AP Chemistry 6.6 Introduction to Enthalpy of Reaction - Exam Style questions - FRQs- New Syllabus
Question
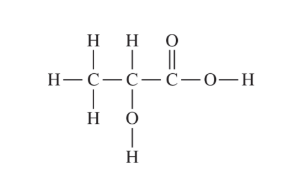
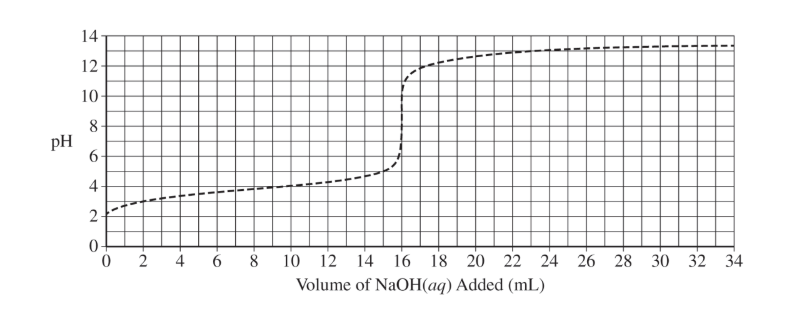
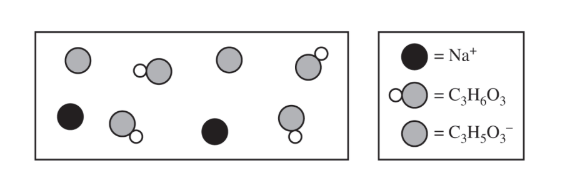
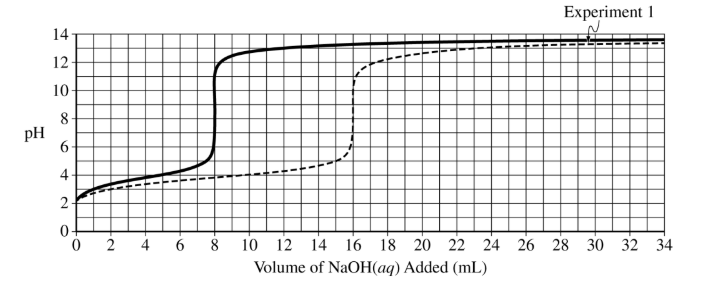
(d) The preceding diagram represents the relative amounts of major species in a sample of the solution in the flask at one point during the titration. (Note that water molecules are omitted.)
(i) Draw an X on the preceding titration curve at a point in the titration where the reaction mixture would be represented by this diagram.
(ii) Justify your answer.
| Experiment | Mass of \( \mathrm{NaOH(s)} \) (grams) | Volume of Solution (mL) | Titration Curve |
|---|---|---|---|
| 1 | \( 10.22 \) | \( 500. \) | Already shown on graph |
| 2 | \( 20.44 \) | \( 500. \) | ? |
(i) Calculate the quantity of heat produced, in J.
(ii) Calculate \( \Delta H_{rxn} \), in \( \mathrm{kJ/mol} \). Include the sign.
(iii) If heat is lost to surroundings, will the calculated \( \Delta H \) be smaller in magnitude? Justify.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 4.6 \) — Introduction to Titration (Part (b))
• Topic \( 8.5 \) — Acid–Base Titrations (Parts (c), (d))
• Topic \( 6.4 \) — Heat Capacity and Calorimetry (Part (e)(i))
• Topic \( 6.6 \) — Introduction to Enthalpy of Reaction (Parts (e)(ii), (e)(iii))
▶️ Answer/Explanation
(a)
The rightmost hydrogen atom should be circled.
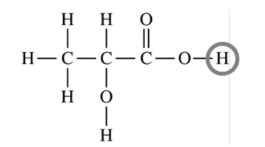
(b)
\( M = \dfrac{10.22}{40.00} \div 0.500 = 0.511\ \mathrm{M} \)
Moles \( = \dfrac{10.22}{40.00} = 0.2555\ \mathrm{mol} \).
(c)
\( pK_a \approx 3.9 \) (acceptable range \( 3.7 – 4.0 \)).
(d)(i)
The X should be at a point greater than or equal to \( 3\ \mathrm{mL} \) and less than \( 8\ \mathrm{mL} \).
(d)(ii)
More acid particles are present than conjugate base particles, meaning the solution is before the half-equivalence point.
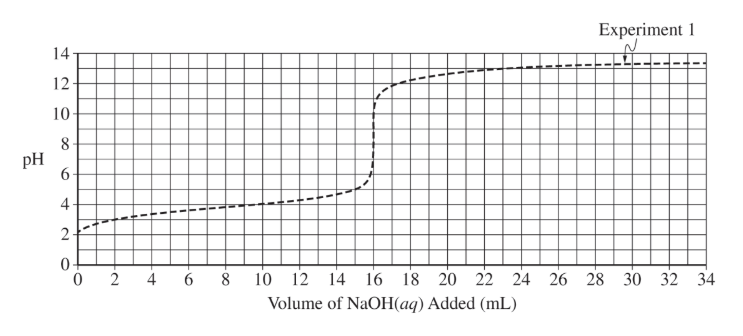
(d)(iii)
The equivalence point should be at \( 8\ \mathrm{mL} \). The curve begins similarly but rises earlier due to higher concentration.
(e)(i)
\( q = mc\Delta T = (200.0)(4.2)(3.2) = 2700\ \mathrm{J} \).
(e)(ii)
\( q_{rxn} = -2.7\ \mathrm{kJ} \)
Moles \( = (0.100)(0.500) = 0.0500\ \mathrm{mol} \)
\( \Delta H_{rxn} = \dfrac{-2.7}{0.0500} = -54\ \mathrm{kJ/mol} \).
(e)(iii)
Agree. Heat loss lowers measured temperature change, so calculated \( \Delta H \) is smaller in magnitude.
