AP Chemistry 3.5 Kinetic Molecular Theory - Exam Style questions - FRQs- New Syllabus
Question
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.5 \) — Kinetic Molecular Theory (Part \( \mathrm{(c)} \))
• Topic \( 3.6 \) — Deviation from Ideal Gas Law (Part \( \mathrm{(d)} \))
▶️ Answer/Explanation
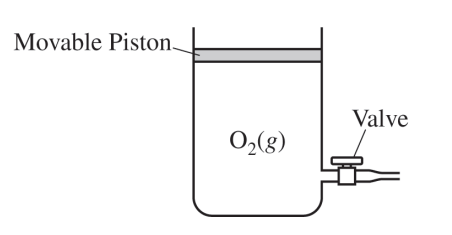
(a)
For the correct calculated value:
Accept one of the following:
• \( 0.325\ \mathrm{mol\ O_2} \times \dfrac{32.00\ \mathrm{g\ O_2}}{1\ \mathrm{mol\ O_2}} = 10.4\ \mathrm{g\ O_2} \)
\( D = \dfrac{m}{V} = \dfrac{10.4\ \mathrm{g}}{7.95\ \mathrm{L}} = 1.31\ \mathrm{g/L} \)
• \( D = \dfrac{m}{V} = \dfrac{P(\mathrm{MM})}{RT} = \dfrac{(1.0\ \mathrm{atm})(32.00\ \mathrm{g/mol})}{(0.08206\ \mathrm{L \cdot atm \cdot mol^{-1} \cdot K^{-1}})(298\ \mathrm{K})} = 1.31\ \mathrm{g/L} \)
So, the density is \( \boxed{1.31\ \mathrm{g/L}} \).
Both methods agree, which is a nice check on the calculation.
(b)
For the correct answer and a valid justification:
Accept one of the following:
• No, the density of the gas remains constant because \( P \), \( R \), and \( T \) remain constant and the mass and volume of \( \mathrm{O_2} \) decrease proportionately.
• A mathematical justification is shown below.
\( D = \dfrac{m}{V} = \dfrac{n\ \mathrm{moles\ of\ O_2} \times \mathrm{molar\ mass\ of\ O_2}}{nRT/P} = \dfrac{P \times (\mathrm{molar\ mass\ of\ O_2})}{RT} \)
Because the temperature stays constant and the piston is movable, the gas readjusts to the same pressure. That keeps the density unchanged.
(c)
For a valid explanation:
Accept one of the following:
• As the gas cools, the average kinetic energy \( (\text{speed}) \) of the \( \mathrm{O_2} \) molecules decreases. The molecules rebound with less energy when they collide with each other and the walls of the container. The spacing between particles decreases, causing the volume occupied by the gas to decrease.
• As the gas cools, the average kinetic energy \( (\text{speed}) \) of the \( \mathrm{O_2} \) molecules decreases. The molecules rebound with less energy when they collide with each other and the walls of the container. The only way for the molecules to maintain a constant rate of collisions with the walls of the container \( (\text{maintaining a pressure of } 1.00\ \mathrm{atm}) \) is for the volume of the gas to decrease.
Lower temperature means slower particles, so the piston moves downward until the gas once again exerts the outside pressure.
(d)
For a valid explanation:
The ideal gas law assumes that gas particles do not experience interparticle attractions. As a real gas cools further, the intermolecular forces have greater effect as the average speed of the molecules decreases, resulting in inelastic collisions. To maintain a gas pressure of \( 1.00\ \mathrm{atm} \), the volume must decrease to accommodate more collisions with less energy.
At \( -180^\circ \mathrm{C} \), the temperature is very close to the boiling point of oxygen, so the gas behaves much less ideally. Attractive forces become important, and some particles are close to condensing, which makes the measured volume smaller than the ideal-gas prediction.
