2.1B Mixtures and Separation Techniques- Pre AP Chemistry Study Notes - New Syllabus.
2.1B Mixtures and Separation Techniques- Pre AP Chemistry Study Notes
2.1B Mixtures and Separation Techniques- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
2.1.B.1 Create and/or evaluate models of mixtures.
2.1.B.2 Interpret the results of an experiment involving the separation of a mixture.
Key Concepts:
- 2.1.B A mixture is composed of two or more different types of particles that are not bonded.
a. Each component of a mixture retains its unique properties.
b. Mixtures can be separated using physical processes such as filtration, evaporation, distillation, and chromatography.
2.1.B.1 — Models of Mixtures
A mixture is composed of two or more different types of particles that are not chemically bonded. Particle models are used to create and evaluate representations of mixtures by examining how different particles are distributed and interact.
In a mixture, each component retains its own unique properties because no chemical bonds form between the particles.
Key Characteristics of Mixtures
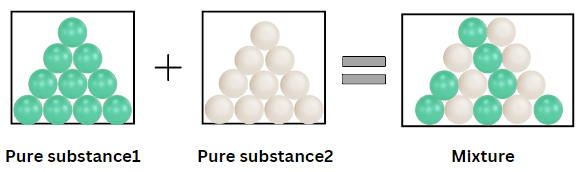
- Contains more than one type of particle
- Particles are physically mixed, not bonded
- Composition can vary
- Components retain their original properties
These features must be visible in accurate particle models of mixtures.
Particle-Level Description of Mixtures
In a correct particle model of a mixture:
- Different particles appear as different colors, sizes, or shapes
- Particles are intermingled but not bonded
- Each particle type remains unchanged
Models should never show particles combining into new particles, as that would represent a chemical reaction, not a mixture.
Types of Mixtures in Particle Models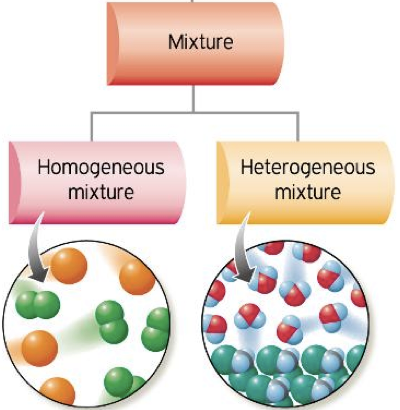
Homogeneous Mixtures
- Composition is uniform throughout
- Particles are evenly distributed
- Also called solutions
Heterogeneous Mixtures
- Composition is not uniform
- Different regions contain different particle ratios
- Components may be visibly separated
Mixtures vs. Pure Substances (Particle Models)
| Feature | Mixture | Pure Substance |
|---|---|---|
| Types of particles | Two or more | One |
| Bonding | No bonding between components | Chemically bonded (if compound) |
| Composition | Variable | Fixed |
| Properties of components | Retained | New properties (if compound) |
Evaluating Models of Mixtures
To evaluate whether a model correctly represents a mixture, check that:
- More than one particle type is present
- Particles are not connected by bonds
- Each particle type remains unchanged
- No new particles are formed
If bonding occurs or all particles are identical, the model does not represent a mixture.
Example
A particle diagram shows red single spheres and blue bonded pairs mixed together, with no bonds between red and blue particles. Identify whether the sample is a mixture and justify your answer.
▶️ Answer / Explanation
The sample represents a mixture.
Two different types of particles are present, and they are not bonded to each other. Each component keeps its original particle structure.
Example
A model shows particles of two substances evenly distributed throughout a container. One substance consists of single atoms; the other consists of bonded molecules. Evaluate whether this represents a homogeneous mixture and explain your reasoning.
▶️ Answer / Explanation
The model represents a homogeneous mixture.
Although there are two different particle types, they are evenly distributed and not chemically bonded. Each component retains its own properties, which is characteristic of a homogeneous mixture.
2.1.B.2 — Interpreting Experiments That Separate Mixtures
Because a mixture contains different types of particles that are not chemically bonded, its components can be separated by physical processes. Interpreting separation experiments involves identifying the method used and explaining which property difference allowed the separation.
During separation, no new substances form; each component retains its original properties.
Key Principle of Separation Methods
A separation method is chosen based on differences in:
- Particle size
- Solubility
- Boiling point
- Attraction to surfaces
Each separation technique works because the components of a mixture differ in a physical property, such as particle size, solubility, boiling point, or attraction to a surface.
Common Separation Methods and What They Show
| Separation Method | Property Used | What the Results Show |
|---|---|---|
| Filtration | Particle size | Insoluble solid remains on filter; liquid passes through |
| Evaporation | Boiling point / volatility | Solute remains after solvent evaporates |
| Distillation | Different boiling points | Lower-boiling liquid is collected first |
| Chromatography | Attraction to stationary/mobile phases | Components separate into distinct bands or spots |
Filtration
Filtration separates an insoluble solid from a liquid.
- Large solid particles are trapped by filter paper
- Liquid and dissolved substances pass through
This method works because the solid particles are larger than the pores in the filter.
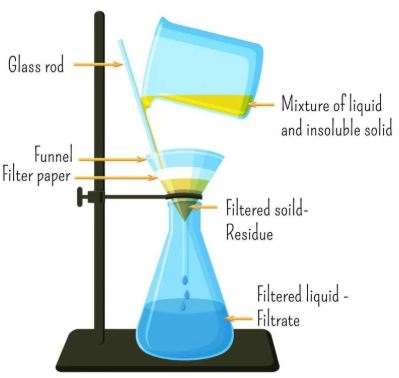
Interpreting Filtration Results
- If a solid remains on the filter paper, it was insoluble
- If liquid passes through, its particles are small enough to pass the pores
Conclusion: The mixture contained a solid–liquid combination that was separated by particle size.
Evaporation
Evaporation separates a dissolved solid from a solution.
- The liquid solvent evaporates
- The solid solute remains behind
This method relies on the solvent having a lower boiling point than the solute.
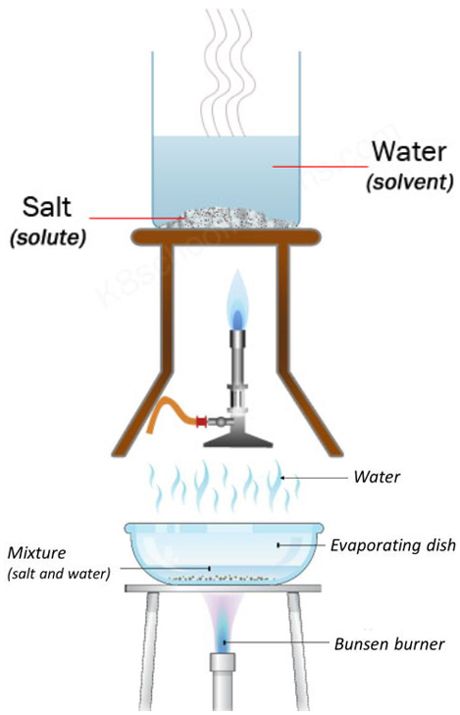
Interpreting Evaporation Results
- Loss of liquid indicates the solvent evaporated
- Solid residue indicates a dissolved solute
Conclusion: The mixture was a solution separated because the solvent had a lower boiling point.
Distillation
Distillation separates components based on different boiling points.
- The substance with the lower boiling point vaporizes first
- The vapor is condensed and collected
Distillation can separate:
- Two liquids
- A liquid from a dissolved solid
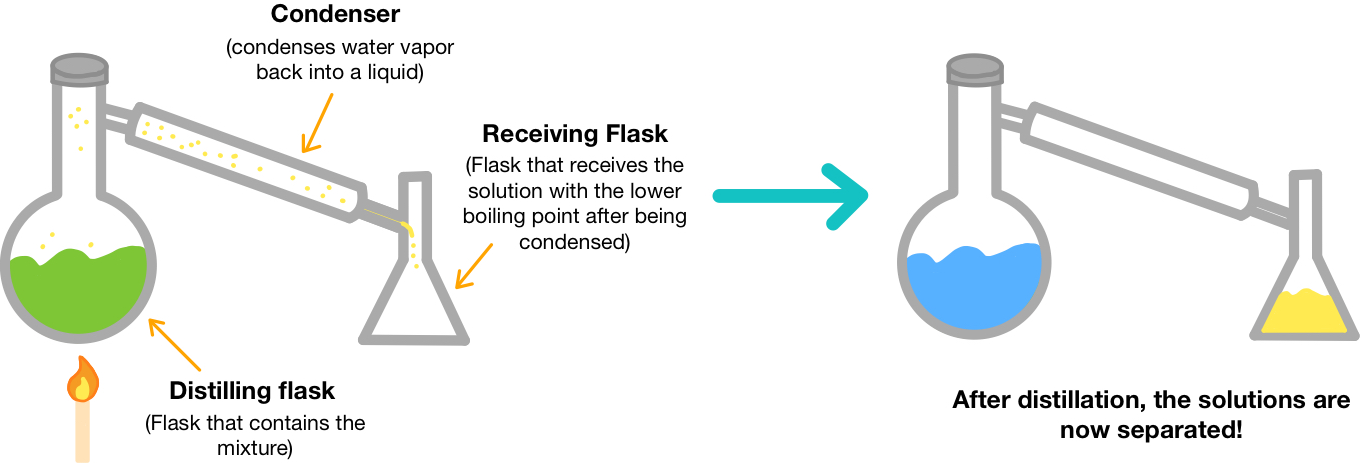
Interpreting Distillation Results
- Collected liquid has a lower boiling point
- Remaining liquid has a higher boiling point
Conclusion: The mixture contained liquids with different boiling points.
Chromatography
Chromatography separates dissolved substances based on their attraction to two phases:
- Stationary phase (paper or solid surface)
- Mobile phase (liquid solvent)
Components that are more attracted to the solvent travel farther, while those more attracted to the surface move less.
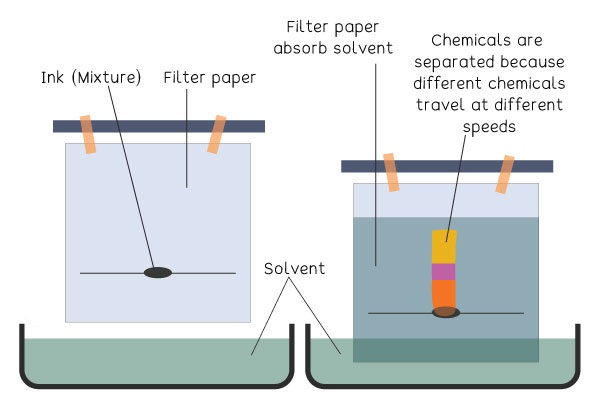
Interpreting Chromatography Results
- More spots than expected → mixture, not a pure substance
- Different travel distances show different attractions
Conclusion: Components separate because they differ in how strongly they are attracted to the stationary phase versus the mobile phase.
Key Interpretation Rule
If a mixture is separated by a physical method and the components remain unchanged, the process is physical, not chemical.
Choosing the Correct Separation Method
| Mixture Type | Best Method |
|---|---|
| Sand and water | Filtration |
| Salt dissolved in water | Evaporation or distillation |
| Two miscible liquids | Distillation |
| Dyes in ink | Chromatography |
If a process separates a mixture and no chemical bonds are broken or formed, the process is physical, not chemical.
Example
A sand–salt mixture is stirred into water and filtered. The sand remains on the filter paper, and salt is found in the filtrate. Interpret these results.
▶️ Answer / Explanation
Sand particles are insoluble and too large to pass through the filter, so they remain on the filter paper.
Salt dissolves in water and passes through the filter with the liquid. This shows the mixture was separated based on particle size and solubility.
Example
A black ink sample produces several colored spots when analyzed using paper chromatography. Interpret what this result reveals about the ink.
▶️ Answer / Explanation
The presence of multiple colored spots shows that the ink is a mixture, not a pure substance.
The different dyes separated because they have different attractions to the paper and solvent, causing them to move different distances.
