Pre AP Biology -CELLS 1.3 Enzymes- MCQ Exam Style Questions -New Syllabus
Pre AP Biology -CELLS 1.3 Enzymes- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Biology -CELLS 1.3 Enzymes- MCQ Exam Style Questions – Pre AP Biology – per latest Pre AP Biology Syllabus.
Question
▶️ Answer/Explanation
The correct answer is a.
In noncompetitive inhibition, the inhibitor binds to an allosteric site rather than the active site.
This binding induces a change in the 3D conformation (shape) of the enzyme.
As a result, the active site can no longer effectively catalyze the conversion of substrate to product.
Unlike competitive inhibition, the inhibitor does not need to resemble the substrate.
Both types of inhibition decrease the reaction rate, so statement c is not unique to noncompetitive.
Inhibition is typically reversible, meaning statement d is generally incorrect for standard noncompetitive models.
Question
B. \(II\) only
C. \(III\) only
D. \(I\) and \(III\) only
B. \(IV\) only
C. \(II\) and \(III\) only
D. \(I\), \(II\), and \(III\) only
B. \(pH\) affects the temperature of the reaction
C. the enzyme has a quaternary structure
D. the enzyme has disulfide bonds
▶️ Answer/Explanation
A. Answer: D (I, II, and III only)
A. Answer: A
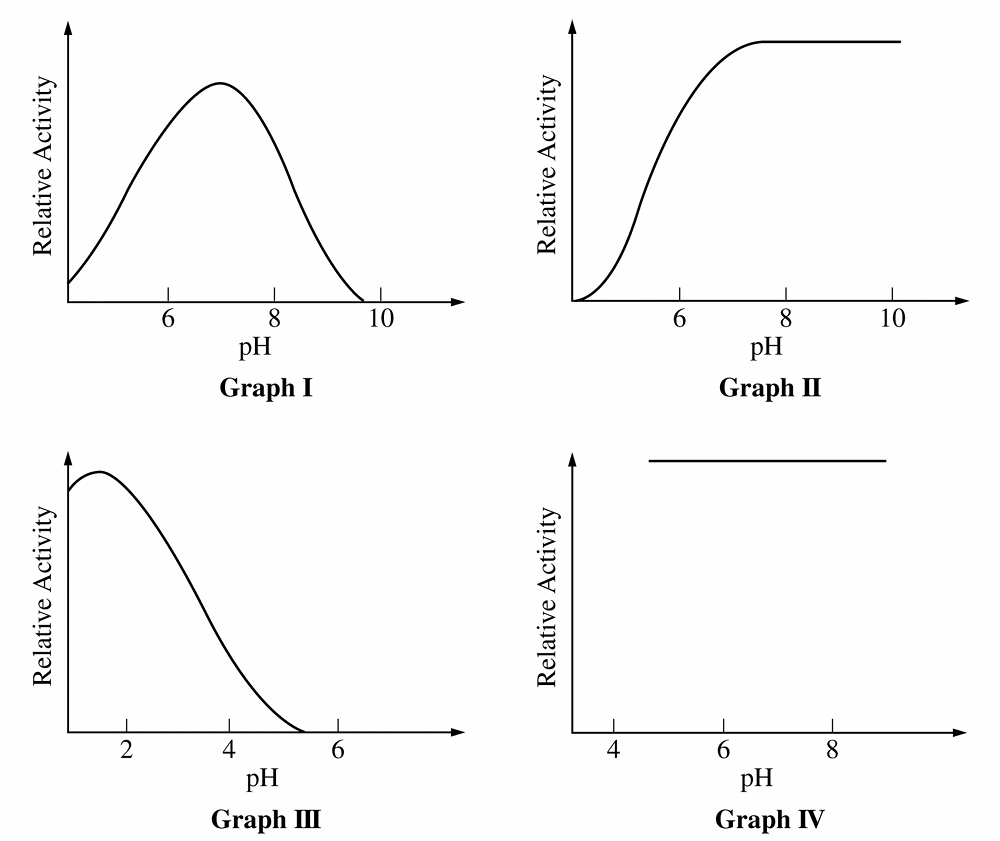
For Q, Graph \(III\) peaks at acidic \(pH\) (\(\approx 2\)) and Graph \(I\) peaks at neutral \(pH\) (\(\approx 7\)).
Graph \(II\) shows activity increasing and remaining high in the alkaline range (\(pH\) \(8\)-\(10\)), identifying it as the alkaline enzyme.
For Q, “sensitive” means enzyme activity varies as \(pH\) changes.
Graph \(IV\) is a horizontal line, indicating activity is constant and independent of \(pH\) (insensitive).
Graphs \(I\), \(II\), and \(III\) all show curves where activity changes with \(pH\), making them the correct choice.
For Q, enzyme activity depends on the precise shape of the active site.
Changes in \(pH\) alter the charge of amino acid side chains, disrupting the bonds holding the active site’s shape.
This loss of specific shape prevents substrate binding, explaining the decline in activity seen in Graph \(I\).
Question
B. Only secondary structure
C. Only quaternary structure
D. It will not have any level of protein structure
▶️ Answer/Explanation
Denaturation involves the disruption of weak bonds such as hydrogen bonds and ionic interactions.
This process causes the protein to lose its quaternary, tertiary, and secondary structures.
The primary structure consists of amino acids linked by strong covalent peptide bonds.
These covalent bonds are not broken by standard denaturing agents like heat or pH changes.
Therefore, the linear sequence of amino acids remains intact after denaturation.
The protein returns to an unfolded polypeptide chain, retaining only its primary level of organization.
