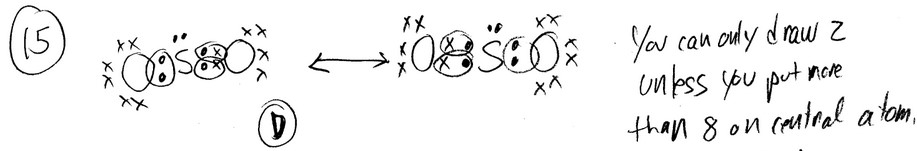AP Chemistry 2.6 Resonance and Formal Charge- MCQs - Exam Style Questions
Questions
The diagram above represents a particle in aqueous solution. Which of the following statements about the particle is correct?
(A) The particle must be a cation because the negative end of each water molecule is pointed toward it.
(B) The particle must be an anion because the positive end of each water molecule is pointed toward it.
(C) The charge of the particle cannot be determined because water molecules have no net charge.
(D) The charge of the particle cannot be determined because the water molecules are arranged symmetrically and their partial charges cancel.
▶️Answer/Explanation
Ans: B
Looking at the diagram, where water molecules surround the particle, let’s analyze the interaction between the water molecules and the particle:
- Water molecules are polar, with a partial negative charge (δ-) on the oxygen atom and a partial positive charge (δ+) on the hydrogen atoms.
- In aqueous solution, the positively charged (cationic) end of water molecules is attracted to negatively charged particles (anions), while the negatively charged (anionic) end is attracted to positively charged particles (cations).
Given this information, let’s analyze the options:
(A) The particle must be a cation because the negative end of each water molecule is pointed toward it. This statement is incorrect. If the negative end of water molecules is pointed toward the particle, it suggests that the particle is positively charged (anion), not a cation.
(B) The particle must be an anion because the positive end of each water molecule is pointed toward it. This statement is incorrect. If the positive end of water molecules is pointed toward the particle, it suggests that the particle is negatively charged (cation), not an anion.
(C) The charge of the particle cannot be determined because water molecules have no net charge. This statement is incorrect. While water molecules themselves are neutral, their orientation around the particle can provide information about the charge of the particle.
(D) The charge of the particle cannot be determined because the water molecules are arranged symmetrically and their partial charges cancel. This statement is incorrect. The arrangement of water molecules around the particle indicates an interaction based on the charge of the particle. If the arrangement were symmetric and canceled out, there would be no preferential orientation of water molecules around the particle.
Given the options, none of them are correct. However, the closest option to being correct is option (B), but it misinterprets the orientation of water molecules. The correct interpretation is that the positive end of water molecules is pointed toward the negatively charged particle, indicating that the particle must be an anion.
Therefore, none of the given options accurately describe the situation.
Question
(i) \(PCl_3\) (ii) \(CH_2Cl_2\) (iii) HCN (iv) \(C_2H_4\) (v) \(NH_3\)
In which Lewis structure does the central atom have a non-zero formal charge?
A) (i) B) (ii) C) (iii) D) (v) E) none
▶️Answer/Explanation
Ans: E

Question
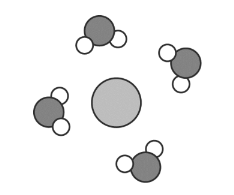
The formal charge on carbon in the molecule below is __________.
A) 0 B) +1 C) -1 D) +3 E) +2
▶️Answer/Explanation
Ans: A
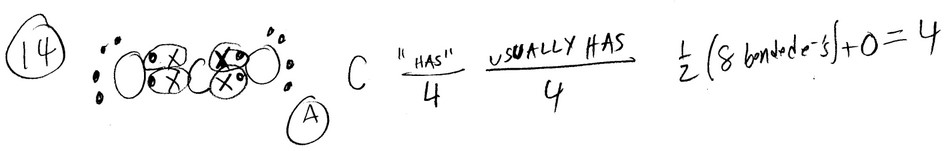
Question
How many equivalent resonance forms can be drawn for SO2 without expanding octet on the sulfur atom
(sulfur is the central atom)?
A) 3 B) 0 C) 4 D) 2 E) 1
▶️Answer/Explanation
Ans: D