AP Physics 2- 15.2 The Bohr Model of Atomic Structure- Exam Style questions - FRQs- New Syllabus
The Bohr Model of Atomic Structure AP Physics 2 FRQ
Unit 15: Modern Physics
Weightage : 15–18%
Question
Most-appropriate topic codes (AP Physics 2):
• Topic \(15.3\) — Emission and Absorption Spectra (Part \( \mathrm{(c)} \), Part \( \mathrm{(d)} \))
• Topic \(15.1\) — Quantum Theory and Wave-Particle Duality (Part \( \mathrm{(d)(i)} \), Part \( \mathrm{(d)(ii)} \))
• Topic \(10.4\) — Electric Potential Energy (Part \( \mathrm{(b)} \), Part \( \mathrm{(d)(iii)} \))
▶️ Answer/Explanation
(a)
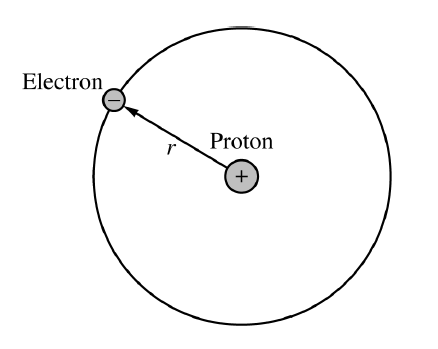
The electrostatic force between the proton and electron provides the centripetal force needed to keep the electron moving in a circular orbit.
\(F_E=F_C\)
The magnitude of the electrostatic force is \(F_E=\dfrac{k e^2}{r^2}\), and the centripetal force on the electron is \(F_C=\dfrac{m_e v^2}{r}\).
Set the two forces equal:
\(\dfrac{k e^2}{r^2}=\dfrac{m_e v^2}{r}\)
Multiply both sides by \(r\):
\(\dfrac{k e^2}{r}=m_e v^2\)
Solve for \(v\):
\(v^2=\dfrac{k e^2}{m_e r}\)
\(\boxed{v=\sqrt{\dfrac{k e^2}{m_e r}}}\)
Here, \(k\) is Coulomb’s constant, \(e\) is the magnitude of the charge of the electron or proton, \(m_e\) is the mass of the electron, and \(r\) is the orbital radius.
(b)
The total energy of the atom is the sum of the electric potential energy and the kinetic energy of the electron.
Since the proton and electron have opposite charges, the electric potential energy is negative:
\(U_E=-\dfrac{k e^2}{r}\)
The kinetic energy of the electron is \(K=\dfrac{1}{2}m_e v^2\).
From part (a), \(v^2=\dfrac{k e^2}{m_e r}\).
Substitute this into the kinetic energy equation:
\(K=\dfrac{1}{2}m_e\left(\dfrac{k e^2}{m_e r}\right)\)
\(K=\dfrac{k e^2}{2r}\)
Now add electric potential energy and kinetic energy:
\(E=U_E+K\)
\(E=-\dfrac{k e^2}{r}+\dfrac{k e^2}{2r}\)
\(\boxed{E=-\dfrac{k e^2}{2r}}\)
The total energy is negative, which means the electron is bound to the proton.
(c)
Yes, the equation is consistent with the description.
From part (b), the total energy is \(E=-\dfrac{k e^2}{2r}\).
When the atom absorbs a photon, the electron moves to an orbit with a larger radius \(r\). As \(r\) increases, the denominator \(2r\) increases, so the magnitude of the negative energy becomes smaller.
This means the total energy becomes less negative, or closer to \(0\). A value closer to \(0\) is greater than a more negative value.
Therefore, as \(r\) increases, the total energy increases.
\(\boxed{\text{The equation is consistent with the model.}}\)
(d)(i)
The energy of a photon is given by \(E=hf\).
Substitute \(h=6.63\times 10^{-34}\,\text{J}\cdot\text{s}\) and \(f=3.2\times 10^{15}\,\text{Hz}\):
\(E=\left(6.63\times 10^{-34}\,\text{J}\cdot\text{s}\right)\left(3.2\times 10^{15}\,\text{s}^{-1}\right)\)
\(E=2.12\times 10^{-18}\,\text{J}\)
\(\boxed{E\approx 2.1\times 10^{-18}\,\text{J}}\)
(d)(ii)
The energy needed to create a particle with the mass of an electron is found using \(E=mc^2\).
For an electron, \(m_e=9.11\times 10^{-31}\,\text{kg}\), and the speed of light is \(c=3.00\times 10^8\,\text{m/s}\).
\(E=\left(9.11\times 10^{-31}\,\text{kg}\right)\left(3.00\times 10^8\,\text{m/s}\right)^2\)
\(E=\left(9.11\times 10^{-31}\right)\left(9.00\times 10^{16}\right)\,\text{J}\)
\(E=8.20\times 10^{-14}\,\text{J}\)
The photon energy from part (d)(i) is \(2.12\times 10^{-18}\,\text{J}\).
Compare the two energies:
\(2.12\times 10^{-18}\,\text{J} \ll 8.20\times 10^{-14}\,\text{J}\)
The photon energy is much smaller than the energy needed to create another electron.
\(\boxed{\text{There is not sufficient energy to add another electron.}}\)
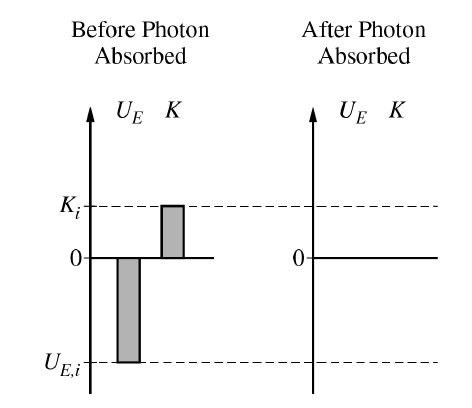
(d)(iii)
After the photon is absorbed, the atom has a greater total energy. The electron moves to a larger orbit, so the electric potential energy becomes less negative.
Since \(U_E=-\dfrac{k e^2}{r}\), increasing \(r\) makes \(U_E\) closer to \(0\). Therefore, the final electric potential energy bar should still be below \(0\), but it should have a smaller magnitude than \(U_{E,i}\).
The kinetic energy is \(K=\dfrac{k e^2}{2r}\). Since \(r\) increases, the kinetic energy decreases. Therefore, the final kinetic energy bar should still be above \(0\), but it should be smaller than \(K_i\).
A correct final bar chart should show \(U_{E,f}<0\), with \(|U_{E,f}|<|U_{E,i}|\), and \(0<K_f<K_i\).
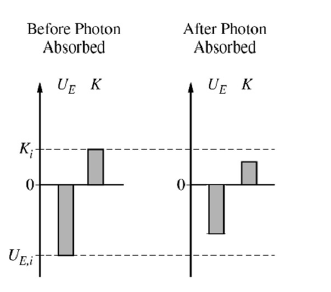
The total energy after absorption is larger because the total energy becomes less negative.
\(\boxed{U_{E,f}\text{ is less negative, and }K_f\text{ is smaller but still positive.}}\)
