AP Chemistry 7.3 Reaction Quotient and Equilibrium Constant - Exam Style questions - FRQs- New Syllabus
Question
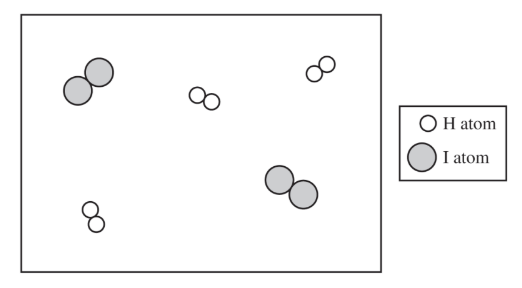
Most-appropriate topic codes (AP Chemistry):
• Topic \( 7.8 \) — Representations of Equilibrium (Part \( \mathrm{(b)(i)} \))
• Topic \( 7.10 \) — Reaction Quotient and Le Châtelier’s Principle (Parts \( \mathrm{(b)(ii)} \), \( \mathrm{(b)(iii)} \))
▶️ Answer/Explanation
(a)
For the correct expression:
\( K_c = \dfrac{[\mathrm{HI}]^2}{[\mathrm{H_2}][\mathrm{I_2}]} \)
The coefficient \( 2 \) in front of \( \mathrm{HI} \) becomes the exponent \( 2 \) in the equilibrium expression.
(b)(i)
For the correct drawing, consistent with part \( \mathrm{(a)} \):
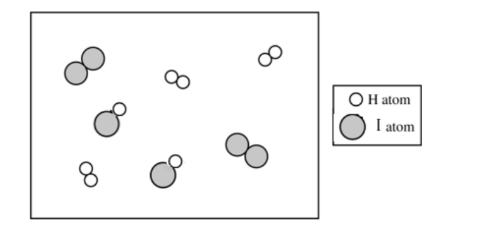
There are \( 2 \) molecules of \( \mathrm{H_2} \) and \( 2 \) molecules of \( \mathrm{I_2} \) shown.
Since \( Q = \dfrac{[\mathrm{HI}]^2}{[\mathrm{H_2}][\mathrm{I_2}]} = 0.67 \), we have \( [\mathrm{HI}]^2 \approx 0.67 \times (2)(2) = 2.68 \), so \( [\mathrm{HI}] \approx \sqrt{2.68} \approx 1.6 \).
Thus, the particulate diagram should show about \( 2 \) molecules of \( \mathrm{HI} \).
(b)(ii)
For a valid hypothesis:
Accept one of the following:
• Decreased the temperature.
• Added more \( \mathrm{H_2} \) and/or \( \mathrm{I_2} \) to the reaction vessel.
A small extra explanation: because the forward reaction is exothermic \(\left(\Delta H_{\mathrm{rxn}} < 0\right)\), decreasing the temperature shifts equilibrium toward products, so the number of moles of \( \mathrm{HI(g)} \) increases.
(b)(iii)
For the correct answer and a valid justification:
Accept one of the following:
• Remain unchanged. The number of moles in the numerator and denominator of \( Q \) (or \( K \)) are equal; changing the volume of the container would not alter the value of \( Q \), which is still equal to \( K \), so the number of moles of \( \mathrm{HI} \) will remain the same.
• Remain unchanged. The increase in volume will decrease the concentration of reactants and products by an equal proportion. Because there are equal moles of gaseous reactants and products in the balanced chemical equation, there is no shift in the equilibrium position, and the number of moles of \( \mathrm{HI} \) will remain the same.
In short, \( \Delta n_{\mathrm{gas}} = 2 – 2 = 0 \), so changing volume at constant temperature does not favor either side.
