AP Chemistry 6.4 Heat Capacity and Calorimetry- Exam Style questions - FRQs- New Syllabus
Question

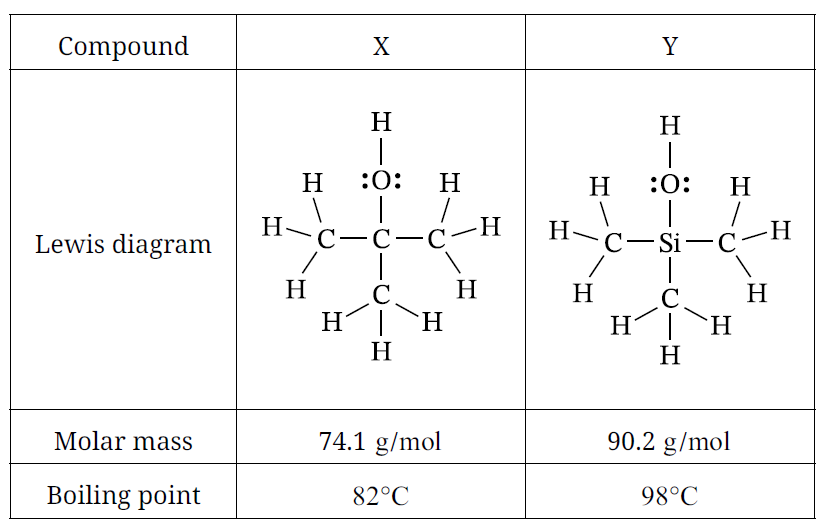
A. Based on VSEPR theory, predict the geometry around the \( \mathrm{Si} \) atom in compound \(Y\).
Most-appropriate topic codes (AP Chemistry):
• Topic \(3.1\) — Intermolecular and Interparticle Forces — Part B
• Topic \(3.3\) — Solids, Liquids, and Gases — Part C
• Topic \(6.4\) — Ideal Gas Law — Part D
▶️ Answer/Explanation
A
For the correct answer:
\( \text{Tetrahedral} \)
The \( \mathrm{Si} \) atom is bonded to four atoms and has no lone pairs on the central atom.
According to VSEPR theory, four electron domains around a central atom give a tetrahedral molecular geometry.
B
For the correct answer and a valid justification:
Agree. Compound \(Y\) has a larger, more polarizable electron cloud because the \( \mathrm{Si} \) atom has more occupied electron shells than the \( \mathrm{C} \) atom, giving compound \(Y\) stronger London dispersion forces and a higher boiling point than compound \(X\).
Since \( \mathrm{Si} \) is larger than \( \mathrm{C} \), its electron cloud is easier to distort.
This increases London dispersion forces, which raises the boiling point.
C
For the correct answer and a valid justification.
Examples of acceptable responses may include the following:
• \( \text{Compound } X \). Compound \(X\) has weaker intermolecular forces than compound \(Y\), so molecules of \(X\) are more likely to be in the gas phase at \(82^\circ \mathrm{C}\) and therefore have a higher vapor pressure.
• \( \text{Compound } X \). At \(82^\circ \mathrm{C}\), compound \(X\) has reached its boiling point, but compound \(Y\) has not. Therefore the proportion of \(X\) molecules in the vapor phase is greater than that of compound \(Y\), giving compound \(X\) the higher vapor pressure.
Because compound \(X\) has the lower boiling point, it is more volatile and thus has the higher vapor pressure at this temperature.
D
For the correct calculated value:
\( PV = nRT \)
\( n = \dfrac{PV}{RT} \)
\( n = \dfrac{(2.30\ \mathrm{atm})(12.5\ \mathrm{L})}{(0.08206\ \mathrm{L\cdot atm\ K^{-1}\ mol^{-1}})(471\ \mathrm{K})} \)
\( n = 0.744\ \mathrm{mol} \)
First convert the temperature to kelvin:
\( 198 + 273 = 471\ \mathrm{K} \).
Substituting the values into the ideal gas law gives the total number of moles of gas particles.
Therefore, the amount of gas in the container is \( \boxed{0.744\ \mathrm{mol}} \).
