AP Chemistry 3.9 Separation of Solutions and Mixtures Chromatography- Exam Style questions - FRQs- New Syllabus
Question
• Balance • \( 100.0\ \mathrm{mL} \) volumetric flask • \( 50.0\ \mathrm{mL} \) graduated cylinder
• Pipet • \( 150\ \mathrm{mL} \) beakers • Chromatography paper
| Step | Step Description and Materials Used |
|---|---|
| 1. | Use the weighing paper and scoop to measure the correct mass of solid \( \mathrm{NaCl} \) on the balance. |
| 2. | |
| 3. | Swirl the mixture to dissolve the solid \( \mathrm{NaCl} \). |
| 4. | |
| 5. | Stopper and invert the mixture several times to ensure that the mixture is homogeneous. |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.9 \) — Separation of Solutions and Mixtures (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value:
\( 0.1000\ \mathrm{L} \times \dfrac{0.340\ \mathrm{mol}}{1\ \mathrm{L}} \times \dfrac{58.44\ \mathrm{g}}{1\ \mathrm{mol}} = 1.99\ \mathrm{g\ NaCl} \)
First find the moles needed:
\( n = MV = (0.340)(0.1000) = 0.0340\ \mathrm{mol} \)
Then convert moles to grams using the molar mass.
(b)
For a correct description of step \( 2 \):
Combine the solid \( \mathrm{NaCl} \) and some distilled water in a \( 100.0\ \mathrm{mL} \) volumetric flask.
A little extra explanation: only add some water first so the solid can dissolve completely before the flask is filled to the final mark.
For a correct description of step \( 4 \):
Fill the volumetric flask with distilled water to the calibration \((100.0\ \mathrm{mL})\) mark.
The volumetric flask is used because it gives the correct final solution volume more accurately than a beaker or graduated cylinder.
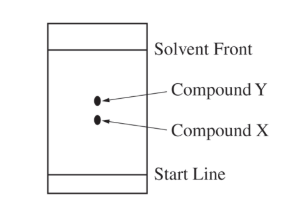
(c)
For the correct prediction and a valid explanation:
It would decrease. The solvent front would not travel as far in the second experiment, so the separation would be smaller.
Because the paper is removed sooner, both compounds have less time to move with the solvent. Therefore, the distance between the \( X \) and \( Y \) spots would be reduced.
