AP Chemistry 8.7 pH and pKa - Exam Style questions - FRQs- New Syllabus
Question
(ii) The mole ratio of carbon \( (\mathrm{C}) \) to oxygen \( (\mathrm{O}) \) is \(1:1\) in ascorbic acid. Based on this information and your answer to part \(A(i)\), determine the empirical formula of ascorbic acid.
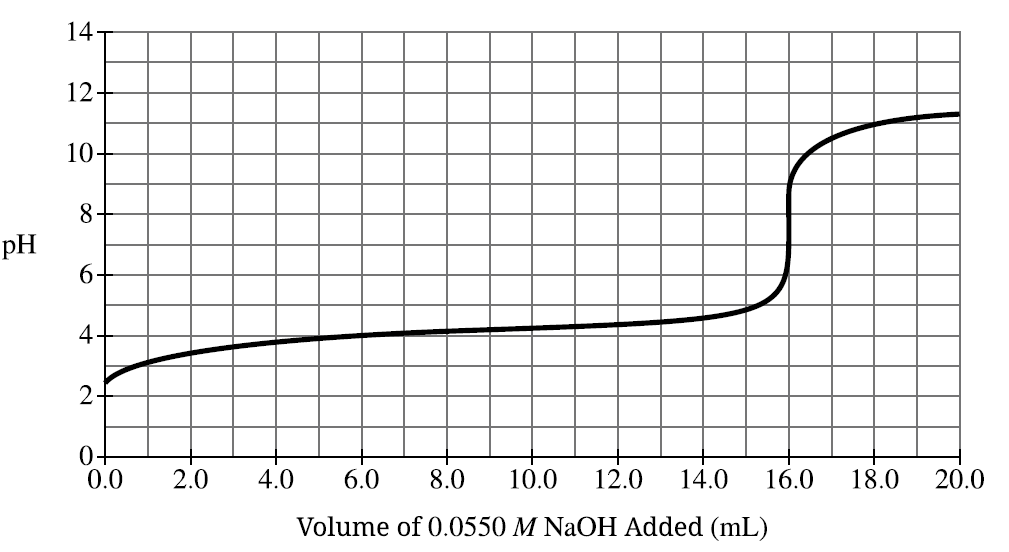
(ii) From the titration curve, determine the approximate \(pK_a\) of ascorbic acid.
(iii) What is the value of the ratio \( \frac{[\mathrm{Asc^-}]}{[\mathrm{HAsc}]} \) when the pH of the solution is \(4.7\)?
| Trial | \( [\mathrm{HAsc}] (M) \) | \( [\mathrm{I_3^-}] (M) \) | Initial Rate of \( \mathrm{DHAsc} \) Formation (M/s) |
|---|---|---|---|
| 1 | 0.450 | 1.200 | \(2.457\times10^{-4}\) |
| 2 | 0.450 | 0.600 | \(1.229\times10^{-4}\) |
| 3 | 0.900 | 1.200 | \(4.914\times10^{-4}\) |
(ii) Calculate the value of the rate constant, \(k\), for the reaction. Include units with your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic 4.6 — Introduction to Titration (moles and concentration from titration data) — Part B(i)
• Topic 8.7 — pH and \(pK_a\) (determining \(pK_a\) from titration curves) — Part B(ii)
• Topic 8.9 — Henderson–Hasselbalch Equation — Part B(iii)
• Topic 5.2 — Introduction to Rate Law (interpreting rate data) — Part C
• Topic 3.1 — Intermolecular and Interparticle Forces (ion–dipole interactions) — Part D
▶️ Answer/Explanation
A(i)
\(2.883\ \mathrm{g\ H_2O} \times \frac{1\ \mathrm{mol\ H_2O}}{18.02\ \mathrm{g\ H_2O}} = 0.1600\ \mathrm{mol\ H_2O}\)
A(ii)
Moles of carbon from \( \mathrm{CO_2} \):
\(0.2400\ \mathrm{mol\ CO_2} \times \frac{1\ \mathrm{mol\ C}}{1\ \mathrm{mol\ CO_2}} = 0.2400\ \mathrm{mol\ C}\)
Hydrogen from part A(i):
\(0.1600\ \mathrm{mol\ H_2O} \times \frac{2\ \mathrm{mol\ H}}{1\ \mathrm{mol\ H_2O}} = 0.3200\ \mathrm{mol\ H}\)
Given the mole ratio \( \mathrm{C:O} = 1:1 \):
\(0.2400\ \mathrm{mol\ O}\)
Ratio:
\(0.2400 : 0.3200 : 0.2400 = 3 : 4 : 3\)
Empirical formula:
\( \boxed{\mathrm{C_3H_4O_3}} \)
B(i)
Moles \( \mathrm{NaOH} \) at equivalence:
\(0.0160\ \mathrm{L} \times 0.0550\ \mathrm{M} = 8.80\times10^{-4}\ \mathrm{mol}\)
Moles \( \mathrm{HAsc} = 8.80\times10^{-4}\)
Volume \( = 0.0100\ \mathrm{L}\)
\([\mathrm{HAsc}] = \frac{8.80\times10^{-4}}{0.0100} = 0.0880\ \mathrm{M}\)
B(ii)
The \(pK_a\) occurs at the half-equivalence point.
From the graph this occurs at pH ≈ \(4.1\).
B(iii)
Henderson–Hasselbalch equation:
\( \mathrm{pH} = pK_a + \log \left(\frac{[\mathrm{Asc^-}]}{[\mathrm{HAsc}]}\right) \)
\(4.7 = 4.1 + \log \left(\frac{[\mathrm{Asc^-}]}{[\mathrm{HAsc}]}\right)\)
\(\log\left(\frac{[\mathrm{Asc^-}]}{[\mathrm{HAsc}]}\right) = 0.6\)
\(\frac{[\mathrm{Asc^-}]}{[\mathrm{HAsc}]} = 10^{0.6} = 4.0\)
C(i)
Compare trials \(1\) and \(3\):
\([\mathrm{HAsc}]\) doubles \(0.450 \rightarrow 0.900\)
Rate doubles \(2.457\times10^{-4} \rightarrow 4.914\times10^{-4}\)
Thus the reaction is first order with respect to \( \mathrm{HAsc} \).
C(ii)
Rate law:
\( \text{rate} = k[\mathrm{HAsc}][\mathrm{I_3^-}] \)
\( k = \frac{2.457\times10^{-4}}{(0.450)(1.200)} \)
\( k = 4.55\times10^{-4}\ \mathrm{M^{-1}\ s^{-1}} \)
D
Ion–dipole attractions occur between \( \mathrm{I_3^-} \) ions and water molecules but not between neutral \( \mathrm{I_2} \) molecules and water. Therefore \( \mathrm{I_3^-} \) is more soluble in water.
