Question

Which of the diagrams above best represents the CH2O molecule, and why?
Answer/Explanation
Ans:D
Diagram 2 represents a molecule in which both C and O have a complete octet and all atoms have a formal charge of zero.
Question
Based on formal charges, which of the following is the best Lewis electron-dot diagram for H3NO?




Answer/Explanation
Ans:A
When the formal charge of each atom in this diagram is determined using the rule (formal charge of an atom = the number of valence electrons of the atom − (the number of electrons in lone pairs + half the number of electrons in bonds with other atoms)), the sum of the formal charges on all the atoms in H3NO is equal to zero. A sum of formal charges on all atoms equal to zero is preferable.
Question

Which of the Lewis diagrams shown above is the more likely structure of CO2, and why?
Answer/Explanation
Ans:A
When the formal charge of each atom in diagram 1 is determined using the rule (formal charge of an atom = the number of valence electrons of the atom − (the number of electrons in lone pairs + half the number of electrons in bonds with other atoms)), the results are zero for each of the three atoms. A molecular structure in which all formal charges are zero is preferable to one in which some formal charges are not zero. And in diagram 2, the results for the three atoms (going left to right) are −1, 0, and +1, respectively.
Question
Which of the following Lewis electron-dot diagrams represents the molecule that contains the smallest bond angle?
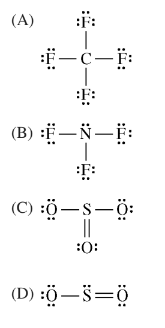
Answer/Explanation
Ans:A
Question
Which of the following Lewis electron-dot diagrams represents the molecule that is the most polar?

Answer/Explanation
Ans:C
