AP Chemistry 9.8 Galvanic (Voltaic) and Electrolytic Cells- Exam Style questions - FRQs- New Syllabus
Question
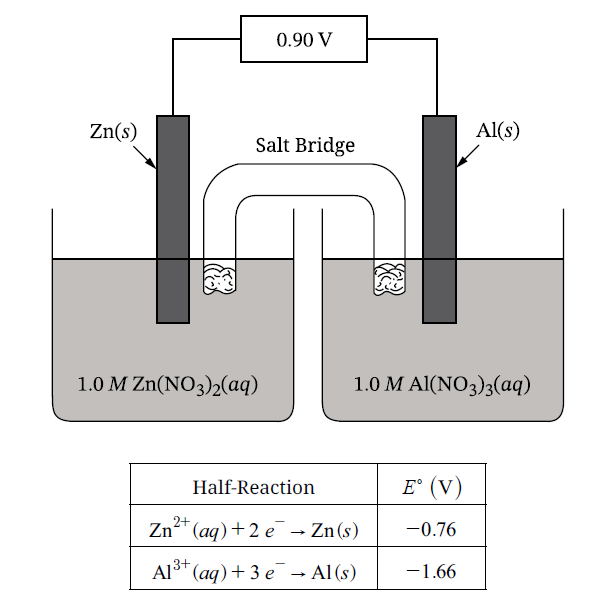
A. Write the half-reaction equation that occurs at the anode for the oxidation that occurs at the anode.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 4.2 \) — Net Ionic Equations — Part B
• Topic \( 9.8 \) — Galvanic (Voltaic) and Electrolytic Cells — Part C
• Topic \( 9.9 \) — Cell Potential and Free Energy — Part D
▶️ Answer/Explanation
A
For the correct equation (state symbols not required):
\( \mathrm{Al(s) \rightarrow Al^{3+}(aq) + 3\ e^-} \)
Because the anode is the site of oxidation, the species losing mass must be the one being oxidized.
Since the \( \mathrm{Al(s)} \) electrode decreases in mass, \( \mathrm{Al} \) is oxidized at the anode.
B
For the correct balanced net ionic equation (state symbols not required):
\( \mathrm{2\ Al(s) + 3\ Zn^{2+}(aq) \rightarrow 2\ Al^{3+}(aq) + 3\ Zn(s)} \)
The reduction half-reaction is \( \mathrm{Zn^{2+}(aq) + 2\ e^- \rightarrow Zn(s)} \).
Balancing electrons between the two half-reactions gives the overall net ionic equation above.
C
For the correct answer and a valid justification that correctly compares the masses of \( \mathrm{Al} \) and \( \mathrm{Zn} \) based on their molar masses and the stoichiometry of the balanced equation.
Examples of acceptable responses may include the following:
• \( \mathrm{Zn} \) experiences a greater change in mass. Assuming the entire \( \mathrm{Al} \) anode reacts:
\( 50.0\ \mathrm{g\ Al} \times \dfrac{1\ \mathrm{mol\ Al}}{26.98\ \mathrm{g\ Al}} \times \dfrac{3\ \mathrm{mol\ Zn}}{2\ \mathrm{mol\ Al}} \times \dfrac{65.38\ \mathrm{g\ Zn}}{1\ \mathrm{mol\ Zn}} = 182\ \mathrm{g\ Zn} \)
• \( \mathrm{Zn} \) experiences a greater change in mass.
\( 1\ \mathrm{mol_{rxn}} \times \dfrac{3\ \mathrm{mol\ Zn}}{1\ \mathrm{mol_{rxn}}} \times \dfrac{65.38\ \mathrm{g\ Zn}}{1\ \mathrm{mol\ Zn}} = 196.1\ \mathrm{g\ Zn} \)
\( 1\ \mathrm{mol_{rxn}} \times \dfrac{2\ \mathrm{mol\ Al}}{1\ \mathrm{mol_{rxn}}} \times \dfrac{26.98\ \mathrm{g\ Al}}{1\ \mathrm{mol\ Al}} = 53.96\ \mathrm{g\ Al} \)
Thus, for however many moles of reaction that proceed, the mass of \( \mathrm{Zn} \) produced will be greater than the mass of \( \mathrm{Al} \) consumed.
• \( \mathrm{Zn} \) experiences a greater change in mass. As the reaction proceeds, three moles of \( \mathrm{Zn} \) are used for every two moles of \( \mathrm{Al} \). Thus, for every \( 196\ \mathrm{g} \) of \( \mathrm{Zn} \) that are produced, \( 54\ \mathrm{g} \) of \( \mathrm{Al} \) are consumed.
So, the \( \mathrm{Zn} \) electrode experiences the greater mass change.
D
For the correct calculated value.
Examples of acceptable responses may include the following:
• \( E^\circ_{\mathrm{cell}} = 1.50\ \mathrm{V} + 0.76\ \mathrm{V} = 2.26\ \mathrm{V} \)
• \( \mathrm{Au^{3+}(aq) + 3\ e^- \rightarrow Au(s)} \qquad +1.50\ \mathrm{V} \)
\( \mathrm{Zn(s) \rightarrow Zn^{2+}(aq) + 2\ e^-} \qquad +0.76\ \mathrm{V} \)
\( E^\circ_{\mathrm{cell}} = 2.26\ \mathrm{V} \)
