Question
N2(g) + O2(g) ⇔2 NO(g)
At high temperatures, N2(g) and O2(g) can react to produce nitrogen monoxide, NO(g), as represented by the equation above.
(a) Write the expression for the equilibrium constant, Kp , for the forward reaction.
(b) A student injects N2(g) and O2(g) into a previously evacuated, rigid vessel and raises the temperature of the vessel to 2000°C. At this temperature the initial partial pressures of N2(g) and O2(g) are 6.01 atm and 1.61 atm, respectively. The system is allowed to reach equilibrium. The partial pressure of NO(g) at equilibrium is 0.122 atm. Calculate the value of Kp .
Nitrogen monoxide, NO(g), can undergo further reactions to produce acids such as HNO2 , a weak acid with a Ka of 4.0 × 10−4 and a pKa of 3.40.
(c) A student is asked to make a buffer solution with a pH of 3.40 by using 0.100 M HNO2(aq) and 0.100 M NaOH(aq).
(i) Explain why the addition of 0.100 M NaOH(aq) to 0.100 M HNO2(aq) can result in the formation of a buffer solution. Include the net ionic equation for the reaction that occurs when the student adds the NaOH(aq) to the HNO2(aq).
(ii) Determine the volume, in mL, of 0.100 M NaOH(aq) the student should add to 100. mL of 0.100 M HNO2(aq) to make a buffer solution with a pH of 3.40. Justify your answer.
(d) A second student makes a buffer by dissolving 0.100 mol of NaNO2(s) in 100. mL of 1.00 M HNO2(aq).
Which is more resistant to changes in pH when a strong acid or a strong base is added, the buffer made by the second student or the buffer made by the first student in part (c) ? Justify your answer.
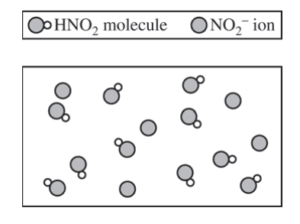
(e) A new buffer is made using HNO2(aq) as one of the ingredients. A particulate representation of a small representative portion of the buffer solution is shown below. (Cations and water molecules are not shown.) Is the pH of the buffer represented in the diagram greater than, less than, or equal to 3.40 ? Justify your answer.
▶️Answer/Explanation
Ans:
(a)
| \(K_{p}=\frac{(P_{NO})^{2}}{(P_{N_{2}})(P_{O_{2}})}\) |
(b)
N2(g) + O2(g) ⇔ 2 NO(g) 2x = 0.122 atm ⇒ x = 0.0610 atm \(K_{p}=\frac{(0.122)^{2}}{(5.95)(1.55)}=0.00161\) |
(c) (i)
NaOH will neutralize some of the HNO2 to produce NO2–. The resulting solution contains a mixture of a weak acid and its conjugate base, which is a buffer solution. HNO2 + OH– → NO2– + H2O |
(ii)
The student should add 50.0 mL of 0.100 M NaOH(aq). When half of the HNO2 is converted to the conjugate base, [HNO2] = [NO2– ], therefore the buffer has a pH equal to pKa. |
(d)
| The buffer made by the second student is more resistant to changes in pH because it contains a higher concentration of HNO2 and NO2– to react with added H+ or OH– ions. |
(e)
The pH of the solution is less than 3.40. If [HNO2] = [NO2– ], pH = pKa, and the pH of the solution would be 3.40. Since [HNO2] > [NO2– ], as represented in the diagram, the solution has a pH less than 3.40. |
