CELLS 1.3 Enzymes- Pre AP Biology Study Notes - New Syllabus.
CELLS 1.3 Enzymes- Pre AP Biology Study Notes
CELLS 1.3 Enzymes- Pre AP Biology Study Notes – New Syllabus.
LEARNING OBJECTIVE
CELLS 1.3(a) Describe the effect of enzymes on the rate of chemical reactions in biological systems.
CELLS 1.3(b) Predict how a change in pH and/or temperature will affect the function of an enzyme.
Key Concepts:
- CELLS 1.3.1 Enzymes are proteins that are catalysts in biochemical reactions and essential for maintaining life processes.
a. The rate of a chemical reaction is affected by the concentration of substrates and enzymes.
b. Enzymes have specific shapes that bind to specific substrates in a precise location called the active site.
c. Enzymes function optimally in a specific pH and temperature range.
Effect of Enzymes on the Rate of Chemical Reactions in Biological Systems
🌱 Introduction
All life depends on chemical reactions.
However, most biological reactions are too slow to support life on their own.
This is where enzymes play a crucial role.
Enzymes speed up chemical reactions in cells without being consumed.
📌 What Are Enzymes?
Enzymes are biological catalysts, usually made of proteins.
Key characteristics:
- Speed up chemical reactions
- Not used up or permanently changed
- Highly specific to their substrates
- Required for nearly all cellular processes
Enzymes increase the rate of chemical reactions without being altered themselves.
⚙️ Why Are Enzymes Necessary in Cells?
Without enzymes:
- Reactions would occur very slowly
- Cells would not get energy fast enough
- Life processes like respiration and digestion would fail
With enzymes:
- Reactions occur at life-supporting speeds
- Cells function efficiently
- Metabolism is maintained
Enzymes make life chemically possible.
🧬 How Enzymes Increase the Rate of Reactions
Activation Energy and Enzymes
Activation energy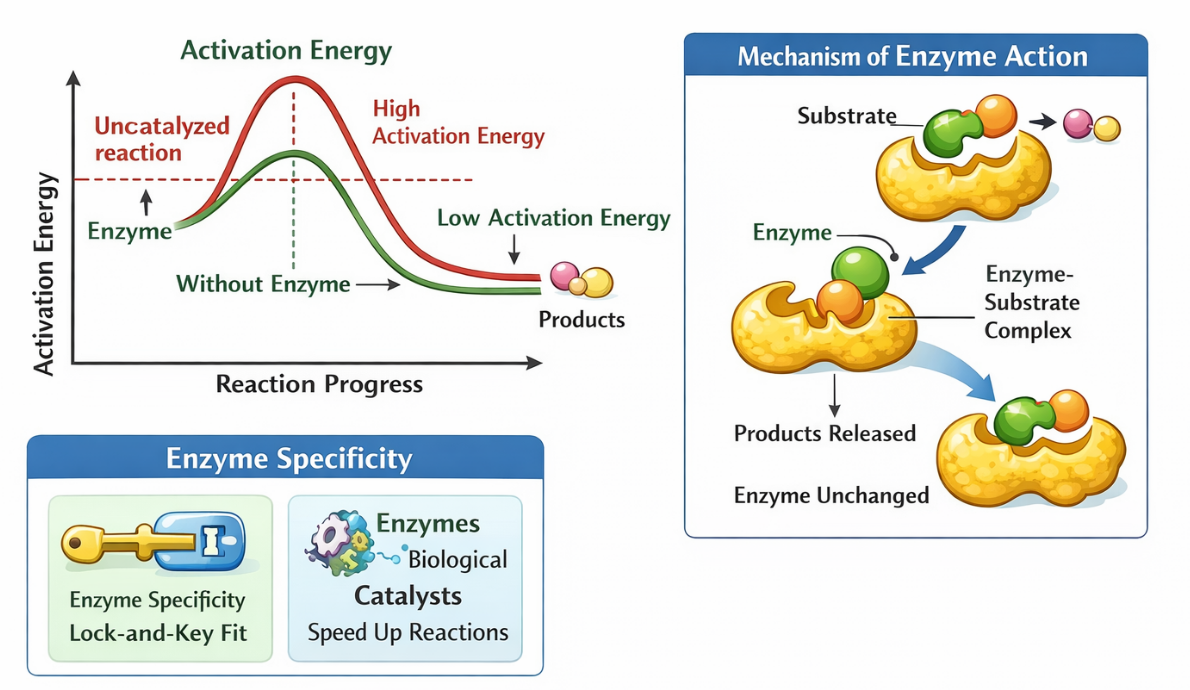
Activation energy is the minimum energy required to start a chemical reaction.
- High activation energy = slow reaction
- Low activation energy = fast reaction
Key role of enzymes:
Enzymes lower the activation energy of reactions.
Mechanism of Enzyme Action
Step-by-step:
- Substrate binds to the enzyme
- Enzyme–substrate complex forms
- Enzyme stabilizes the transition state
- Products are released
- Enzyme is free to be reused
📌 Important:
The enzyme is unchanged after the reaction.
🧬 How Enzymes Lower Activation Energy
Enzymes lower activation energy by:
- Bringing reactants closer together
- Correctly orienting substrates
- Weakening existing chemical bonds
- Creating a favorable microenvironment
Result: More molecules can react in less time.
⚡ Effect on Reaction Rate
With enzymes:
- Reaction rate increases dramatically
- Reactions occur in milliseconds instead of years
Without enzymes:
- Same reactions may occur extremely slowly
Enzymes affect reaction rate but do not change the final products or equilibrium.
🎯 Enzyme Specificity and Reaction Rate
Enzymes are substrate-specific.
- Each enzyme works on a particular substrate
- Correct binding ensures efficient reactions
Lock-and-key or induced-fit concept supports this specificity.
🧬 Enzymes and Biological Systems
Enzymes control:
- Cellular respiration
- Photosynthesis
- DNA replication
- Protein synthesis
- Digestion
Every metabolic pathway depends on enzymes.
📌 Important Clarifications
- Enzymes do not create energy
- Enzymes do not change reaction direction
- Enzymes do not affect overall energy released
- Enzymes only speed up the rate
📊 Summary Table: Enzymes and Reaction Rate
| Aspect | Without Enzymes | With Enzymes |
|---|---|---|
| Activation energy | High | Lowered |
| Reaction speed | Very slow | Very fast |
| Cell survival | Not possible | Possible |
| Enzyme use | Not applicable | Reusable |
🧠 Key Statements
- Enzymes are biological catalysts
- They lower activation energy
- They increase reaction rate
- They are not consumed
- They are specific to substrates
📦 Quick Recap
Enzymes speed up chemical reactions
They lower activation energy
They form enzyme substrate complexes
They are reusable and specific
They do not change reaction products
They are essential for life
Effect of pH and Temperature on Enzyme Function
🌱 Introduction
Enzymes are proteins, and like all proteins, their function depends on their shape.
Changes in environmental conditions such as temperature and pH can alter enzyme structure and therefore affect enzyme activity.
Any change in temperature or pH that alters enzyme shape will affect the rate of the reaction it catalyzes.
📌 Why Are pH and Temperature Important for Enzymes?
- Enzymes work best under specific conditions
- These conditions are called optimum temperature and optimum pH
- Deviations from these conditions can:
- Slow enzyme activity
- Stop enzyme activity
- Permanently damage the enzyme
🌡️ Effect of Temperature on Enzyme Function
Optimum Temperature
Each enzyme has an optimum temperature at which it works fastest.
- In human enzymes, this is usually around 37°C
At optimum temperature:
- Molecules move efficiently
- Enzyme–substrate collisions increase
- Reaction rate is highest
Increase in Temperature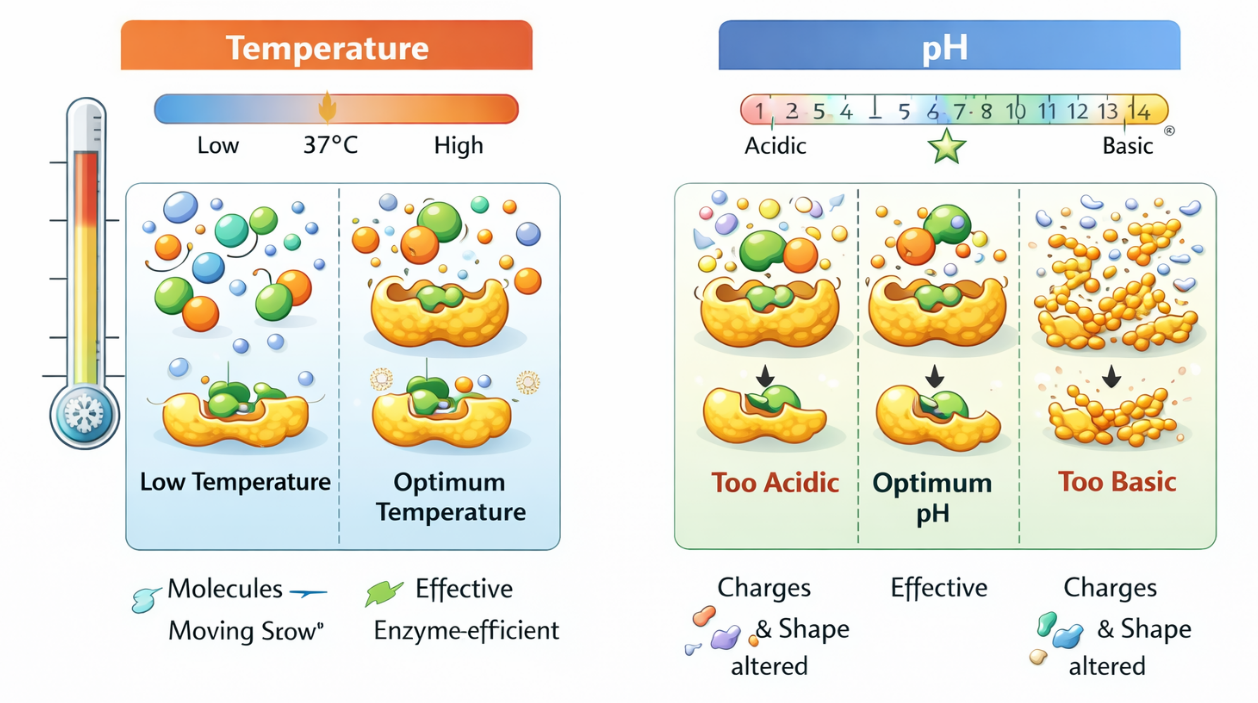
Moderate increase:
- Molecules move faster
- More collisions between enzyme and substrate
- Reaction rate increases
This happens up to the optimum temperature.
High temperature (above optimum):
- Excess heat disrupts weak bonds in the enzyme
- Enzyme loses its specific shape
- Active site changes
This process is called denaturation.
📌 Result:
- Substrate no longer fits
- Reaction rate decreases sharply
- Enzyme may stop functioning permanently
Decrease in Temperature
- Molecules move slowly
- Fewer enzyme–substrate collisions
- Reaction rate decreases
📌 Important note:
Low temperature usually does not denature enzymes.
The enzyme can work again if temperature rises.
📊 Temperature Summary
| Temperature Change | Effect on Enzyme |
|---|---|
| Below optimum | Reaction slows |
| At optimum | Maximum activity |
| Above optimum | Denaturation, activity decreases |
⚖️ Effect of pH on Enzyme Function
Optimum pH
Each enzyme also has an optimum pH.
- Most human enzymes work best near pH 7
- Some enzymes work best at extreme pH values
- Example: stomach enzymes work best in acidic pH
pH affects charge and shape of the enzyme.
Change in pH (Too Acidic or Too Basic)
How pH change affects enzymes:
- Alters charges on amino acids
- Disrupts hydrogen and ionic bonds
- Changes active site shape
Result:
- Substrate binding decreases
- Enzyme activity reduces
Extreme pH Change
- Enzyme structure is permanently altered
- Active site is destroyed
- Enzyme becomes denatured
📊 pH Summary
| pH Condition | Effect on Enzyme |
|---|---|
| Optimum pH | Maximum activity |
| Slight pH change | Reduced activity |
| Extreme pH | Denaturation |
🧠 Predicting Enzyme Behavior
- If temperature increases slightly: Reaction rate increases
- If temperature becomes very high: Enzyme denatures and stops working
- If temperature decreases: Reaction rate slows but enzyme remains intact
- If pH moves away from optimum: Enzyme activity decreases
- If pH becomes extreme: Enzyme denatures and loses function permanently
📌 Key Concept for Predictions
Enzyme function depends on maintaining proper shape of the active site.
Any factor that disrupts this shape will reduce enzyme efficiency.
📦 Quick Recap
Enzymes have optimum temperature and pH
Moderate temperature increase speeds reactions
High temperature causes denaturation
Low temperature slows reactions
pH affects enzyme charge and shape
Extreme pH denatures enzymes
Shape change leads to loss of function
