1.2B Endothermic and Exothermic Processes- Pre AP Chemistry Study Notes - New Syllabus.
1.2B Endothermic and Exothermic Processes- Pre AP Chemistry Study Notes
1.2B Endothermic and Exothermic Processes- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
1.2.B.1 Use data to explain the direction of energy flow into or out of a system.
Key Concepts:
- 1.2.B Energy transfers are classified as endothermic or exothermic.
a. In endothermic changes, energy flows from the surroundings to the system.
b. In exothermic changes, energy flows from the system to the surroundings.
1.2.B.1 — Direction of Energy Flow: Endothermic vs. Exothermic
Energy transfer during physical or chemical changes can be understood by defining a system and its surroundings. Using experimental data (such as temperature change), scientists explain the direction of energy flow and classify the process as endothermic or exothermic.
System and Surroundings
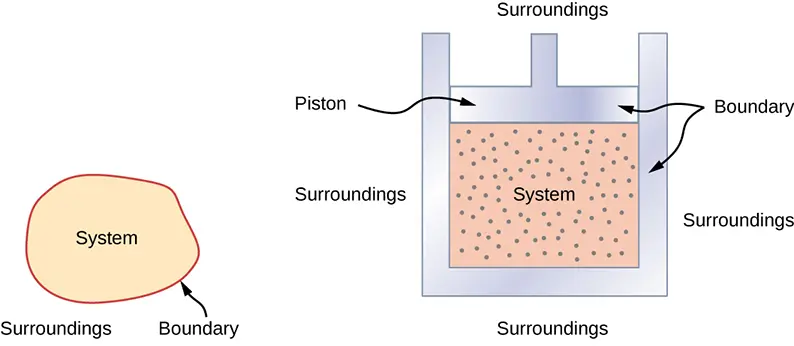
- System: the part of the universe being studied (reaction, dissolving substance, phase change)
- Surroundings: everything outside the system (solution, air, container)
Energy transfer is tracked by observing changes in the surroundings, most commonly temperature change.
Classifying Energy Transfers
Energy transfers are classified based on the direction energy flows:
| Type of Change | Direction of Energy Flow | Temperature of Surroundings |
|---|---|---|
| Endothermic | Surroundings → System | Decreases |
| Exothermic | System → Surroundings | Increases |
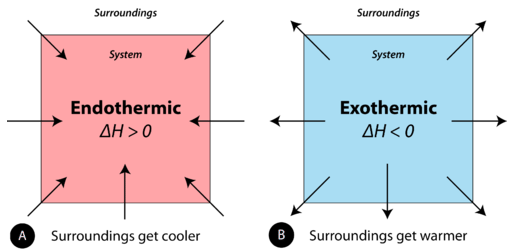
Exothermic Energy Transfers
In an exothermic change:
- Energy flows from the system to the surroundings
- The system releases energy
- The surroundings gain energy
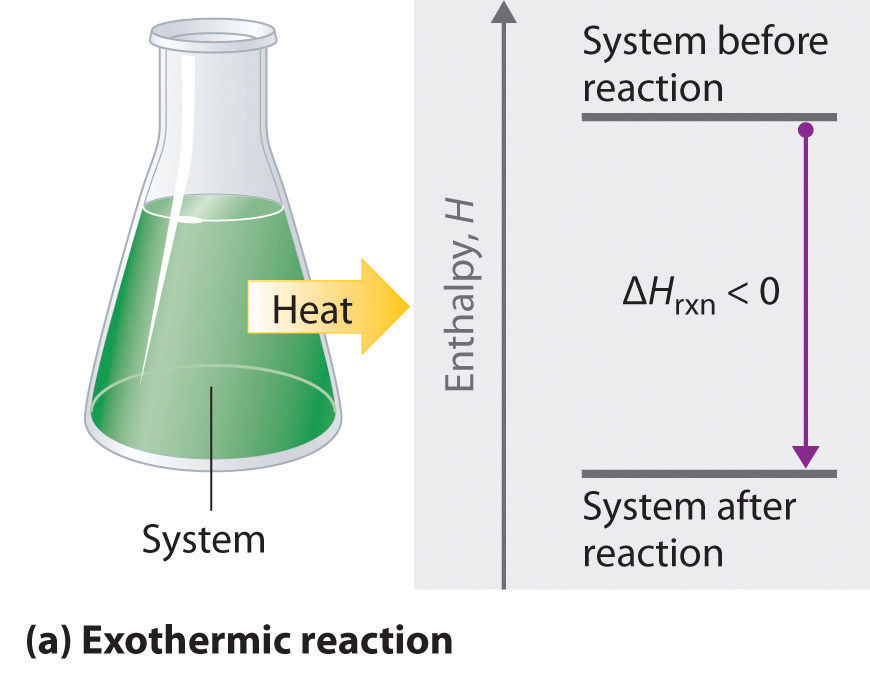
Using data, this is identified by an increase in the temperature of the surroundings.
Examples include combustion and neutralization reactions.
Endothermic Energy Transfers
In an endothermic change:
- Energy flows from the surroundings to the system
- The system absorbs energy
- The surroundings lose energy
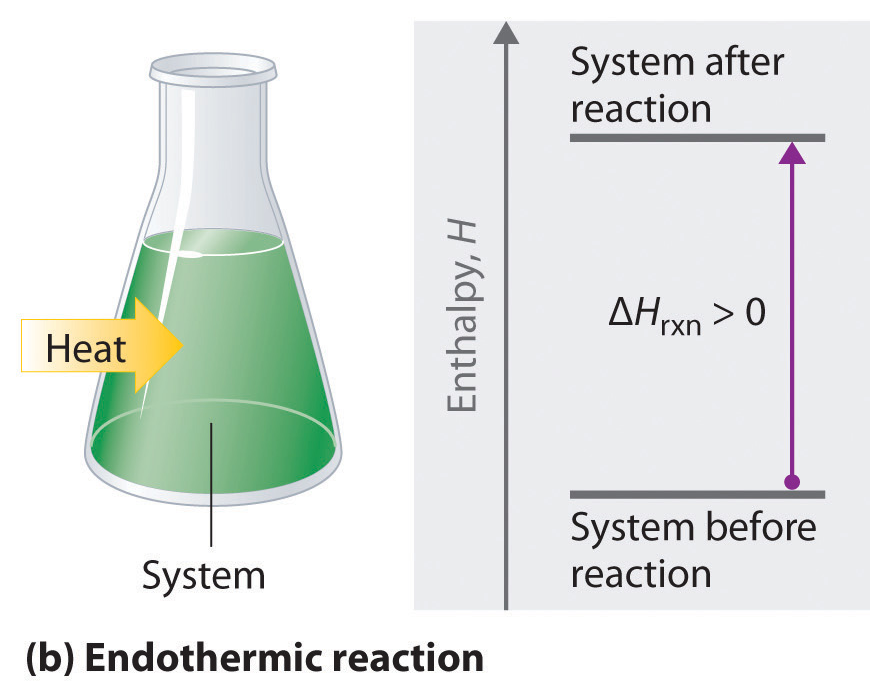
Using data, this is identified by a decrease in the temperature of the surroundings.
Examples include melting ice and dissolving ammonium nitrate in water.
Using Data to Explain Energy Flow
To explain the direction of energy flow using data:
- Identify whether the temperature of the surroundings increases or decreases
- Determine whether energy was gained or lost by the surroundings
- Infer the opposite energy change for the system
- Classify the process as endothermic or exothermic
Example
During a reaction, the temperature of the solution drops from \( \mathrm{25.0^\circ C} \) to \( \mathrm{21.5^\circ C} \). Use this data to explain the direction of energy flow.
▶️ Answer / Explanation
The temperature of the surroundings decreases, meaning energy is lost by the surroundings.
Therefore, energy flows from the surroundings into the system. The process is endothermic.
Example
A calorimetry experiment shows \( \mathrm{q_{solution} = +2.10 \times 10^{3}\ J} \). Use this data to determine the direction of energy flow and classify the change.
▶️ Answer / Explanation
A positive value of \( \mathrm{q_{solution}} \) means the surroundings gained energy.
Energy must have flowed from the system to the surroundings. Therefore, the change is exothermic.
