1.3A Gas Pressure and Particle Collisions- Pre AP Chemistry Study Notes - New Syllabus.
1.3A Gas Pressure and Particle Collisions- Pre AP Chemistry Study Notes
1.3A Gas Pressure and Particle Collisions- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
1.3.A.1 Create and/or evaluate models that illustrate how a gas exerts pressure.
1.3.A.2 Explain the relationship between pressure in a gas and collisions.
Key Concepts:
- 1.3.A The pressure of a gas is the force the gas applies to a unit area of the container it is in.
a. Pressure arises from collisions of particles with the walls of the container.
b. Pressure is measured using several different units that are proportional to each other.
1.3.A.1 — How Gases Exert Pressure (Particle Models)
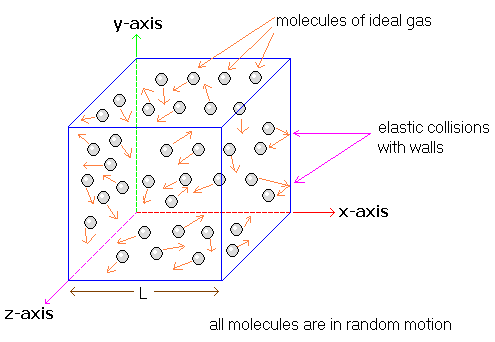
A gas exerts pressure because its particles are in constant, random motion and collide with the walls of their container. Particle (particulate) models are used to illustrate how these collisions create force per unit area.
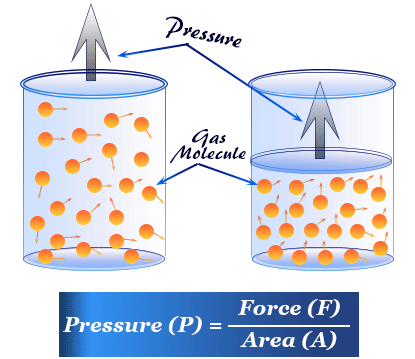
What Is Gas Pressure?
Pressure is the force exerted per unit area. For gases, pressure results from collisions of gas particles with container walls.
![]()
Each collision applies a tiny force; the combined effect of many collisions produces measurable pressure.
Particle-Level Explanation of Gas Pressure
According to particle models:
![]()
- Gas particles move in straight lines between collisions
- Collisions with container walls are frequent
- Each collision transfers momentum to the wall
More frequent and more forceful collisions result in higher gas pressure.
Factors That Affect Gas Pressure in Models
Number of Particles
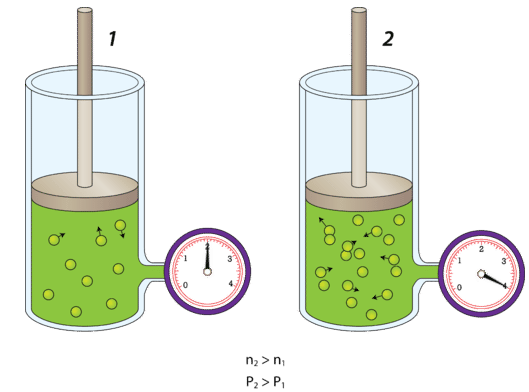
- More particles → more collisions per second
- Pressure increases if volume and temperature are constant
Temperature
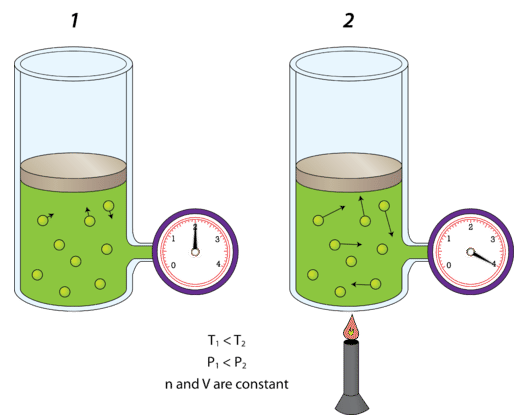
- Higher temperature → faster particle motion
- Collisions are more forceful and more frequent
- Pressure increases if volume is constant
Volume of the Container
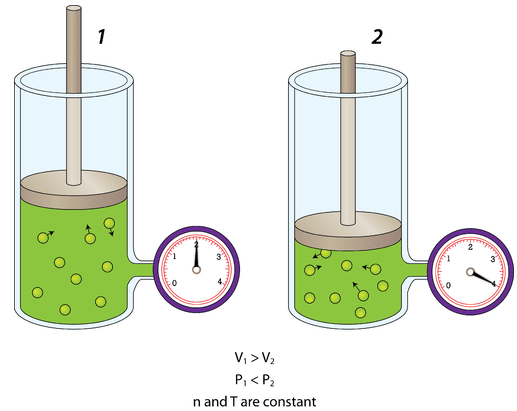
- Smaller volume → particles hit walls more often
- Pressure increases if temperature and particle number are constant
Interpreting Particulate Models of Pressure
| Model Feature | What It Represents |
|---|---|
| More collision arrows | Higher pressure |
| Longer arrows | Greater collision force (higher temperature) |
| Smaller container | More frequent wall collisions |
Evaluating the Accuracy of Pressure Models
A correct model illustrating gas pressure should:
- Show particles moving randomly
- Show collisions with container walls
- Keep particle size constant
- Relate pressure to collision frequency and force
Models that show particles pushing steadily on walls without motion are incorrect.
Example
Two particle models show the same container volume. Model A has fewer particles than Model B. Which model represents a gas with higher pressure? Explain.
▶️ Answer / Explanation
Model B represents the higher pressure.
With more particles in the same volume, there are more collisions with the container walls per second, resulting in greater force on the walls and higher pressure.
Example
A gas is heated while kept in a rigid container. Describe how a particle model would change and explain how this leads to increased pressure.
▶️ Answer / Explanation
Heating increases particle speed, so a particle model would show longer motion arrows.
Faster particles collide with the container walls more frequently and with greater force, increasing the pressure even though the volume remains constant.
1.3.A.2 — Gas Pressure and Collisions
The pressure of a gas is directly related to the collisions made by gas particles. Using particle models, scientists explain gas pressure by examining how often particles collide and how much force those collisions exert on the walls of a container.
Collision-Based Explanation of Gas Pressure
Gas particles are in constant, random motion. Each time a particle collides with a container wall, it exerts a small force. Gas pressure is the result of the combined effect of these collisions.
Two collision-related factors determine gas pressure:
- Collision frequency — how often particles hit the walls
- Collision force — how hard particles hit the walls
Collision Frequency and Pressure
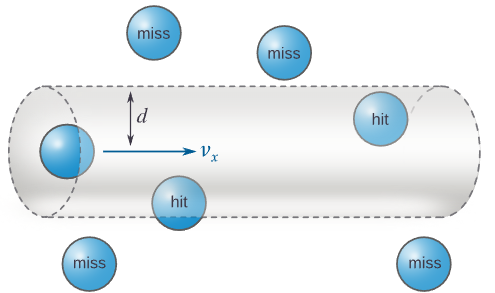
Collision frequency depends on:
- Number of gas particles
- Volume of the container
- Speed of particles
When collision frequency increases, pressure increases because the walls experience more forces per unit time.
Collision Force and Pressure
Collision force depends on the speed of the gas particles.
- Faster-moving particles transfer more momentum per collision
- Greater momentum change produces a larger force
Higher collision force leads to higher gas pressure.
How Temperature Affects Collisions
Temperature is a measure of the average kinetic energy of particles.
![]()
- Increasing temperature increases particle speed
- Collisions become more frequent and more forceful
- Pressure increases if volume is constant
Summarizing the Relationship
| Collision Change | Effect on Pressure |
|---|---|
| More frequent collisions | Pressure increases |
| More forceful collisions | Pressure increases |
| Fewer or weaker collisions | Pressure decreases |
Evaluating Particle Models
A correct particle model explaining gas pressure must:
- Show random particle motion
- Show collisions with container walls
- Relate pressure to collision frequency and force
Models that show particles exerting constant pressure without motion do not correctly explain gas behavior.
Example
A gas in a sealed container experiences an increase in pressure when heated. Explain this change using particle collisions.
▶️ Answer / Explanation
Heating increases the speed of gas particles.
Faster particles collide with the container walls more often and with greater force, increasing the pressure.
Example
Two containers have equal volumes and temperatures. Container A holds twice as many gas particles as Container B. Use collision reasoning to explain which container has the higher pressure.
▶️ Answer / Explanation
Container A has the higher pressure.
With twice as many particles in the same volume, collision frequency with the container walls is greater, resulting in a larger total force and higher pressure.
