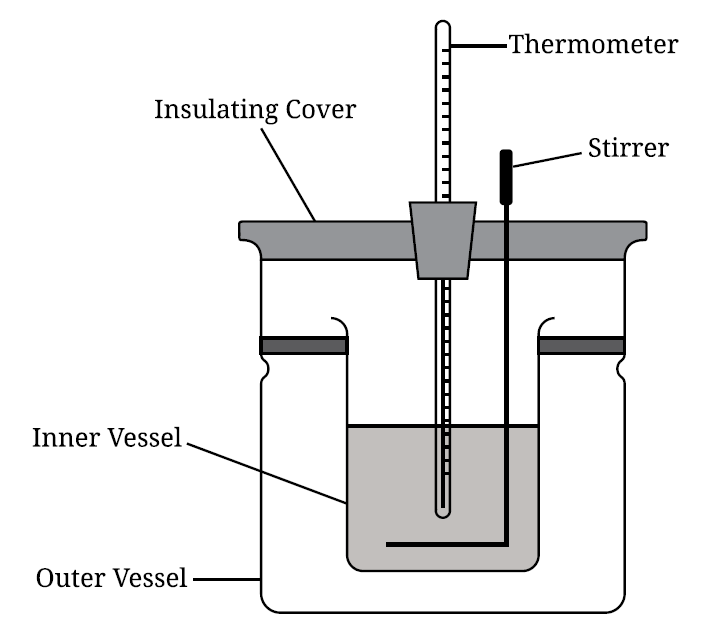
| Mass of \( \mathrm{P_4O_{10}} \) | \(0.100\ \mathrm{g}\) |
| Mass of \( \mathrm{H_2O} \) | \(100.0\ \mathrm{g}\) |
| Initial temperature | \(22.00^\circ\mathrm{C}\) |
| Final temperature | \(22.38^\circ\mathrm{C}\) |
| Molar mass of \( \mathrm{P_4O_{10}} \) | \(283.9\ \mathrm{g\ mol^{-1}}\) |
| Specific heat of \( \mathrm{H_2O} \) | \(4.18\ \mathrm{J\ g^{-1}\ ^\circ C^{-1}}\) |
Most-appropriate topic codes (AP Chemistry CED)
• Topic 9.1 — Introduction to Entropy (entropy changes in reactions)
• Topic 9.3 — Gibbs Free Energy and Thermodynamic Favorability (relationship \( \Delta G^\circ = \Delta H^\circ – T\Delta S^\circ \))
• Topic 6.3 — Heat Transfer and Thermal Equilibrium (calorimetry and \( q = mc\Delta T \))
• Topic 6.4 — Heat Capacity and Calorimetry (calculating enthalpy change)
• Topic 6.9 — Hess’s Law (combining thermochemical equations)
• Topic 7.4 — Calculating the Equilibrium Constant (\(K_p\) from partial pressures)
• Topic 7.10 — Reaction Quotient and Le Châtelier’s Principle (temperature effects on equilibrium)
▶️ Answer/Explanation
A
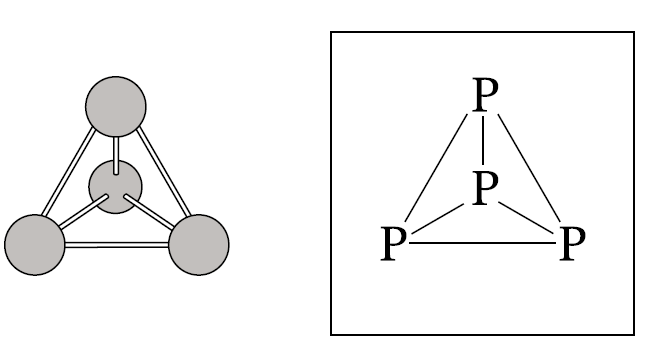
Each phosphorus atom forms three bonds and therefore has one lone pair of electrons. Thus one lone pair is present on each \( \mathrm{P} \) atom in the tetrahedral \( \mathrm{P_4} \) structure.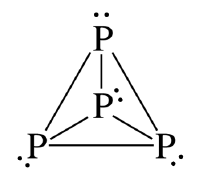
B(i)
Gas particles have more possible arrangements than solids. During the reaction gaseous \( \mathrm{O_2} \) molecules are converted into solid \( \mathrm{P_4O_{10}} \), so particle freedom decreases. Therefore entropy decreases and \( \Delta S^\circ < 0 \).
B(ii)
Yes. \( \Delta G^\circ = \Delta H^\circ – T\Delta S^\circ \). Since \( \Delta H^\circ < 0 \), the reaction releases energy and favors spontaneity. Because \( \Delta S^\circ < 0 \), entropy does not favor spontaneity. Therefore the reaction is driven by enthalpy.
C(i)
\( q = mc\Delta T \)
\( q = (100.1)(4.18)(22.38-22.00) \)
\( q = 160\ \mathrm{J} = 0.160\ \mathrm{kJ} \)
C(ii)
\( q_{rxn} = -q_{solution} = -0.160\ \mathrm{kJ} \)
moles \( \mathrm{P_4O_{10}} = \frac{0.100}{283.9} = 3.52\times10^{-4}\ \mathrm{mol} \)
\( \Delta H^\circ_{rxn} = \frac{-0.160}{3.52\times10^{-4}} = -450\ \mathrm{kJ\ mol^{-1}_{rxn}} \)
D
Less than. Because some \( \mathrm{P_4O_{10}} \) did not react, fewer moles reacted and less heat was released. Therefore the temperature increase is smaller.
E
Using Hess’s law:
\( \Delta H_f^\circ = \frac14\Delta H_1^\circ + \Delta H_2^\circ \)
\( \Delta H_f^\circ = \frac14(-1148) + (-88) = -375\ \mathrm{kJ\ mol^{-1}} \)
F(i)
\( K_p = \frac{P_{PCl_5}}{P_{PCl_3}P_{Cl_2}} \)
\( K_p = \frac{4.00}{(2.00)(6.00)} = 0.333 \)
F(ii)
Decrease. Because \( \Delta H^\circ_2 \) is negative, the forward reaction is exothermic. Increasing temperature favors the reverse reaction, so \( K_p \) decreases.
