AP Chemistry 3.3 Solids, Liquids, and Gases- Exam Style questions - FRQs- New Syllabus
Question

| Substance | Melting Point \((K)\) | Boiling Point \((K)\) | Enthalpy of Vaporization \((\mathrm{kJ/mol})\) |
|---|---|---|---|
| \( \mathrm{CH_3OH} \) | \(176\) | \(338\) | \(37.6\) |
| \( \mathrm{H_2CO} \) | \(181\) | \(254\) | \(24.2\) |
Most-appropriate topic codes (AP Chemistry):
• Topic \(3.1\) — Intermolecular and Interparticle Forces — Part B
• Topic \(3.3\) — Solids, Liquids, and Gases — Part C(i)
• Topic \(6.5\) — Energy of Phase Changes — Part C(ii)
▶️ Answer/Explanation
A
For the correct answer:
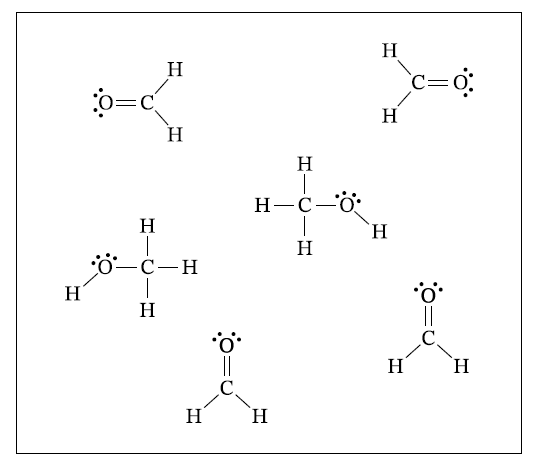
\( \mathrm{sp^2} \)
The \( \mathrm{C} \) atom in \( \mathrm{H_2CO} \) has three regions of electron density around it: two single bonds and one double bond.
A molecule with three electron domains around the central atom has \( \mathrm{sp^2} \) hybridization.
B
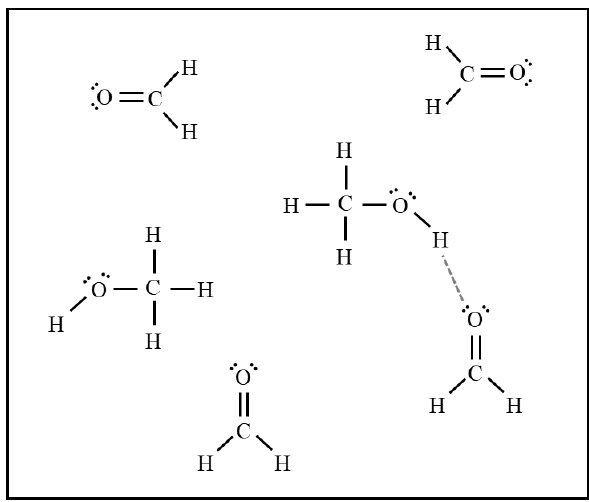
For a correct diagram:
The diagram should show a dashed line between the \( \mathrm{O} \) atom in one \( \mathrm{H_2CO} \) molecule and the \( \mathrm{H} \) atom in the \( \mathrm{-OH} \) group of one \( \mathrm{CH_3OH} \) molecule. See example response below.
This is because hydrogen bonding occurs when an \( \mathrm{H} \) attached to \( \mathrm{O} \) is attracted to a lone pair on another electronegative atom such as \( \mathrm{O} \).

C(i)
For a correct proposal:
The proposed temperature should be in the range \(181\ \mathrm{K} – 254\ \mathrm{K}\).
To be liquids, the temperature must be above each substance’s melting point and below each substance’s boiling point.
Any temperature in this overlap range is acceptable, for example \(200\ \mathrm{K}\).
C(ii)
For the correct calculated value:
\[ 8.59\ \mathrm{g\ CH_3OH} \times \frac{1\ \mathrm{mol\ CH_3OH}}{32.04\ \mathrm{g\ CH_3OH}} \times \frac{-37.6\ \mathrm{kJ}}{1\ \mathrm{mol\ CH_3OH}} = -10.1\ \mathrm{kJ} \]
First convert grams of \( \mathrm{CH_3OH} \) to moles, then multiply by the enthalpy of vaporization.
The negative sign shows energy is released by the substance during condensation.
Therefore, \(10.1\ \mathrm{kJ}\) are removed.
