AP Physics 2- 9.3 Thermal Energy Transfer and Equilibrium- Exam Style questions - FRQs- New Syllabus
Thermal Energy Transfer and Equilibrium AP Physics 2 FRQ
Unit 9: Thermodynamics
Weightage : 15–18%
Question
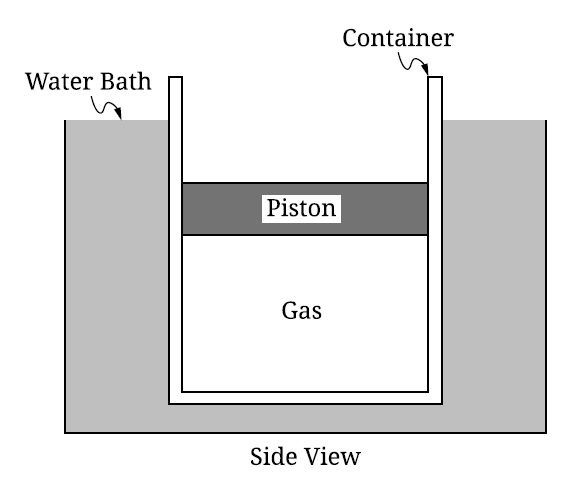
_____ \( T_{\mathrm{new}} < T_0 \)
_____ \( T_{\mathrm{new}} = T_0 \)
Most-appropriate topic codes (AP Physics 2):
• Topic \( 9.3 \) — Thermal Energy Transfer and Equilibrium (Part \( \mathrm{A} \), Part \( \mathrm{C} \), Part \( \mathrm{D} \))
• Topic \( 9.4 \) — The First Law of Thermodynamics (Part \( \mathrm{B} \), Part \( \mathrm{C} \), Part \( \mathrm{D} \))
▶️ Answer/Explanation
A.
The piston has three forces acting on it:
\( \bullet \) Upward force exerted by the gas, \( F_{\mathrm{gas}} \)
\( \bullet \) Downward weight of the piston, \( F_g = Mg \)
\( \bullet \) Downward force from the atmosphere, \( F_{\mathrm{atm}} = P_{\mathrm{atm}}A \)
A correct force diagram is shown below.
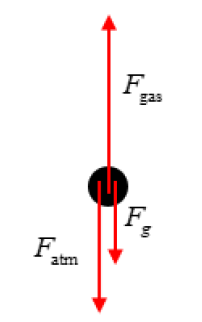
Because the piston is at rest, these forces balance.
B.
For a monatomic ideal gas,
\( U = \dfrac{3}{2}nRT \)
and from the ideal gas law,
\( PV = nRT \)
so
\( U = \dfrac{3}{2}PV \)
Now find the gas pressure using force balance on the piston:
Upward force from the gas: \( PA \)
Downward forces: \( P_{\mathrm{atm}}A + Mg \)
Since the piston is at rest,
\( PA – P_{\mathrm{atm}}A – Mg = 0 \)
\( P = P_{\mathrm{atm}} + \dfrac{Mg}{A} \)
Therefore,
\( U = \dfrac{3}{2}PV_0 \)
\( \boxed{U = \dfrac{3}{2}\left(P_{\mathrm{atm}} + \dfrac{Mg}{A}\right)V_0} \)
This works nicely because the gas is ideal and monatomic, so internal energy depends only on temperature, and \( PV \) gives the same result through the ideal gas law.
C.
Because the container is thermally conducting and the water bath remains at constant temperature, the process is approximately isothermal.
When the extra block is slowly added, the external pressure on the gas increases, so the gas is compressed. For an isothermal compression, \( PV = \text{constant} \), so the \( P\text{-}V \) curve is a hyperbola.
The process moves from a point at lower pressure and larger volume to a point at higher pressure and smaller volume, so the arrow points up and to the left along a concave-up curve.
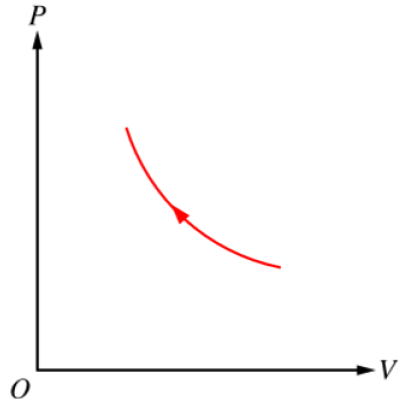
D.
\( \boxed{T_{\mathrm{new}} > T_0} \)
With the extra block still on the piston, the gas must support a larger downward force than it did initially. Therefore, at equilibrium the gas pressure is larger than before.
In part \( \mathrm{B} \), the initial pressure was \( P_{\mathrm{atm}} + \dfrac{Mg}{A} \). With an added block of the same mass \( M \), the final equilibrium pressure at volume \( V_0 \) is
\( P_{\mathrm{final}} = P_{\mathrm{atm}} + \dfrac{2Mg}{A} \)
Since the gas ends at the same volume \( V_0 \) but at a larger pressure, the ideal gas law implies a larger temperature:
\( PV = nRT \)
With \( n \) constant and \( V \) unchanged, larger \( P \) means larger \( T \). Therefore, \( T_{\mathrm{new}} > T_0 \).
This is also consistent with the \( P\text{-}V \) picture in part \( \mathrm{C} \): to return to the original volume while supporting the added block, the gas must end at a higher pressure than it had initially.
