AP Chemistry 7.10 Reaction Quotient and Le Châtelier’s Principle - Exam Style questions - FRQs- New Syllabus
Question
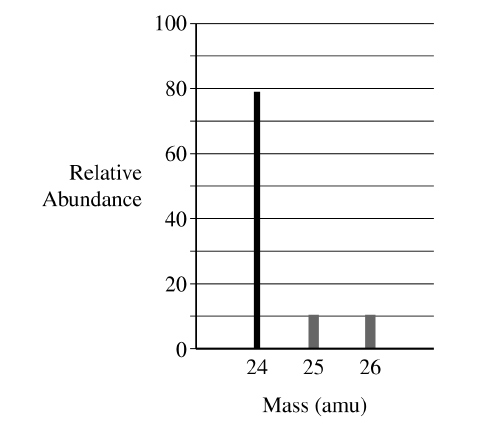
(ii) Describe the difference in atomic structure that accounts for the difference in mass between magnesium-\(25\) and magnesium-\(26\).
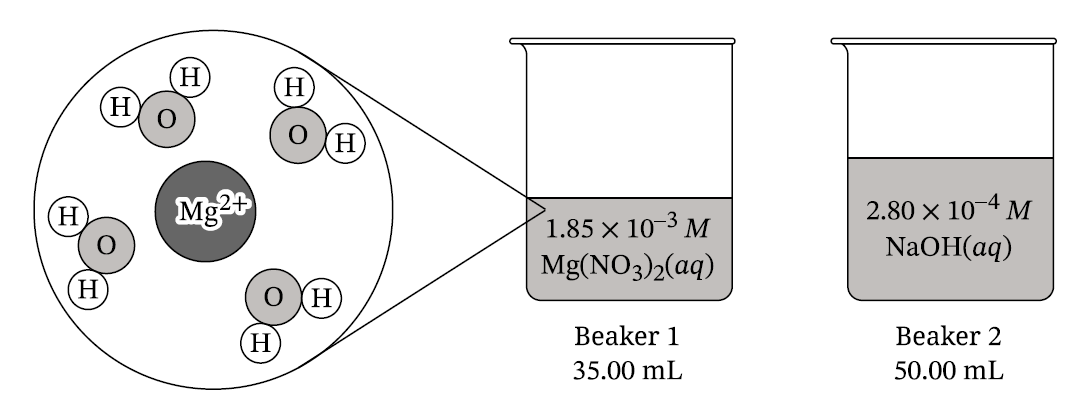
(ii) The relative radii of the ions
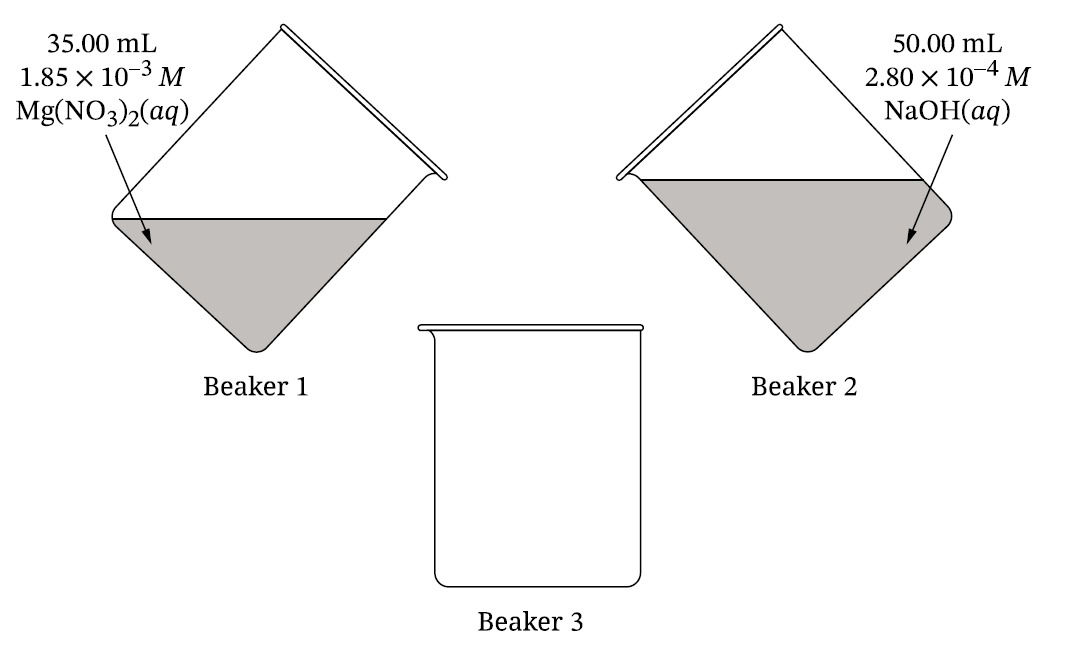
(ii) After the two solutions are combined in beaker \(3\) as described in part \(D\), but before any reaction takes place, \( [\text{OH}^-]=1.65\times10^{-4}\ \text{M}\). Using your answer to part \(D\), calculate the value of the reaction quotient, \(Q\).
(iii) Using the reaction quotient, \(Q\), predict whether a precipitate should form as the mixture in beaker \(3\) approaches equilibrium. Justify your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic 3.1 — Intermolecular and Interparticle Forces: Coulombic attraction between ions and water molecules — part B
• Topic 8.2 — pH and pOH of Strong Acids and Bases: calculating pH from hydroxide concentration — part C
• Topic 3.7 — Solutions and Mixtures: dilution and concentration after mixing solutions — part D
• Topic 7.11 — Solubility Equilibria: writing \(K_{sp}\) expressions — part E(i)
• Topic 7.3 — Reaction Quotient and Equilibrium Constant: calculating \(Q\) — part E(ii)
• Topic 7.10 — Reaction Quotient and Le Châtelier’s Principle: predicting precipitation from \(Q\) vs \(K_{sp}\) — part E(iii)
• Topic 8.11 — pH and Solubility: effect of added acid on solubility equilibrium — part F
▶️ Answer/Explanation
A(i)
The abundances for magnesium-\(25\) and magnesium-\(26\) should each be between \(10\) and \(11\%\). Since magnesium-\(24\) is \(79\%\), the remaining \(21\%\) is approximately equally divided between the other two isotopes.
A(ii)
Magnesium-\(26\) has one more neutron than magnesium-\(25\). Both isotopes have the same number of protons, but the difference in neutron number results in different masses.
B(i)
The charge on the sodium ion is less than the charge on the magnesium ion. A smaller ionic charge produces a weaker Coulombic attraction between \( \text{Na}^+\) and water molecules compared with \( \text{Mg}^{2+}\).
B(ii)
The \( \text{Na}^+\) ion is larger than the \( \text{Mg}^{2+}\) ion. Because the distance between the ion and the oxygen atom of water is greater, the Coulombic attraction is weaker.
C
\[ \text{pOH}=-\log(2.80\times10^{-4})=3.553 \] \[ \text{pH}=14-3.553=10.447 \]
D
\[ M_1V_1=M_2V_2 \] \[ M_2=\frac{(1.85\times10^{-3})(0.03500)}{0.03500+0.05000} \] \[ [\text{Mg}^{2+}]=7.62\times10^{-4}\ \text{M} \]
E(i)
\[ K_{sp}=[\text{Mg}^{2+}][\text{OH}^-]^2 \]
E(ii)
\[ Q=[\text{Mg}^{2+}][\text{OH}^-]^2 \] \[ Q=(7.62\times10^{-4})(1.65\times10^{-4})^2 \] \[ Q=2.07\times10^{-11} \]
E(iii)
Because \(Q>K_{sp}\), the ion concentrations exceed equilibrium values. Therefore a precipitate of \( \text{Mg(OH)}_2(s)\) will form.
F
The amount of undissolved \( \text{Mg(OH)}_2(s)\) will decrease. The \( \text{H}^+\) from \( \text{HNO}_3\) reacts with \( \text{OH}^-\), decreasing \( [\text{OH}^-]\). As a result, more \( \text{Mg(OH)}_2\) dissolves until equilibrium is re-established.
