AP Chemistry 4.2 Net Ionic Reaction -MCQs - Exam Style Questions
Question
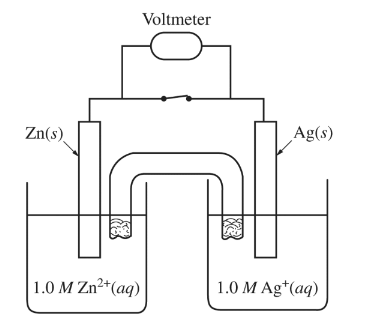
Standard reduction potentials for the half-reactions associated with the electrochemical cell shown above are given in the table below.
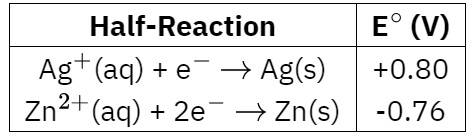
What is the value of E$^\circ$ for the cell?
A. 2.36 V
B. 0.04 V
C. 1.56 V
D. 0.84 V
▶️Answer/Explanation
Ans: B
Question
A student combines a solution of NaCl(aq) with a solution of \(AgNO_3\)(aq) , and a precipitate forms. Which of the following is the balanced net ionic equation for the formation of the precipitate?
A \(Ag^+(aq)+Cl^-(aq)→AgCl(s)\)
B \(Na^+(aq)+NO3^-(aq)→NaNO3(s)\)
C \(NaCl(aq)+AgNO_3(aq)→NaNO_3(s)+AgCl(aq)\)
D \(NaCl(aq)+AgNO_3(aq)→NaNO_3(aq)+AgCl(s)\)
▶️Answer/Explanation
Ans: A
Only the \(Ag^+\)(aq) and \(Cl^-\)(aq) ions react, forming the insoluble precipitate AgCl(s). The solution mixture also contains the nonreacting spectator ions \(Na^+\)(aq) and \(NO3^-\)(aq), which are not included in a net ionic equation
Question
Which of the following is the correct net ionic equation of the neutralization reaction between hydrofluoric acid and sodium hydroxide in aqueous solution?
A \(HF(aq)+NaOH(aq)→H_2O(l)+NaF(aq)\)
B \(HF(aq)+NaOH(aq)→NaH(aq)+FOH(aq)\)
C \(H^+(aq)+OH^−(aq)→H_2O(l)\)
D \(HF(aq)+OH^−(aq)→H2O(l)+F^−(aq)\)
▶️Answer/Explanation
Ans: D
The correct net ionic equation includes the reacting species (HF and \(OH^−\)) and the product(s) (\(H_2O\) and \(F^−\)) and does not include any spectator ions. HF is a weak acid and ionizes only partially in aqueous solution; thus, HF is correctly represented as undissociated (molecular) in the net ionic equation. NaOH is a strong base, and would dissociate completely into \(Na^+ \)and \(OH^-\) ions. The net ionic equation should include the reacting species (HF and \(OH^−\) and the product(s) (\(H_2O\) and \(F^−\)). Na+ is a spectator ion in this reaction and is not included in the net ionic equation.
Question
Which of the following is the correct net ionic equation of the reaction that occurs when aqueous solutions of ammonia and hydrochloric acid are combined?
A \(NH_3(aq)+HCl(aq)→NH_4Cl(aq)\)
B \(NH_3(aq)+H^+(aq)→NH_4^+(aq)\)
C \(NH_3(aq)+HCl(aq)→NH_4^+(aq)+Cl^−(aq)\)
D \(NH_3(aq)+H^+(aq)+Cl^−(aq)→NH_4+(aq)+Cl^−(aq)\)
▶️Answer/Explanation
Ans: B
The net ionic equation includes the reacting species (\(NH_3 and H^+\)) and the product (\(NH_4^+\)). HCl is a strong acid and completely dissociates in water. \(Cl^−\) is a spectator ion; thus, it is not included in the net ionic equation.
Question
Which of the following equations best represents the species that react and the species that are produced when \(CaCO_3\)(s) and HCl(aq) are combined?
(A)\( 2 H^{+}(aq) + CaCO_{3}(s) \rightarrow Ca^{2+}(aq) + CO_{3}^{2-}(aq) + H_{2}\)(g)
(B) \(2 H^{+}(aq) + CaCO_{3}(s) \rightarrow Ca^{2+}(aq) + H_2O(l) + CO_2(g)\)
(C) \(2 H^{+}(aq) + CaCO_3(s) \rightarrow Ca^{2+}(aq) + 2 OH^{-} (aq) + CO(g) \)
(D) \(2 HCl(aq) + CaCO_{3}(s) \rightarrow Ca^{2+}(aq) + H_2O(l) + CO_{2}(g) + 2 Cl^{-}\)(aq)
▶️Answer/Explanation
Ans:D
