AP Chemistry 5.11 Catalyst -Exam Style Questions- FRQs- New Syllabus
Question
Step \( 2 \): \( \mathrm{C_2H_5O_3^+ + H_2O \rightleftharpoons HC_2H_3O_3 + H_3O^+} \)
Overall: \( \mathrm{C_3H_6O_3 + H_2O \rightleftharpoons HC_2H_3O_3 + CH_3OH} \)
Most-appropriate topic codes (AP Chemistry):
• Topic \( 8.3 \) — Weak Acid and Base Equilibria — Part B(i)
• Topic \( 8.3 \) — Weak Acid and Base Equilibria — Part B(ii)
• Topic \( 5.11 \) — Catalysis — Part C
▶️ Answer/Explanation
A
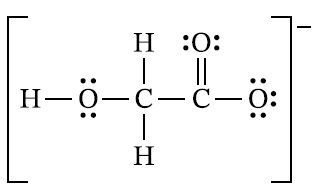
For a correct circled atom:
Accept one of the following:
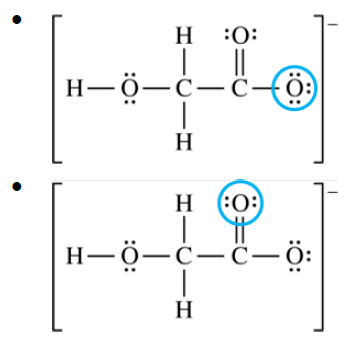

Because of resonance, the two \( \mathrm{C-O} \) bonds on the right are equivalent.
So either of those two \( \mathrm{O} \) atoms can be accepted as the proton-accepting atom.
B(i)
For the correct calculated value:
\( K_b = \dfrac{[\mathrm{HC_2H_3O_3}][\mathrm{OH^-}]}{[\mathrm{C_2H_3O_3^-}]} \)
At equilibrium, \( [\mathrm{HC_2H_3O_3}] = [\mathrm{OH^-}] = 1.3 \times 10^{-5}\ \mathrm{M} \), and
\( [\mathrm{C_2H_3O_3^-}] = 2.5 – 1.3 \times 10^{-5}\ \mathrm{M} \).
\( K_b = \dfrac{(1.3 \times 10^{-5})(1.3 \times 10^{-5})}{2.5 – 1.3 \times 10^{-5}} \approx \dfrac{(1.3 \times 10^{-5})^2}{2.5} = 6.8 \times 10^{-11} \)
So, \( \boxed{K_b = 6.8 \times 10^{-11}} \).
B(ii)
For the correct calculated value, consistent with part B(i):
\( K_a = \dfrac{K_w}{K_b} \)
\( K_a = \dfrac{1.0 \times 10^{-14}}{6.8 \times 10^{-11}} = 1.5 \times 10^{-4} \)
Using \( K_w = K_aK_b \), divide \( K_w \) by the \( K_b \) value from part B(i).
Thus, \( \boxed{K_a = 1.5 \times 10^{-4}} \).
C
For the correct answer and a valid justification:
Agree. \( \mathrm{H_3O^+} \) is consumed in step \( 1 \) and regenerated in step \( 2 \), which is consistent with the behavior of a catalyst.
A catalyst participates in the mechanism but does not appear in the overall reaction.
Since \( \mathrm{H_3O^+} \) is used and then produced again, its net amount does not change.
