AP Chemistry 8.8 Properties of Buffers - Exam Style questions - FRQs- New Syllabus
Question
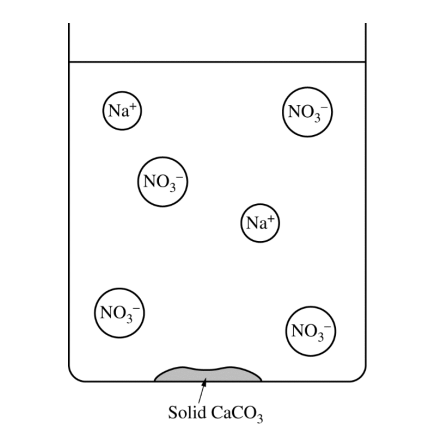
| Volume of \( \mathrm{Na_2CO_3} \) solution | \( 50.0\ \mathrm{mL} \) |
| Volume of \( 1.0\ \mathrm{M\ Ca(NO_3)_2} \) added | \( 100.0\ \mathrm{mL} \) |
| Mass of \( \mathrm{CaCO_3} \) precipitate collected | \( 0.93\ \mathrm{g} \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.8 \) — Representations of Solutions (Part \( \mathrm{(b)} \))
• Topic \( 4.5 \) — Stoichiometry (Part \( \mathrm{(c)} \), Part \( \mathrm{(d)} \))
• Topic \( 3.10 \) — Solubility (Part \( \mathrm{(a)} \), Part \( \mathrm{(b)} \), Part \( \mathrm{(e)} \))
• Topic \( 3.7 \) — Solutions and Mixtures (Part \( \mathrm{(e)} \))
• Topic \( 8.3 \) — Weak Acid and Base Equilibria (Part \( \mathrm{(f)} \), Part \( \mathrm{(g)} \))
• Topic \( 8.7 \) — \( \mathrm{pH} \) and \( \mathrm{p}K_a \) (Part \( \mathrm{(h)} \))
• Topic \( 8.8 \) — Properties of Buffers (Part \( \mathrm{(h)} \))
▶️ Answer/Explanation
(a)
\( \mathrm{Ca^{2+}}(aq) + \mathrm{CO_3^{2-}}(aq) \rightarrow \mathrm{CaCO_3}(s) \)
\( \mathrm{Na^+} \) and \( \mathrm{NO_3^-} \) are spectator ions, so they are not included in the net ionic equation.
(b)
The completed particulate diagram should show one \( \mathrm{Ca^{2+}} \) ion still present in the solution, along with the \( \mathrm{Na^+} \) and \( \mathrm{NO_3^-} \) ions already shown.
Because \( \mathrm{Ca(NO_3)_2} \) was added in excess, some \( \mathrm{Ca^{2+}} \) remains in the solution after all of the \( \mathrm{CO_3^{2-}} \) has precipitated as \( \mathrm{CaCO_3}(s) \).
(c)
First calculate moles of \( \mathrm{CaCO_3} \):
\( n = \dfrac{m}{M} = \dfrac{0.93\ \mathrm{g}}{100.1\ \mathrm{g/mol}} = 9.29 \times 10^{-3}\ \mathrm{mol} \)
Since the reaction ratio is \( 1:1 \), \( \mathrm{Na_2CO_3} : \mathrm{CaCO_3} = 1:1 \), the moles of \( \mathrm{Na_2CO_3} \) originally present are also \( 9.29 \times 10^{-3}\ \mathrm{mol} \).
To appropriate significant figures: \( 0.0093\ \mathrm{mol\ Na_2CO_3} \)
(d)
No, I do not agree.
If the precipitate was not completely dry, the measured mass would be greater than the actual mass of pure \( \mathrm{CaCO_3} \), because some of the mass would come from water.
That would make the calculated moles of \( \mathrm{CaCO_3} \) too large, which would also make the calculated moles and molarity of \( \mathrm{Na_2CO_3} \) too high, not too low.
(e)
The liquid would conduct electricity.
Even after the precipitate is removed, dissolved ions remain in the filtrate, including \( \mathrm{Na^+}(aq) \), \( \mathrm{NO_3^-}(aq) \), and excess \( \mathrm{Ca^{2+}}(aq) \). These mobile ions allow the solution to conduct an electric current.
(f)(i)
The student could determine the \( \mathrm{pH} \) of the solution using a \( \mathrm{pH} \) meter.
(f)(ii)
First, use the measured \( \mathrm{pH} \) to find \( \mathrm{pOH} \):
\( \mathrm{pOH} = 14 – \mathrm{pH} \)
Then determine \( [\mathrm{OH^-}] \):
\( [\mathrm{OH^-}] = 10^{-\mathrm{pOH}} \)
Next, use an ICE table for \( \mathrm{CO_3^{2-}}(aq) + \mathrm{H_2O}(l) \rightleftharpoons \mathrm{HCO_3^-}(aq) + \mathrm{OH^-}(aq) \). If the equilibrium concentration of \( \mathrm{OH^-} \) is \( x \), then the equilibrium concentration of \( \mathrm{HCO_3^-} \) is also \( x \), and the equilibrium concentration of \( \mathrm{CO_3^{2-}} \) is \( c_i – x \), where \( c_i \) is the initial concentration of \( \mathrm{CO_3^{2-}} \).
| \( \mathrm{CO_3^{2-}}(aq) \) | \( \mathrm{H_2O}(l) \) | \( \mathrm{HCO_3^-}(aq) \) | \( \mathrm{OH^-}(aq) \) | |
|---|---|---|---|---|
| I | \( c_i \) | — | \( 0 \) | \( 0 \) |
| C | \( -x \) | — | \( +x \) | \( +x \) |
| E | \( c_i – x \) | — | \( x \) | \( x \) |
Substitute into the expression for \( K_b \):
\( K_b = \dfrac{[\mathrm{HCO_3^-}][\mathrm{OH^-}]}{[\mathrm{CO_3^{2-}}]} = \dfrac{(x)(x)}{c_i – x} \)
Then solve for \( c_i \):
\( c_i = \dfrac{x^2}{K_b} + x \)
Thus, using the measured \( \mathrm{pH} \) value to find \( x = [\mathrm{OH^-}] \), the student can determine the initial concentration of \( \mathrm{CO_3^{2-}} \).
(g)
The concentration of \( \mathrm{HCO_3^-}(aq) \) is less than the concentration of \( \mathrm{CO_3^{2-}}(aq) \).
Because \( K_b = 2.1 \times 10^{-4} \) is small, the hydrolysis reaction is reactant-favored. Therefore, most of the dissolved species remains as \( \mathrm{CO_3^{2-}} \), and only a smaller amount is converted to \( \mathrm{HCO_3^-} \).
(h)
No, the \( \mathrm{Na_2CO_3} \) solution is not suitable for making a buffer with \( \mathrm{pH} = 6 \).
A buffer works best when the desired \( \mathrm{pH} \) is close to the \( \mathrm{p}K_a \) of the weak acid in the conjugate acid-base pair. Here, the weak acid is \( \mathrm{HCO_3^-} \), and its \( \mathrm{p}K_a \) is approximately \( 10.32 \).
Since \( 6 \) is far from \( 10.32 \), the \( \mathrm{CO_3^{2-}/HCO_3^-} \) pair would not function as an effective buffer at \( \mathrm{pH} = 6 \).
