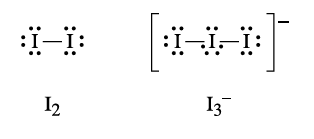Question 1
A. An incomplete mass spectrum for magnesium is shown in the diagram.
The percent abundance of magnesium‑24 is 79%. The percent abundances of the other two natural isotopes of magnesium, magnesium‑25 and magnesium‑26, are approximately equal.
i. Complete the mass spectrum in part A by drawing thick lines in the appropriate locations to represent the percent abundance of magnesium‑25 and magnesium‑26.
ii. Describe the difference in atomic structure that accounts for the difference in mass between magnesium‑25 and magnesium‑26.
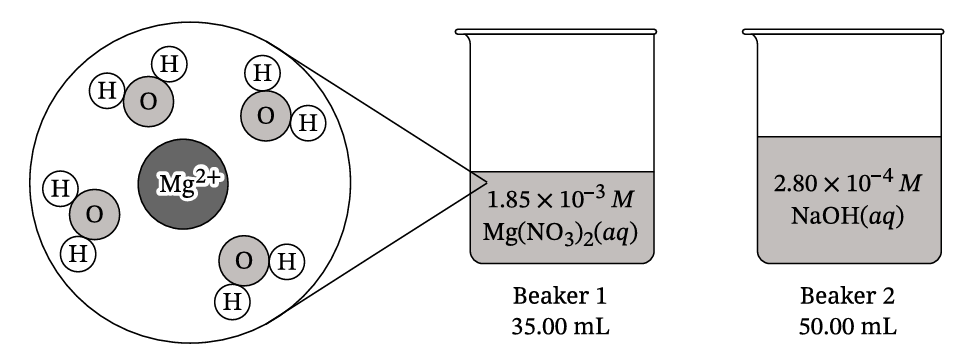
B. The particle diagram shown represents a magnesium ion, Mg²⁺, in beaker 1. A sodium ion, Na⁺, in beaker 2 has a weaker attraction to water than the Mg²⁺ does. Explain this phenomenon using Coulomb’s law and each of the following.
i. The relative charge of the ions
ii. The relative radii of the ions
C. Calculate the pH of the solution in beaker 2.
D. A student combines 35.00 mL of \(1.85 \times 10^{-3} \, M \, \text{Mg}(NO_3)_2(aq)\) with 50.00 mL of \(2.80 \times 10^{-4} \, M \, \text{NaOH}(aq)\). Calculate \(\left[ \text{Mg}^{2+} \right]\) after the two solutions are combined but before any reaction takes place. (Assume that volumes are additive.)
E. The dissolution of magnesium hydroxide is represented by the following equation.
\(\text{Mg(OH)}_2(s) = \text{Mg}^{2+}(aq) + 2 \, \text{OH}^- (aq) \quad K_{sp} = 5.61 \times 10^{-12}\)
i. Write the expression for the solubility product constant, \(K_{sp}\).
ii. After the two solutions are combined in beaker 3 as described in part D, but before any reaction takes place, \(\left[ \text{OH}^- \right] = 1.65 \times 10^{-4} \, M\). Using your answer to part D, calculate the value of the reaction quotient, Q.
iii. Using the reaction quotient, Q, predict whether a precipitate should form as the mixture in beaker 3 approaches equilibrium. Justify your answer.
F. In a separate experiment, the student adds \(HNO_3(aq)\) to decrease the pH of a saturated solution containing undissolved Mg(OH)₂(s). Does the amount of undissolved Mg(OH)₂(s) increase, decrease, or remain the same as the \(HNO_3(aq)\) is added? Justify your answer.
Most‑appropriate topic codes :
• SAP‑3: Coulomb’s law and electrostatic interactions in solutions — parts B(i), B(ii)
• SAP‑9: Solution stoichiometry and dilution calculations — part C
• SAP‑9: Solution stoichiometry and dilution calculations — part D
• SPQ‑4: The solubility product constant \(K_{sp}\) and the reaction quotient \(Q\) — parts E(i), E(ii), E(iii)
• SPQ‑5: Le Châtelier’s principle and equilibrium shifts — part F
▶️ Answer/Explanation
A(i)
Remaining abundance = 100% – 79% = 21%. Since Mg‑25 and Mg‑26 have approximately equal abundances, each contributes about 21%/2 = 10.5%. On the mass spectrum, draw vertical bars at mass 25 and mass 26 with heights corresponding to a relative abundance of about 10–11 (on the 0‑100 scale).
✅ Answer: \(\boxed{\text{Lines drawn at ≈10–11 abundance at masses 25 and 26.}}\)
A(ii)
Both isotopes have the same number of protons (12) and electrons. The mass difference arises from a different number of neutrons.
✅ Answer: \(\boxed{\text{Magnesium‑26 has one more neutron than magnesium‑25.}}\)
B(i)
Coulomb’s law states \(F \propto \frac{q_1 q_2}{r^2}\). Mg²⁺ has a charge of +2, while Na⁺ has a charge of +1. The larger charge on Mg²⁺ results in a stronger electrostatic attraction to the partial negative charge on the oxygen atom of water molecules.
✅ Answer: \(\boxed{\text{Mg}^{2+}\text{ has a larger charge than Na}^+, \text{ leading to a stronger Coulombic attraction to water.}}\)
B(ii)
Na⁺ has a larger ionic radius than Mg²⁺. According to Coulomb’s law, increasing the distance \(r\) between the ion and the water molecule decreases the attractive force. Thus, the larger Na⁺ has a weaker attraction to water.
✅ Answer: \(\boxed{\text{Na}^+ \text{ is larger than Mg}^{2+}, \text{ so the greater distance reduces the Coulombic attraction.}}\)
C
[OH⁻] = \(2.80 \times 10^{-4} \, M\).
pOH = \(- \log(2.80 \times 10^{-4}) = 3.553\).
pH = 14 – pOH = 14 – 3.553 = 10.447.
✅ Answer: \(\boxed{10.45 \ (\text{or } 10.447)}\)
D
Moles of Mg²⁺ = \((1.85 \times 10^{-3} \, \text{mol/L})(0.03500 \, \text{L}) = 6.475 \times 10^{-5} \, \text{mol}\).
Total volume after mixing = 35.00 mL + 50.00 mL = 85.00 mL = 0.08500 L.
\([Mg^{2+}] = \frac{6.475 \times 10^{-5} \, \text{mol}}{0.08500 \, \text{L}} = 7.62 \times 10^{-4} \, M\).
✅ Answer: \(\boxed{7.62 \times 10^{-4} \, M}\)
E(i)
\(K_{sp} = [Mg^{2+}][OH^-]^2\).
✅ Answer: \(\boxed{K_{sp} = [Mg^{2+}][OH^-]^2}\)
E(ii)
\([Mg^{2+}] = 7.62 \times 10^{-4} \, M\) (from part D). \([OH^-] = 1.65 \times 10^{-4} \, M\).
\(Q = [Mg^{2+}][OH^-]^2 = (7.62 \times 10^{-4})(1.65 \times 10^{-4})^2 = 2.07 \times 10^{-11}\).
✅ Answer: \(\boxed{2.07 \times 10^{-11}}\)
E(iii)
\(K_{sp} = 5.61 \times 10^{-12}\); \(Q = 2.07 \times 10^{-11}\).
Since \(Q > K_{sp}\), the ion product exceeds the solubility product, so the solution is supersaturated and a precipitate will form as the system shifts toward equilibrium.
✅ Answer: \(\boxed{\text{Precipitate will form because } Q > K_{sp}.}\)
F
Adding HNO₃ increases [H⁺], which reacts with OH⁻: \(H^+ + OH^- \rightarrow H_2O\). This decreases [OH⁻]. According to Le Châtelier’s principle, lowering [OH⁻] shifts the dissolution equilibrium \(\text{Mg(OH)}_2(s) \rightleftharpoons Mg^{2+}(aq) + 2OH^-(aq)\) to the right, dissolving more solid. Therefore, the amount of undissolved Mg(OH)₂(s) decreases.
✅ Answer: \(\boxed{\text{Decrease.}}\)
Question 2
| Trial | [HAsc] (M) | [\( I_3^-\)] (M) | Initial Rate of DHAsc Formation (M/s) |
|---|---|---|---|
| 1 | 0.450 | 1.200 | \( 2.457 \times 10^{-4} \) |
| 2 | 0.450 | 0.600 | \( 1.229 \times 10^{-4} \) |
| 3 | 0.900 | 1.200 | \( 4.914 \times 10^{-4} \) |
Most-appropriate topic codes :
• SPQ-2.C: Explain the relationship between mass, molecular weight, and number of particles — part A(i)
• TRA-1.A: Represent the law of conservation of mass quantitatively and qualitatively — part A(ii)
• SPQ-4.A: Identify the equivalence point in a titration based on the quantities of the titrant and analyte — part B(i)
• SAP-9.E: Explain results from the titration of a mono- or polyprotic acid or base solution — part B(i), B(ii)
• SAP-10.A: Explain the relationship between the predominant form of a weak acid or base in solution and the pH of the solution — part B(ii), B(iii)
• SAP-10.B: Explain the relationship between pH and pKₐ — part B(ii), B(iii)
• SAP-10.C: Use the Henderson-Hasselbalch equation to calculate pH or pKₐ — part B(iii)
• TRA-3.A: Explain the relationship between the rate of a chemical reaction and experimental parameters — part C(i), C(ii)
• TRA-3.B: Represent experimental data with a consistent rate law expression — part C(i), C(ii)
• SAP-5.A: Explain the relationship between the solubility of ionic and molecular compounds in water and the intermolecular interactions between particles — part D
▶️ Answer/Explanation
A(i)
To calculate moles of \( H_2O \): \( 2.883 \, \text{g} \, H_2O \times \frac{1 \, \text{mol} \, H_2O}{18.02 \, \text{g} \, H_2O} = 0.1600 \, \text{mol} \, H_2O \) ✅ Answer: \( \boxed{0.1600 \, \text{mol}} \)
A(ii)
Step 1: Calculate moles of C from \( CO_2 \): \( 0.2400 \, \text{mol} \, CO_2 \times \frac{1 \, \text{mol} \, C}{1 \, \text{mol} \, CO_2} = 0.2400 \, \text{mol} \, C \) Step 2: Calculate moles of H from \( H_2O \): \( 0.1600 \, \text{mol} \, H_2O \times \frac{2 \, \text{mol} \, H}{1 \, \text{mol} \, H_2O} = 0.3200 \, \text{mol} \, H \) Step 3: Given C:O ratio is 1:1, so moles of O = moles of C = 0.2400 mol Step 4: Find simplest ratio: C: \( 0.2400/0.2400 = 1 \)
H: \( 0.3200/0.2400 = 1.333 \) (×3 = 4)
O: \( 0.2400/0.2400 = 1 \) (×3 = 3)
Empirical formula: \( C_3H_4O_3 \) ✅ Answer: \( \boxed{C_3H_4O_3} \)
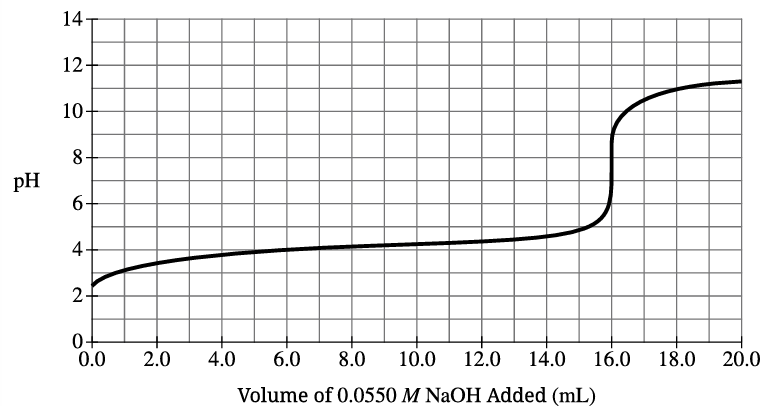
B(i)
From titration curve, equivalence point is at 16.0 mL NaOH. Step 1: Calculate moles of NaOH: \( 0.0160 \, \text{L} \times 0.0550 \, \text{M} = 8.80 \times 10^{-4} \, \text{mol} \, \text{NaOH} \) Step 2: At equivalence point, moles HAsc = moles NaOH: \( 8.80 \times 10^{-4} \, \text{mol} \, \text{HAsc} \) Step 3: Calculate concentration: \( \frac{8.80 \times 10^{-4} \, \text{mol}}{0.0100 \, \text{L}} = 0.0880 \, \text{M} \) ✅ Answer: \( \boxed{0.0880 \, \text{M}} \)
B(ii)
From titration curve, half-equivalence point is at 8.0 mL where pH = pKₐ. Reading from graph at 8.0 mL, pH ≈ 4.1 ✅ Answer: \( \boxed{4.1} \) (acceptable range: 4.0–4.3)
B(iii)
Using Henderson-Hasselbalch equation: \( \text{pH} = pK_a + \log\left(\frac{[\text{Asc}^-]}{[H\text{Asc}]}\right) \) \( 4.7 = 4.1 + \log\left(\frac{[\text{Asc}^-]}{[H\text{Asc}]}\right) \) \( \log\left(\frac{[\text{Asc}^-]}{[H\text{Asc}]}\right) = 0.6 \) \( \frac{[\text{Asc}^-]}{[H\text{Asc}]} = 10^{0.6} = 4.0 \) ✅ Answer: \( \boxed{4.0} \)
C(i)
Comparing Trials 1 and 3: [HAsc] doubles from 0.450 M to 0.900 M while [\( I_3^-\)] remains constant at 1.200 M. The rate doubles from \( 2.457 \times 10^{-4} \, \text{M/s} \) to \( 4.914 \times 10^{-4} \, \text{M/s} \). This direct proportionality shows the reaction is first order with respect to [HAsc].
✅ Answer: When [HAsc] doubles with constant [ I 3 − I 3 − ], the rate doubles, indicating first order dependence.
C(ii)
Using the rate law \( rate = k[HAsc][I_3^-] \) and Trial 1 data: \( k = \frac{rate}{[HAsc][I_3^-]} = \frac{2.457 \times 10^{-4} \, \text{M/s}}{(0.450 \, \text{M})(1.200 \, \text{M})} = 4.55 \times 10^{-4} \, \text{M}^{-1}\text{s}^{-1} \)
✅ Answer: \(4.55 × 10^ {−4} M ^{−1} s^{−1} \)
D
\( I_3^- \) is an ion while \( I_2 \) is a nonpolar molecule. \( I_3^- \) can form ion-dipole interactions with water molecules, where the charged ion attracts the partial charges on water molecules. \( I_2 \) can only form weaker London dispersion forces with water. ✅ Answer: Ion-dipole interactions Ion-dipole interactions
Question 3
B. The reaction of white phosphorus with oxygen to form \( P_4O_{10}(s) \) is represented below:
ii. A student claims that the thermodynamic favorability of this reaction is driven by enthalpy and not by entropy. Is the student’s claim correct? Justify your answer using the relationship between \( \Delta G^\circ \), \( \Delta H^\circ \), and \( \Delta S^\circ \).
C. \( P_4O_{10}(s) \) reacts exothermically with water: \( P_4O_{10}(s) + 6 H_2O(l) \rightarrow 4 H_3PO_4(aq) \). A chemist performs a calorimetry experiment with the following data:
| Mass of \( P_4O_{10} \) | 0.100 g |
|---|---|
| Mass of \( H_2O \) | 100.0 g |
| Initial Temperature | 22.00 °C |
| Final Temperature | 22.38 °C |
| Molar Mass of \( P_4O_{10} \) | 283.9 g/mol |
| Specific Heat of \( H_2O \) | 4.18 J/(g·°C) |
ii. Calculate the value of \( \Delta H_{rxn}^\circ \) for the reaction in kJ/mol\(_{rxn} \). Include the sign in your answer.
E. White phosphorus also reacts with chlorine. Use the equations below and Hess’s Law to calculate the standard enthalpy of formation of \( PCl_5(g) \):
(4) \( PCl_3(g) + Cl_2(g) \rightleftharpoons PCl_5(g) \) \( \Delta H^\circ = -88 \text{ kJ/mol}_{rxn} \)
(5) \( \frac{1}{4} P_4(s) + \frac{5}{2} Cl_2(g) \rightarrow PCl_5(g) \) \( \Delta H_f^\circ = ? \)
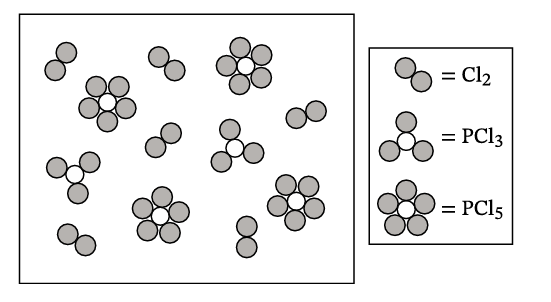
F. The equilibrium for reaction (4) is studied. The following particle diagram represents the contents of a vessel at equilibrium at 546 K (each particle represents a partial pressure of 1.00 atm).

ii. Does the value of \( K_p \) increase, decrease, or remain the same when the temperature is increased to 596 K? Justify your answer based on the sign of \( \Delta H^\circ \) for reaction (4).
Most-appropriate topic codes :
• Topic 5.1: Reaction Rates — Not applicable to this question
• Topic 7.9: Introduction to Entropy — Part B(i)
• Topic 9.2: Absolute Entropy and Entropy Change — Part B(i)
• Topic 9.3: Gibbs Free Energy and Thermodynamic Favorability — Part B(ii)
• Topic 6.5: Energy of Phase Changes — Not applicable to this question
• Topic 6.6: Introduction to Enthalpy of Reaction — Part C
• Topic 6.9: Hess’s Law — Part E
• Topic 7.7: Calculating Equilibrium Concentrations — Part F(i)
• Topic 7.10: Reaction Quotient and Equilibrium Constant — Part F(i)
• Topic 7.13: Le Châtelier’s Principle — Part F(ii)
▶️ Answer/Explanation
A.
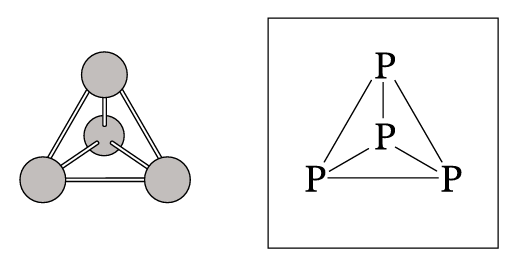
The Lewis structure for \( P_4 \) is a tetrahedron where each P atom is single-bonded to three others. Each P atom has five valence electrons. After forming three single bonds (using 3 electrons), each P atom has two nonbonding (lone pair) electrons remaining.
✅ Answer: Two lone pair electrons (:) should be drawn on each P atom in the diagram.
B(i).
The reaction consumes 5 moles of gaseous \( O_2 \) and produces zero moles of gas. Gaseous molecules have significantly higher entropy (more microstates, greater dispersal of energy) than solid molecules. The net loss of gas molecules causes a decrease in the disorder (entropy) of the system.
✅ Answer: \(\boxed{\text{The reaction converts gaseous reactants into a solid product. Solids have far fewer microstates and less dispersal than gases, so the system’s entropy decreases.}}\)
B(ii).
Yes, the student’s claim is correct.
The relationship is \( \Delta G^\circ = \Delta H^\circ – T\Delta S^\circ \). For a reaction to be thermodynamically favorable (spontaneous) at 298 K, \( \Delta G^\circ < 0 \).
• \( \Delta H^\circ < 0 \) (exothermic) contributes negatively to \( \Delta G^\circ \), favoring the reaction.
• \( \Delta S^\circ < 0 \) contributes positively to \( \Delta G^\circ \) (since \( -T\Delta S^\circ > 0 \)), opposing the reaction.
Therefore, the favorability must be driven by the negative \( \Delta H^\circ \)** overcoming the unfavorable negative \( \Delta S^\circ \).
✅ Answer: \(\boxed{\text{Yes. The reaction is exothermic (\(\Delta H^\circ < 0\)), which favors spontaneity, while the negative \(\Delta S^\circ\) disfavors it. Since \(\Delta G^\circ\) is negative overall, the favorability is driven by enthalpy.}}\)
C(i).
The heat absorbed by the water (and solution) is equal to the heat released by the reaction (\( q_{rxn} = -q_{surr} \)). Assume the specific heat of the solution is that of water.
\[ \begin{aligned} q_{surr} &= m \cdot c \cdot \Delta T \\ &= (100.0 \, \text{g}) \times (4.18 \, \text{J g}^{-1} \text{°C}^{-1}) \times (22.38 – 22.00 \, \text{°C}) \\ &= (100.0) \times (4.18) \times (0.38) \\ &= 158.84 \, \text{J} \\ &= 0.15884 \, \text{kJ} \end{aligned} \]
Rounded to two significant figures (limited by \( \Delta T = 0.38 \, \text{°C} \), which has two sig figs):
✅ Answer: \(\boxed{q = 0.16 \, \text{kJ}}\)
C(ii).
First, find moles of \( P_4O_{10} \):
\[ \text{moles of } P_4O_{10} = \frac{0.100 \, \text{g}}{283.9 \, \text{g/mol}} = 3.522 \times 10^{-4} \, \text{mol} \]
The heat calculated in (i) was released by this amount of reactant. The enthalpy change is the heat released per mole of reaction as written.
\[ \begin{aligned} \Delta H_{rxn}^\circ &= \frac{-q}{\text{mol } P_4O_{10}} = \frac{-0.15884 \, \text{kJ}}{3.522 \times 10^{-4} \, \text{mol}} \\ &= -451 \, \text{kJ/mol}_{rxn} \end{aligned} \]
Rounded to two significant figures:
✅ Answer: \(\boxed{\Delta H_{rxn}^\circ = -4.5 \times 10^2 \, \text{kJ/mol}_{rxn}}\) (The negative sign is required.)
D.
Less than. Since \( P_4O_{10} \) is the limiting reactant, the amount of heat released in the reaction is directly proportional to the amount of \( P_4O_{10} \) that reacts. If less \( P_4O_{10} \) is transferred into the calorimeter, less heat will be released. With the same mass of water, a smaller release of heat will result in a smaller temperature increase (\( \Delta T \)).
✅ Answer: \(\boxed{\text{Less than. Less } P_4O_{10} \text{ results in less heat released, causing a smaller } \Delta T.}\)
E.
Use Hess’s Law to combine equations (3) and (4) to yield target equation (5).
1. Multiply equation (3) by \( \frac{1}{4} \) to get \( \frac{1}{4}P_4(s) + \frac{3}{2}Cl_2(g) \rightarrow PCl_3(g) \) with \( \Delta H^\circ = \frac{1}{4}(-1148) = -287 \, \text{kJ} \).
2. Write equation (4) as is: \( PCl_3(g) + Cl_2(g) \rightarrow PCl_5(g) \) with \( \Delta H^\circ = -88 \, \text{kJ} \).
3. Add the two manipulated equations: \( PCl_3(g) \) cancels, and \( \frac{3}{2}Cl_2 + Cl_2 = \frac{5}{2}Cl_2 \).
\[ \begin{aligned} \Delta H_f^\circ (\text{for eq. 5}) &= \frac{1}{4}(-1148) + (-88) \\ &= (-287) + (-88) \\ &= -375 \, \text{kJ/mol} \end{aligned} \]
✅ Answer: \(\boxed{\Delta H_f^\circ = -375 \, \text{kJ/mol}}\)
F(i).
From the diagram:
• Particles of \( PCl_5 \): 4 → Partial pressure \( P_{PCl_5} = 4.00 \, \text{atm} \)
• Particles of \( PCl_3 \): 2 → Partial pressure \( P_{PCl_3} = 2.00 \, \text{atm} \)
• Particles of \( Cl_2 \): 6 → Partial pressure \( P_{Cl_2} = 6.00 \, \text{atm} \)
For \( PCl_3(g) + Cl_2(g) \rightleftharpoons PCl_5(g) \):
\[ K_p = \frac{P_{PCl_5}}{P_{PCl_3} \cdot P_{Cl_2}} = \frac{4.00}{(2.00)(6.00)} = \frac{4.00}{12.00} = \frac{1}{3} \]
✅ Answer: \(\boxed{K_p = 0.333}\)
F(ii).
Decrease.
For reaction (4), \( \Delta H^\circ = -88 \, \text{kJ/mol} \), which is exothermic. According to Le Châtelier’s principle, increasing the temperature favors the endothermic direction (the reverse reaction, which absorbs heat). Therefore, at a higher temperature, the equilibrium shifts toward the reactants (\( PCl_3 \) and \( Cl_2 \)), decreasing the value of \( K_p \).
✅ Answer: \(\boxed{\text{Decrease. The reaction is exothermic (\(\Delta H^\circ < 0\)), so increasing temperature shifts equilibrium toward reactants, decreasing \(K_p\).}}\)
Question 4
C.
| Substance | Melting Point (K) | Boiling Point (K) | Enthalpy of Vaporization (kJ/mol) |
|---|---|---|---|
| CH3OH | 176 | 338 | 37.6 |
| H2CO | 181 | 254 | 24.2 |
ii. The scientist analyzes the mixture after it is cooled and determines that 8.59 g of CH3OH(l) is present. Calculate the amount of thermal energy, in kJ, that was removed to condense the 8.59 g of CH3OH at its boiling point.
Most-appropriate topic codes :
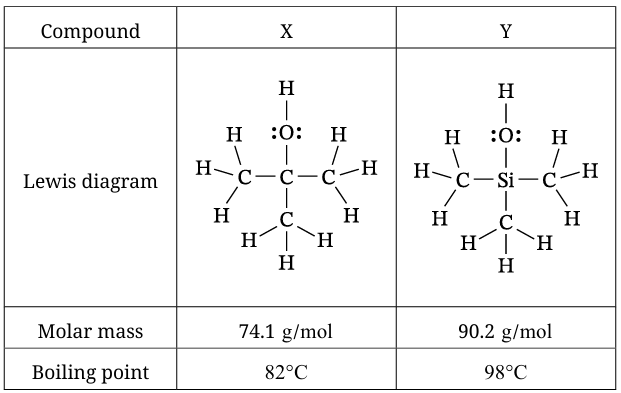
SAP-4.C: Represent a molecule with a Lewis diagram. — Part A & B (molecular structure)
SAP-8.A: Explain the relationship between the chemical structures of molecules and the relative strength of their intermolecular forces. — Part B
SAP-8.B: Explain the relationship among the macroscopic properties of a substance, the particulate-level structure of the substance, and the interactions between these particles. — Part C(i) & C(ii)
ENE-2.B: Calculate the heat (q) absorbed or released by a system undergoing heating/cooling based on the amount of the substance, the heat capacity, and the change in temperature. — Part C(ii)
ENE-3.A: Explain changes in the heat q absorbed or released by a system undergoing a phase transition. — Part C(ii)
▶️ Answer/Explanation
(a)
The carbon atom in H2CO has three electron domains (two single bonds to hydrogen and one double bond to oxygen). Three electron domains correspond to \( sp^2 \) hybridization.
✅ Answer: \(\boxed{sp^2}\)
(b)
Hydrogen bonding occurs between a hydrogen atom bonded to an electronegative atom (O, N, or F) and a lone pair on another electronegative atom. In this mixture, the strongest hydrogen bond forms between the hydrogen atom of the -OH group in CH3OH and the oxygen atom in H2CO. A dashed line should connect the H (from CH3OH’s OH) to the O (in H2CO).
(c i)
Data from table:
• CH3OH: Melting Point = 176 K, Boiling Point = 338 K
• H2CO: Melting Point = 181 K, Boiling Point = 254 K
Method: For both substances to be liquids, the temperature must be above their melting points and below their boiling points.
• Minimum temperature: must be above the higher melting point (181 K for H2CO)
• Maximum temperature: must be below the lower boiling point (254 K for H2CO)
Therefore, the temperature range is between 181 K and 254 K.
✅ Answer: \(\boxed{\text{Any temperature between 181 K and 254 K (e.g., 200 K, 215 K, 240 K)}}\)
(c ii)
Given:
• Mass of CH3OH = 8.59 g
• Molar mass of CH3OH = 32.04 g/mol
• Enthalpy of vaporization, \(\Delta H_{\text{vap}}\) = 37.6 kJ/mol (negative for condensation)
Step 1: Calculate moles of CH3OH
\( n = \frac{\text{mass}}{\text{molar mass}} = \frac{8.59 \text{ g}}{32.04 \text{ g/mol}} \)
\( n = 0.268 \text{ mol} \)
Step 2: Calculate heat removed during condensation
During condensation, heat is released (exothermic), so \(\Delta H\) is negative.
\( q = n \times (-\Delta H_{\text{vap}}) \)
\( q = 0.268 \text{ mol} \times (-37.6 \text{ kJ/mol}) \)
\( q = -10.1 \text{ kJ} \)
The negative sign indicates heat is removed from the system.
✅ Answer: \(\boxed{10.1 \text{ kJ}}\) removed
Question 5
Most‑appropriate topic codes :
• 3.1: Intermolecular Forces — part (b)
• 3.3: Solids, Liquids, and Gases — part (c)
• 3.4: Ideal Gas Law — part (d)
▶️ Answer/Explanation
(a)
In compound Y, the Si atom is bonded to four atoms (three H atoms and one O atom) and has no lone pairs. According to VSEPR theory, four bonding domains with zero lone pairs produce a tetrahedral electron‑pair geometry and molecular geometry.
✅ Answer: \(\boxed{\text{Tetrahedral}}\)
(b)
Agree. Both compounds are nonpolar and experience only London dispersion forces (LDF). LDF strength increases with the size of the electron cloud. Si has more occupied electron shells than C, so the electron cloud in compound Y is larger and more polarizable than in compound X. This results in stronger London dispersion forces and a higher boiling point for compound Y.
✅ Scoring: The point is earned for agreeing and justifying based on larger/more polarizable electron cloud due to more electron shells on Si.
(c)
Compound X will have the higher vapor pressure at 82 °C. At this temperature, compound X is at its boiling point (82 °C), so a significant fraction of its molecules are in the vapor phase. Compound Y has a boiling point of 98 °C, so at 82 °C it is still predominantly liquid. Because vapor pressure depends on the fraction of molecules in the vapor phase, compound X will have the higher vapor pressure.
Alternative valid justification: Compound X has weaker intermolecular forces (smaller LDF) than compound Y, so molecules escape more easily into the vapor phase at any given temperature, leading to a higher vapor pressure.
✅ Scoring: The point is earned for correctly choosing compound X and providing a valid justification based on relative boiling points or relative IMF strengths.
(d)
Use the ideal gas law: \(PV = nRT\).
Given:
• \(P = 2.30\ \text{atm}\)
• \(V = 12.5\ \text{L}\)
• \(T = 198^\circ\text{C} = 198 + 273 = 471\ \text{K}\)
• \(R = 0.08206\ \frac{\text{L·atm}}{\text{K·mol}}\)
\[ n = \frac{PV}{RT} = \frac{(2.30)(12.5)}{(0.08206)(471)} \]
\[ n = \frac{28.75}{38.63} \approx 0.744\ \text{mol} \]
✅ Answer: \(\boxed{0.744\ \text{mol}}\) (to three significant figures)
Question 6
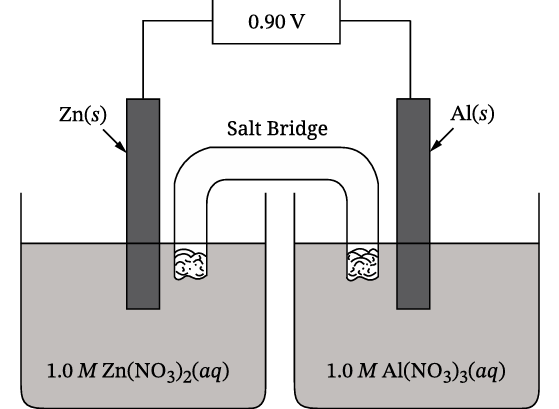
| Reduction Half-Reaction | \( E^{\circ} \) (V) |
|---|---|
| \( \text{Zn}^{2+}(aq) + 2 e^{-} \rightarrow \text{Zn}(s) \) | −0.76 |
| \( \text{Al}^{3+}(aq) + 3 e^{-} \rightarrow \text{Al}(s) \) | −1.66 |
| Reduction Half-Reaction | \( E^{\circ} \) (V) |
|---|---|
| \( \text{Au}^{3+}(aq) + 3 e^{-} \rightarrow \text{Au}(s) \) | +1.50 |
| \( \text{Zn}^{2+}(aq) + 2 e^{-} \rightarrow \text{Zn}(s) \) | −0.76 |
| \( \text{Mn}^{2+}(aq) + 2 e^{-} \rightarrow \text{Mn}(s) \) | −1.19 |
| \( \text{Al}^{3+}(aq) + 3 e^{-} \rightarrow \text{Al}(s) \) | −1.66 |
| \( \text{Be}^{2+}(aq) + 2 e^{-} \rightarrow \text{Be}(s) \) | −1.85 |
Corrected Topic Codes:
• ENE-6.A.2: Oxidation occurs at the anode, and reduction occurs at the cathode. The half-reactions are added to give the overall cell reaction. — Parts (A) and (B).
• ENE-6.A.3: In a galvanic cell, the anode is negatively charged and the cathode is positively charged. Electrons flow from the anode to the cathode through an external circuit. — Implicit in cell operation described.
• ENE-6.B.1: The standard cell potential (\(E^{\circ}_{cell}\)) is the difference between the standard reduction potentials of the cathode and anode half-reactions: \(E^{\circ}_{cell} = E^{\circ}_{cathode} – E^{\circ}_{anode}\). — Part (D).
• ENE-6.B.2: The standard reduction potential of a half-reaction is measured relative to the standard hydrogen electrode (SHE). — Basis for the table values.
• ENE-6.B.3: The standard cell potential is positive for a galvanic cell and indicates the spontaneity of the redox reaction. — Implicit in seeking a “greater voltage”.
• TRA-1.B.1: The mass of substances in a redox reaction can be related using stoichiometry and the mole concept. — Part (C) calculation.
▶️ Answer/Explanation
(A)
The anode is where oxidation (loss of electrons) occurs. Since the \( \text{Al}(s) \) electrode decreases in mass, \( \text{Al} \) is being oxidized to \( \text{Al}^{3+} \). The oxidation half-reaction is the reverse of the given reduction half-reaction for \( \text{Al} \).
✅ Answer: \( \boxed{\text{Al}(s) \rightarrow \text{Al}^{3+}(aq) + 3 e^{-}} \)
(B)
The overall reaction combines the oxidation of \( \text{Al} \) and the reduction of \( \text{Zn}^{2+} \). To balance the electrons, multiply the \( \text{Al} \) half-reaction by 2 and the \( \text{Zn}^{2+} \) half-reaction by 3, then add.
Oxidation (×2): \( 2\text{Al}(s) \rightarrow 2\text{Al}^{3+}(aq) + 6 e^{-} \)
Reduction (×3): \( 3\text{Zn}^{2+}(aq) + 6 e^{-} \rightarrow 3\text{Zn}(s) \)
Net ionic equation: \( 2\text{Al}(s) + 3\text{Zn}^{2+}(aq) \rightarrow 2\text{Al}^{3+}(aq) + 3\text{Zn}(s) \)
✅ Answer: \( \boxed{2\text{Al}(s) + 3\text{Zn}^{2+}(aq) \rightarrow 2\text{Al}^{3+}(aq) + 3\text{Zn}(s)} \)
(C)
From the balanced equation, 2 mol of \( \text{Al} \) are consumed while 3 mol of \( \text{Zn} \) are deposited. Using molar masses (\( \text{Al} = 26.98 \ \text{g/mol} \), \( \text{Zn} = 65.38 \ \text{g/mol} \)):
Mass change per mole of reaction (based on 2 mol \( \text{Al} \) and 3 mol \( \text{Zn} \)):
Mass of \( \text{Al} \) lost: \( 2 \times 26.98 = 53.96 \ \text{g} \)
Mass of \( \text{Zn} \) gained: \( 3 \times 65.38 = 196.14 \ \text{g} \)
Thus, for the same extent of reaction (same moles of electrons transferred), the mass gain of the \( \text{Zn} \) electrode is greater than the mass loss of the \( \text{Al} \) electrode. Even if only part of the 50.0 g \( \text{Al} \) reacts, the stoichiometric ratio ensures the \( \text{Zn} \) electrode’s mass changes more.
Calculation example: Assuming all 50.0 g \( \text{Al} \) reacts:
Moles of \( \text{Al} \): \( \frac{50.0}{26.98} = 1.853 \ \text{mol} \)
Moles of \( \text{Zn} \) produced: \( 1.853 \ \text{mol Al} \times \frac{3 \ \text{mol Zn}}{2 \ \text{mol Al}} = 2.780 \ \text{mol Zn} \)
Mass of \( \text{Zn} \) produced: \( 2.780 \times 65.38 \approx 182 \ \text{g} \) (gain)
Mass of \( \text{Al} \) lost: 50.0 g (loss)
The gain of ~182 g for \( \text{Zn} \) is larger than the loss of 50.0 g for \( \text{Al} \).
✅ Answer: The \( \boxed{\text{Zn electrode}} \)’s mass changed the most, because the mass of Zn deposited per mole of electrons transferred is greater than the mass of Al lost.
(D)
To maximize \( E_{cell}^{\circ} \), pair the half-cell with the most positive reduction potential (cathode) with the Zn half-cell as the anode (oxidation). Zn must be oxidized: \( \text{Zn}(s) \rightarrow \text{Zn}^{2+}(aq) + 2 e^{-} \), \( E^{\circ}_{\text{ox}} = +0.76 \ \text{V} \).
From the table, the cathode with the highest reduction potential is \( \text{Au}^{3+}/\text{Au} \) (\( E^{\circ}_{\text{red}} = +1.50 \ \text{V} \)).
\( E_{cell}^{\circ} = E^{\circ}_{\text{cathode}} – E^{\circ}_{\text{anode}} = E^{\circ}_{\text{red}}(\text{cathode}) – E^{\circ}_{\text{red}}(\text{anode}) \)
For Zn anode: \( E^{\circ}_{\text{red}}(\text{Zn}^{2+}/\text{Zn}) = -0.76 \ \text{V} \).
For Au cathode: \( E^{\circ}_{\text{red}}(\text{Au}^{3+}/\text{Au}) = +1.50 \ \text{V} \).
\( E_{cell}^{\circ} = 1.50 – (-0.76) = 2.26 \ \text{V} \).
Checking other options (e.g., Be with \( E^{\circ}_{\text{red}} = -1.85 \ \text{V} \)) gives lower voltages. Thus, the maximum voltage is 2.26 V.
✅ Answer: \( \boxed{2.26 \ \text{V}} \)
Question 7
Most-appropriate topic codes :
• Topic 6.9: pH and pKa — Part B
• Topic 5.7: Introduction to Reaction Mechanisms — Part C
▶️ Answer/Explanation
A.
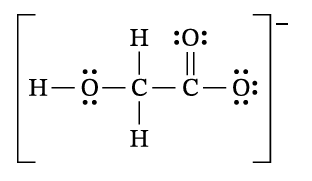
In the glycolate ion \( C_2H_3O_3^- \), the negatively charged oxygen atom is the basic site that accepts a proton (\( H^+ \)) from water. On the Lewis diagram, one of the terminal oxygen atoms with a negative formal charge should be circled. Due to resonance, both terminal oxygen atoms are equivalent.
✅ Answer: Circle either terminal oxygen atom.
B (i).
Given: \( [C_2H_3O_3^-]_{\text{initial}} = 2.5 \, M \), \( [OH^-]_{\text{eq}} = 1.3 \times 10^{-5} \, M \).
From stoichiometry: \( [HC_2H_3O_3]_{\text{eq}} = [OH^-]_{\text{eq}} = 1.3 \times 10^{-5} \, M \).
\( [C_2H_3O_3^-]_{\text{eq}} \approx 2.5 \, M – 1.3 \times 10^{-5} \, M \approx 2.5 \, M \) (since \( 1.3 \times 10^{-5} \) is negligible compared to 2.5).
Using \( K_b \) expression:
\( K_b = \frac{[HC_2H_3O_3][OH^-]}{[C_2H_3O_3^-]} = \frac{(1.3 \times 10^{-5})(1.3 \times 10^{-5})}{2.5} \)
\( K_b = \frac{(1.3 \times 10^{-5})^2}{2.5} = \frac{1.69 \times 10^{-10}}{2.5} = 6.76 \times 10^{-11} \)
✅ Answer: \( \boxed{K_b = 6.8 \times 10^{-11}} \)
B (ii).
At 25°C, \( K_w = 1.0 \times 10^{-14} \).
For conjugate acid-base pair: \( K_a \times K_b = K_w \)
\( K_a = \frac{K_w}{K_b} = \frac{1.0 \times 10^{-14}}{6.76 \times 10^{-11}} = 1.48 \times 10^{-4} \)
✅ Answer: \( \boxed{K_a = 1.5 \times 10^{-4}} \)
C.
Agree. The student’s claim is correct.
Justification:
1. In Step 1, \( H_3O^+ \) is consumed as a reactant.
2. In Step 2, \( H_3O^+ \) is regenerated as a product.
3. \( H_3O^+ \) does not appear in the overall reaction equation.
4. The species is used in the first step and reformed in the second, which is the definition of a catalyst.
✅ Answer: \( \boxed{\text{Agree}} \) with the justification that \( H_3O^+ \) is consumed and then regenerated.