AP Chemistry 4.2 Net-Ionic Equations Study Notes - New Syllabus Effective fall 2024
AP Chemistry 4.2 Net-Ionic Equations Study Notes- New syllabus
AP Chemistry 4.2 Net-Ionic Equations Study Notes – AP Chemistry – per latest AP Chemistry Syllabus.
LEARNING OBJECTIVE
Represent changes in matter with a balanced chemical or net ionic equation:
i. For physical changes.
ii. For given information about the identity of the reactants and/or product.
iii. For ions in a given chemical reaction.
Key Concepts:
- Symbolic Representation of Physical and Chemical Processes
- Conservation of Mass and Charge in Chemical Equations
- Symbolic Forms of Chemical Equations: Molecular, Complete Ionic, and Net Ionic Representations
Symbolic Representation of Physical and Chemical Processes
All physical and chemical processes can be represented symbolically using balanced equations that describe the transformation of matter. In these symbolic representations, chemical symbols and formulas are used to express substances, while coefficients indicate relative amounts involved in the process.
1. Representation of Processes: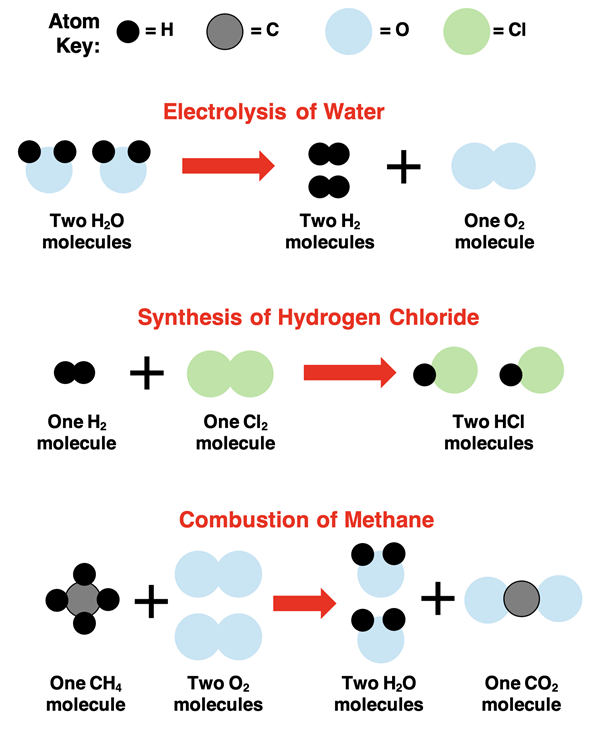
Physical Processes: Represented using phase symbols to indicate changes in state, but without changing chemical identity.
- Example: \(\mathrm{H_2O(s) \rightarrow H_2O(l)}\) — melting of ice (physical change).
- Example: \(\mathrm{NaCl(s) \rightarrow Na^+(aq) + Cl^-(aq)}\) — dissolving of salt in water (no new substance).
Chemical Processes: Represented by chemical equations that show the conversion of reactants into products via bond breaking and formation.
- Example: \(\mathrm{2H_2(g) + O_2(g) \rightarrow 2H_2O(l)}\) — chemical reaction producing water.
2. Components of a Symbolic Equation:
| Component | Description |
|---|---|
| Reactants | Substances present before the reaction or change |
| Products | Substances formed as a result of the process |
| Arrow (→) | Indicates the direction of change from reactants to products |
| Coefficients | Show the relative number of moles or particles of each substance |
| State Symbols | Indicate phase: (s), (l), (g), (aq) |
3. Purpose of Symbolic Representation:
- To concisely describe chemical or physical processes.
- To track the quantitative relationships between substances (via stoichiometry).
- To communicate information about energy changes, states, and reaction conditions (e.g., heat, catalysts, or solvents).
All physical and chemical processes can be expressed symbolically through equations that represent the rearrangement of matter — either through physical state changes or chemical transformations.
Example:
Represent the following processes symbolically:
- Evaporation of liquid water
- Combustion of methane gas in oxygen
▶️ Answer / Explanation
1. Physical Process: \(\mathrm{H_2O(l) \rightarrow H_2O(g)}\) — phase change from liquid to gas; composition remains \(\mathrm{H_2O}\).
2. Chemical Process: \(\mathrm{CH_4(g) + 2O_2(g) \rightarrow CO_2(g) + 2H_2O(g)}\) — new substances (carbon dioxide and water) are formed; bonds are broken and reformed.
Final Insight: Both processes are symbolically represented, but only the second involves a chemical transformation.
Conservation of Mass and Charge in Chemical Equations
A chemical equation represents a chemical change where atoms are rearranged into new combinations, forming new substances. Because atoms are neither created nor destroyed during chemical reactions, balanced chemical equations must show the same number of each type of atom and the same total electric charge on both sides of the equation.
1. Conservation of Mass:
Based on the Law of Conservation of Mass — the total mass of reactants equals the total mass of products.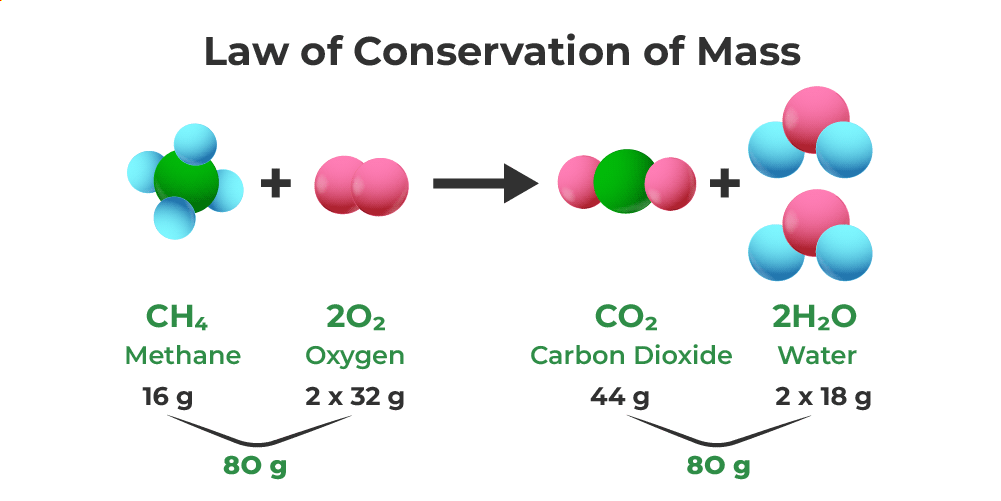
- Atoms are rearranged, not lost or created, during a chemical reaction.
- The coefficients in a balanced equation ensure that equal numbers of atoms of each element are present on both sides.
Example: \(\mathrm{2H_2(g) + O_2(g) \rightarrow 2H_2O(l)}\)
- Left: 4 H atoms, 2 O atoms
- Right: 4 H atoms, 2 O atoms
Mass is conserved.
2. Conservation of Charge:
In ionic and redox reactions, total charge must also be conserved.
- This means that the algebraic sum of charges on the reactant side must equal that on the product side.
- This principle ensures that no net charge is lost or gained in any reaction process.
Example: \(\mathrm{Zn(s) + Cu^{2+}(aq) \rightarrow Zn^{2+}(aq) + Cu(s)}\)
- Left total charge: \(0 + (+2) = +2\)
- Right total charge: \((+2) + 0 = +2\)
Charge is conserved.
3. Meaning of a Balanced Chemical Equation:
- Represents the stoichiometric relationship between reactants and products.
- Shows the relative numbers of particles (atoms, molecules, or ions) involved.
- Reflects both mass conservation and charge conservation.
A balanced equation is not merely symbolic it expresses the quantitative relationships required for stoichiometric calculations (mole ratios, limiting reactants, etc.).
4. Steps to Balance a Chemical Equation:
- Write correct formulas for all reactants and products.
- Count the number of each type of atom on both sides.
- Adjust coefficients (not subscripts) to balance one element at a time.
- Check that all elements and total charge are balanced.
- Verify that coefficients represent the simplest whole-number ratio.
Chemical equations represent the rearrangement of atoms not their creation or destruction. They must always demonstrate that mass and charge are conserved across the reaction.
Example:
Balance and verify conservation of mass and charge for the reaction between aluminum metal and copper(II) chloride solution.
Unbalanced equation: \(\mathrm{Al(s) + CuCl_2(aq) \rightarrow AlCl_3(aq) + Cu(s)}\)
▶️ Answer / Explanation
Step 1: Balance atoms.
- Aluminum: 2 on each side → \(\mathrm{2Al}\)
- Chlorine: 6 on each side → \(\mathrm{3CuCl_2}\)
- Copper: 3 on each side → \(\mathrm{3Cu}\)
Balanced equation: \(\mathrm{2Al(s) + 3CuCl_2(aq) \rightarrow 2AlCl_3(aq) + 3Cu(s)}\)
Step 2: Verify conservation.
- Atoms: 2 Al, 3 Cu, 6 Cl on each side
- Charge: Total 0 (neutral) on both sides
Final Answer: The balanced equation satisfies conservation of both mass and charge.
Symbolic Forms of Chemical Equations: Molecular, Complete Ionic, and Net Ionic Representations
Chemical equations can be written in different symbolic forms to represent reactions at varying levels of detail. The three main forms are the molecular equation, the complete ionic equation, and the net ionic equation. Each provides insight into how atoms and ions are rearranged during a chemical reaction.
1. Molecular Equation:
- Represents all reactants and products as neutral compounds or molecules, even if they exist as ions in solution.
- It shows the overall reaction but does not explicitly reveal the ionic nature of aqueous species.
- Purpose: Useful for general representation and stoichiometric calculations.
Example (Molecular Form): \(\mathrm{Na_2SO_4(aq) + BaCl_2(aq) \rightarrow 2NaCl(aq) + BaSO_4(s)}\)
Shows the overall double-replacement reaction but does not show ion dissociation.
2. Complete Ionic Equation:
- Represents all strong electrolytes (soluble ionic compounds, strong acids, and bases) as ions.
- Shows all species present in the solution before and after the reaction.
- Purpose: Helps visualize all particles that participate in or remain unchanged during the reaction.
Example (Complete Ionic Form):
\(\mathrm{2Na^+(aq) + SO_4^{2-}(aq) + Ba^{2+}(aq) + 2Cl^-(aq) \rightarrow 2Na^+(aq) + 2Cl^-(aq) + BaSO_4(s)}\)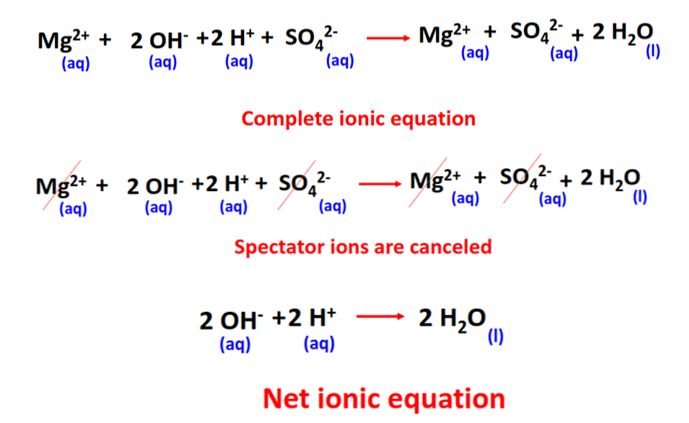
Now, all aqueous ionic species are explicitly shown as dissociated ions.
3. Net Ionic Equation:
- Shows only the species that actually participate in the chemical change.
- Spectator ions (ions that remain unchanged on both sides) are eliminated.
- Purpose: Emphasizes the chemical essence of the reaction, making it simpler and more meaningful.
Example (Net Ionic Form):
\(\mathrm{Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)}\)
This equation highlights that the formation of solid barium sulfate is the core chemical change.
4. Spectator Ions:
- Ions that do not participate in the chemical change but remain in solution throughout the process.
- In the above example, \(\mathrm{Na^+}\) and \(\mathrm{Cl^-}\) are spectator ions.
\(\mathrm{Na^+(aq)}\) and \(\mathrm{Cl^-(aq)}\) → present on both sides → canceled out in the net ionic equation.
| Type of Equation | Description | Includes Spectator Ions? |
|---|---|---|
| Molecular Equation | Shows all species as compounds (no ion dissociation) | Yes |
| Complete Ionic Equation | Shows all aqueous ionic species as dissociated ions | Yes |
| Net Ionic Equation | Shows only the species that undergo change | No |
Each type of equation communicates a different level of chemical detail. All balanced forms must obey the law of conservation of mass and charge, ensuring that atoms and charges are equal on both sides.
Example:
Represent the reaction between aqueous sodium carbonate and calcium chloride in all three symbolic forms.
▶️ Answer / Explanation
Step 1: Molecular Equation
\(\mathrm{Na_2CO_3(aq) + CaCl_2(aq) \rightarrow 2NaCl(aq) + CaCO_3(s)}\)
Step 2: Complete Ionic Equation
\(\mathrm{2Na^+(aq) + CO_3^{2-}(aq) + Ca^{2+}(aq) + 2Cl^-(aq) \rightarrow 2Na^+(aq) + 2Cl^-(aq) + CaCO_3(s)}\)
Step 3: Net Ionic Equation
\(\mathrm{Ca^{2+}(aq) + CO_3^{2-}(aq) \rightarrow CaCO_3(s)}\)
Step 4: Identify Spectator Ions
\(\mathrm{Na^+}\) and \(\mathrm{Cl^-}\) are unchanged → removed in the net ionic form.
Final Answer: The net ionic equation shows only the formation of insoluble calcium carbonate, highlighting the actual chemical change.
