Pre AP Biology -CELLS 1.3 Enzymes- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Biology -CELLS 1.3 Enzymes- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Biology -CELLS 1.3 Enzymes- MCQ Exam Style Questions – Pre AP Biology – per latest Pre AP Biology Syllabus.
Question
▶️ Answer/Explanation
The correct answer is a.
In noncompetitive inhibition, the inhibitor binds to an allosteric site rather than the active site.
This binding induces a change in the 3D conformation (shape) of the enzyme.
As a result, the active site can no longer effectively catalyze the conversion of substrate to product.
Unlike competitive inhibition, the inhibitor does not need to resemble the substrate.
Both types of inhibition decrease the reaction rate, so statement c is not unique to noncompetitive.
Inhibition is typically reversible, meaning statement d is generally incorrect for standard noncompetitive models.
Question
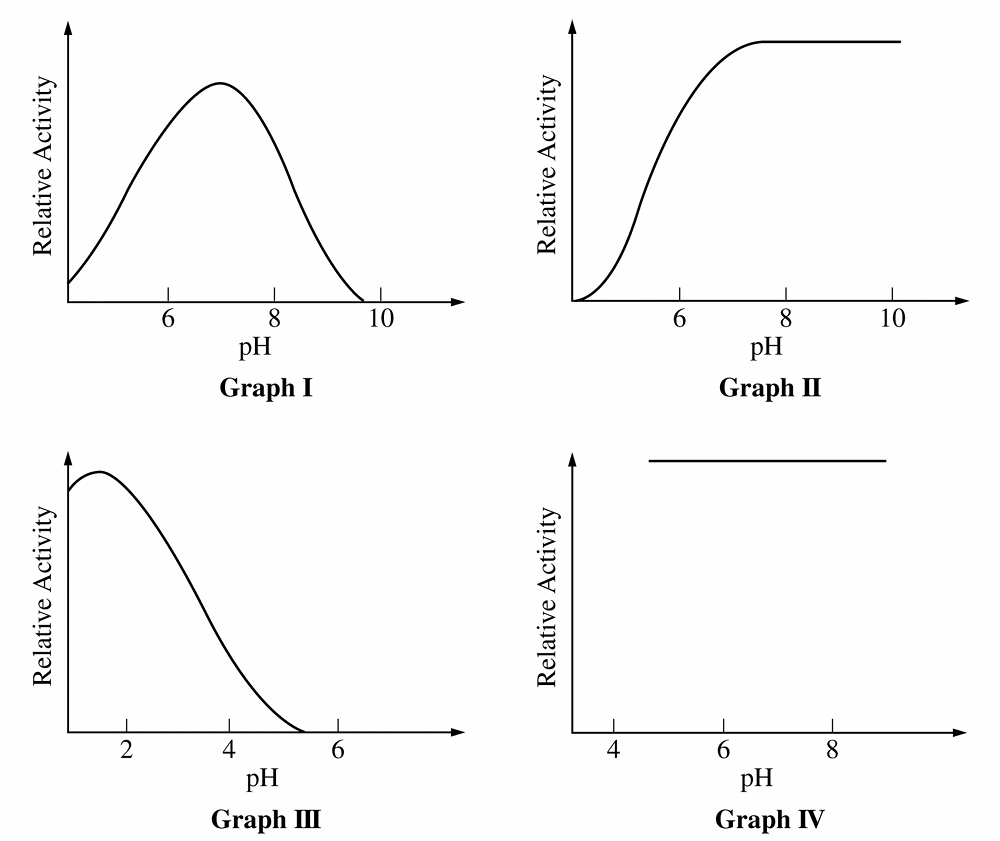
B. \(II\) only
C. \(III\) only
D. \(I\) and \(III\) only
B. \(IV\) only
C. \(II\) and \(III\) only
D. \(I\), \(II\), and \(III\) only
B. \(pH\) affects the temperature of the reaction
C. the enzyme has a quaternary structure
D. the enzyme has disulfide bonds
▶️ Answer/Explanation
A. Answer: D (I, II, and III only)
A. Answer: A
For Q, Graph \(III\) peaks at acidic \(pH\) (\(\approx 2\)) and Graph \(I\) peaks at neutral \(pH\) (\(\approx 7\)).
Graph \(II\) shows activity increasing and remaining high in the alkaline range (\(pH\) \(8\)-\(10\)), identifying it as the alkaline enzyme.
For Q, “sensitive” means enzyme activity varies as \(pH\) changes.
Graph \(IV\) is a horizontal line, indicating activity is constant and independent of \(pH\) (insensitive).
Graphs \(I\), \(II\), and \(III\) all show curves where activity changes with \(pH\), making them the correct choice.
For Q, enzyme activity depends on the precise shape of the active site.
Changes in \(pH\) alter the charge of amino acid side chains, disrupting the bonds holding the active site’s shape.
This loss of specific shape prevents substrate binding, explaining the decline in activity seen in Graph \(I\).
Question
B. Only secondary structure
C. Only quaternary structure
D. It will not have any level of protein structure
▶️ Answer/Explanation
Denaturation involves the disruption of weak bonds such as hydrogen bonds and ionic interactions.
This process causes the protein to lose its quaternary, tertiary, and secondary structures.
The primary structure consists of amino acids linked by strong covalent peptide bonds.
These covalent bonds are not broken by standard denaturing agents like heat or pH changes.
Therefore, the linear sequence of amino acids remains intact after denaturation.
The protein returns to an unfolded polypeptide chain, retaining only its primary level of organization.
Question
The human insulin protein is comprised of $51$ amino acids.
How many bases was the mRNA made up of?
▶️ Answer/Explanation
The genetic code is a triplet code, meaning $3$ mRNA bases (a codon) code for $1$ amino acid.
To find the number of coding bases, multiply the number of amino acids by $3$: $51 \times 3 = 153$ bases.
However, every mRNA sequence must also include a stop codon to signal the end of translation.
A stop codon consists of an additional $3$ bases that do not code for an amino acid.
The total number of bases is calculated as: $153 \text{ (coding bases)} + 3 \text{ (stop codon)} = 156$ bases.
Therefore, the correct option is (A).
Question
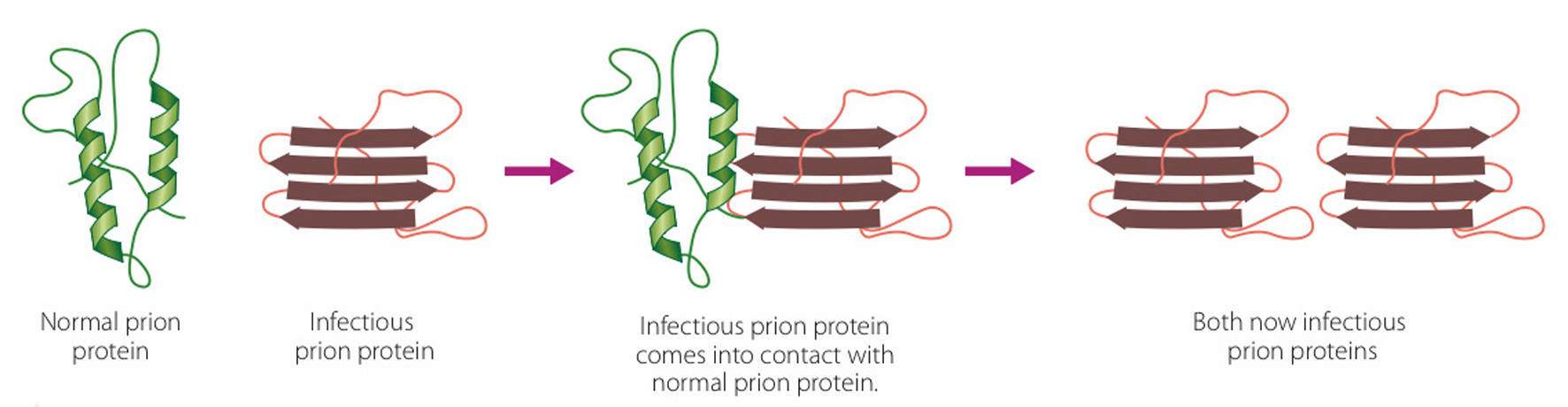
(B) Prions can be transmitted by consuming prion-infected brain leading to diseases such as Kuru.
(C) The protein is caused by a mutation that is inherited by offspring.
(D) The diagram shows the communicable nature of the protein as it induces abnormal folding patterns in normal proteins when they come into contact.
▶️ Answer/Explanation
The diagram illustrates a normal prion protein (green helix structure) interacting with an infectious prion protein (red sheet structure).
Upon contact, the infectious prion acts as a template, inducing the normal protein to misfold into the abnormal shape.
This process results in two infectious prions, demonstrating how the condition propagates within an organism.
This mechanism highlights the communicable nature of the protein at a molecular level.
Option (A) is incorrect as the diagram shows the protein “spreading” its state to others.
Option (B) mentions a specific transmission route (ingestion) not explicitly shown in the molecular interaction diagram.
Option (C) refers to genetic inheritance, whereas the diagram focuses on protein-to-protein conformational change.
Question
▶️ Answer/Explanation
The correct answer is D.
Enzymes function most efficiently at their optimal temperature, which is $25^\circ\text{C}$ in this case.
Increasing the temperature from $15^\circ\text{C}$ toward $25^\circ\text{C}$ provides more kinetic energy, increasing molecular collisions.
As the temperature reaches the optimum, the rate of reaction hits its maximum point.
Option B is incorrect because temperatures above the optimum can cause the enzyme to denature.
Options A and C would either have no effect or would decrease the reaction rate.
Question
Experiment 1:
- Hydrogen peroxide was poured on a slice of raw potato.
- The peroxide bubbled vigorously.
- The peroxide was broken down into water and oxygen.
Experiment 2:
- A slice of raw potato was heated and then hydrogen peroxide was poured on it.
- There was no visible reaction.
B. Heating the potato caused the hydrogen peroxide to evaporate, so the reaction did not occur.
C. Heating the potato caused the potato enzymes to release the water and oxygen from the hydrogen peroxide.
D. Heating the potato caused a shape change in the potato enzymes, so they could no longer function properly.
▶️ Answer/Explanation
The correct answer is D.
Potatoes contain the enzyme catalase, which breaks down $\text{H}_2\text{O}_2$ into $\text{H}_2\text{O}$ and $\text{O}_2$.
In Experiment 1, the enzyme is active, resulting in vigorous bubbling (oxygen release).
In Experiment 2, heating the potato provides thermal energy that disrupts chemical bonds.
This causes the enzyme to denature, permanently changing the shape of its active site.
Because the shape is lost, the substrate can no longer bind, and the reaction stops.
