AP Chemistry 5.6 Reaction Energy Profile - MCQs - Exam Style Questions
Questions
To catalyze a biochemical reaction, an enzyme typically
(A) drives the reaction to completion by consuming byproducts of the reaction
(B) binds temporarily to reactant molecules to lower the activation energy of the reaction
(C) dissociates into additional reactant molecules, thereby increasing the reaction rate
(D) decomposes and releases energy to increase the number of successful collisions between reactant molecules
▶️Answer/Explanation
Ans: B
The correct answer is:
(B) binds temporarily to reactant molecules to lower the activation energy of the reaction.
Enzymes are biological catalysts that facilitate biochemical reactions by lowering the activation energy required for the reaction to proceed. They achieve this by binding to reactant molecules, forming enzyme-substrate complexes. This binding stabilizes the transition state, making it easier for the reaction to occur, thus lowering the activation energy. Enzymes do not alter the equilibrium constant or drive reactions to completion (A), dissociate into additional reactant molecules (C), or decompose and release energy to increase the number of successful collisions (D).
Question
\(XY_{2} → X + Y_{2}\) The equation above represents the decomposition of a compound \(XY_{2}\). The diagram below shows two reaction profiles (path one and path two) for the decomposition of\( XY_{2}\).
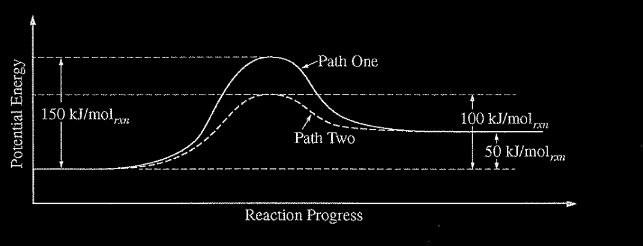
Which of the following most likely accounts for the difference between reaction path one and reaction path two?
(A) A higher temperature in path one
(B) A higher temperature in path two
(C) The presence of a catalyst in path one
(D) The presence of a catalyst in path two
▶️Answer/Explanation
Ans:D
Question
Which energy difference in the energy profile below corresponds to the activation energy for the forward
reaction?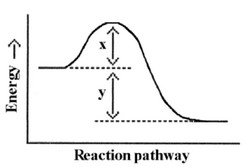
A) x B) y C) x + y D) y – x E) x – y
▶️Answer/Explanation
Ans: A
Question
In the energy profile of a reaction, the species that exists at the maximum on the curve is called the __________.
A) product
B) enthalpy of reaction
C) atomic state
D) activated complex
E) activation energy
▶️Answer/Explanation
Ans: D