AP Chemistry 5.3 Concentration Change Over Time- Exam Style questions - FRQs- New Syllabus
Question
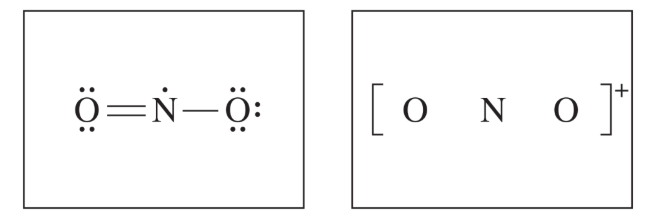
(ii) A student makes the claim that the bond angles in \( \mathrm{NO_2} \) and \( \mathrm{NO_2^+} \) are different from each other. Do you agree or disagree with the student’s claim? Justify your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 4.5 \) — Stoichiometry (Part \( \mathrm{(b)} \))
• Topic \( 2.5 \) — Lewis Diagrams (Part \( \mathrm{(c)(i)} \))
• Topic \( 2.7 \) — VSEPR and Hybridization (Part \( \mathrm{(c)(ii)} \))
▶️ Answer/Explanation
(a)
For a correct explanation:
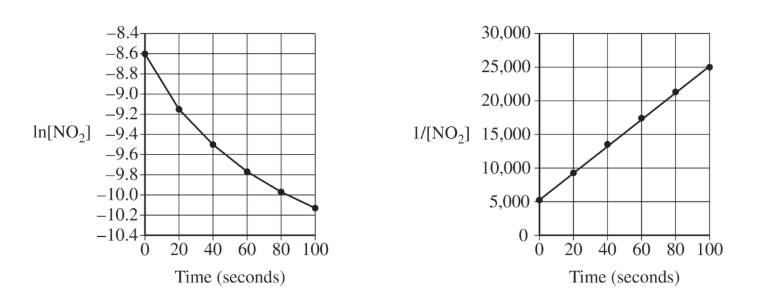
The plot of \( \dfrac{1}{[\mathrm{NO_2}]} \) versus time is the most linear, indicating that the reaction is second order with respect to \( \mathrm{NO_2} \).
For a second-order reaction in one reactant, the integrated rate law predicts a linear relationship between \( \dfrac{1}{[\mathrm{A}]} \) and time, so the straight-line graph supports the claim.
(b)
For the correct calculated value:
From the balanced equation, \( \mathrm{2\,NO_2 \rightarrow 2\,NO + O_2} \)
This means: \( \dfrac{1\ \mathrm{mol\ O_2}}{2\ \mathrm{mol\ NO_2}} \)
Therefore, \( 6.52 \times 10^{-7}\ \mathrm{M/s} \times \dfrac{1\ \mathrm{mol\ O_2}}{2\ \mathrm{mol\ NO_2}} = 3.26 \times 10^{-7}\ \mathrm{M/s} \)
So, the rate of appearance of \( \mathrm{O_2} \) is \( \boxed{3.26 \times 10^{-7}\ \mathrm{M/s}} \).
(c)(i)
For the correct Lewis diagram:
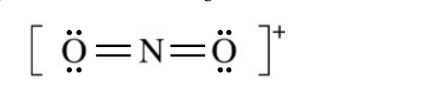
In \( \mathrm{NO_2^+} \), nitrogen forms two double bonds to oxygen and has no unpaired electron.
Each oxygen has two lone pairs, and the entire structure is enclosed in brackets with an overall \( + \) charge.
(c)(ii)
For the correct answer and a valid justification, consistent with part \( \mathrm{(c)(i)} \):
Agree. The angle of \( \mathrm{NO_2^+} \) is different from the angle in \( \mathrm{NO_2} \) because there would no longer be a nonbonding electron on the central atom in \( \mathrm{NO_2^+} \), and the oxygen atoms would spread farther apart, forming a linear structure with a \( 180^\circ \) bond angle.
Another valid way to justify this is by hybridization:
The hybridization of \( \mathrm{N} \) in \( \mathrm{NO_2} \) is \( \mathrm{sp^2} \), which gives a bond angle of approximately \( 120^\circ \). The hybridization of \( \mathrm{N} \) in \( \mathrm{NO_2^+} \) is \( \mathrm{sp} \), which gives a bond angle of \( 180^\circ \).
