2.2A Intermolecular Forces- Pre AP Chemistry Study Notes - New Syllabus.
2.2A Intermolecular Forces- Pre AP Chemistry Study Notes
2.2A Intermolecular Forces- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
2.2.A.1 Create and/or evaluate models that illustrate how molecular properties influence the type(s) of intermolecular force(s) present in a substance.
2.2.A.2 Create and/or evaluate a claim about the type(s), strength(s), and origin(s) of intermolecular forces present in a substance.
Key Concepts:
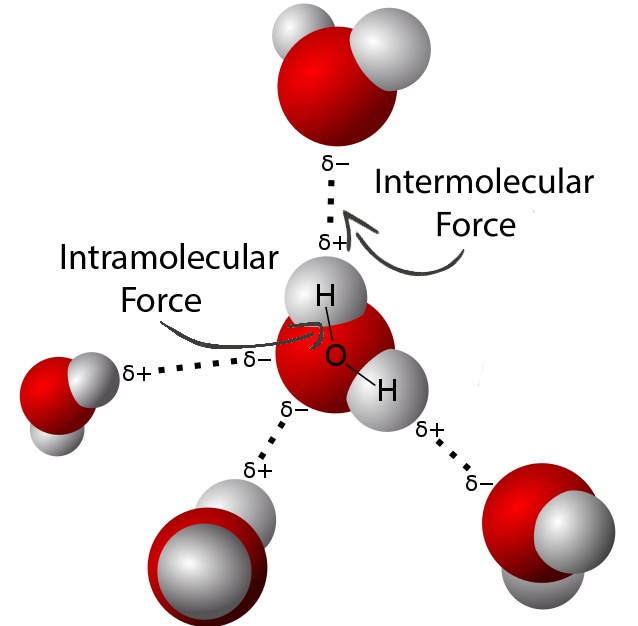
- 2.2.A Intermolecular forces occur between molecules and are the result of electrostatic interactions.
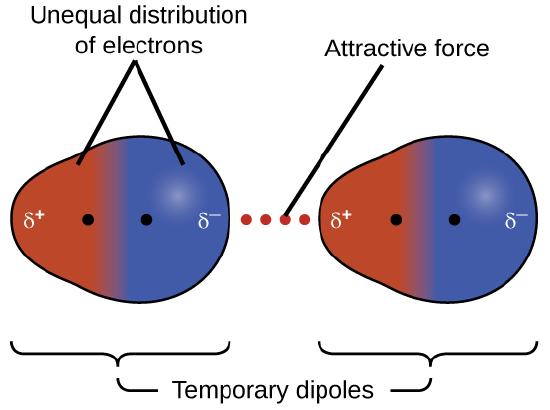
a. London dispersion forces are attractions among temporary dipoles created by the random movement of electrons; these attractions occur between all types of molecules. Molecules with more electrons tend to have stronger London dispersion forces.
b. Dipole–dipole forces are attractions among permanent dipoles on interacting molecules.
c. Hydrogen bonding forces exist when hydrogen atoms covalently bonded to highly electronegative atoms (N, O, or F) are attracted to the negative ends of dipoles formed by highly electronegative atoms (N, O, or F) in other molecules.
2.2.A.1 — Molecular Properties and Intermolecular Forces
Intermolecular forces (IMF) are attractive forces between particles that arise from electrostatic interactions. The type(s) of intermolecular forces present in a substance depend on specific molecular properties, such as polarity, shape, and the presence of certain atoms.
Particle models are used to illustrate and evaluate how these molecular properties determine which intermolecular forces act between particles.
Key Molecular Properties That Affect IMF
- Molecular polarity
- Presence of polar bonds (especially O–H, N–H, or F–H)
- Molecular size and number of electrons
- Molecular shape and symmetry
These properties determine how electrons are distributed and how molecules attract each other.
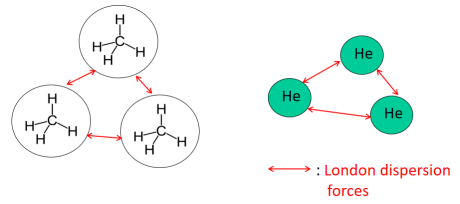
London Dispersion Forces (LDF)
London dispersion forces arise from temporary fluctuations in electron distribution, creating instantaneous dipoles.
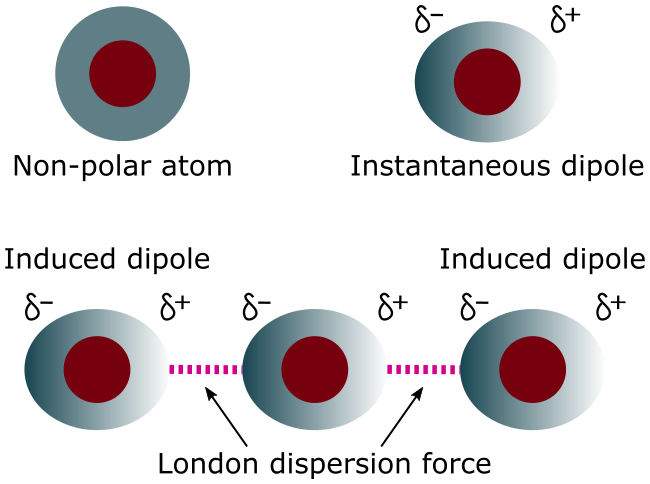
- Present in all substances
- Only IMF present in nonpolar molecules
- Strength increases with molecular size and number of electrons
In particle models, LDF are shown as weak, momentary attractions between nearby particles.
Dipole–Dipole Forces
Dipole–dipole forces occur between polar molecules, where partial positive and partial negative ends attract.
![]()
- Require molecules with a permanent dipole
- Stronger than London dispersion forces (for similar-sized molecules)
Particle models show alignment of opposite partial charges between neighboring molecules.
Hydrogen Bonding
Hydrogen bonding is a particularly strong type of dipole–dipole interaction. It occurs when hydrogen is bonded to a highly electronegative atom:
- Oxygen (O)
- Nitrogen (N)
- Fluorine (F)
![]()
Hydrogen bonding requires:
- A highly polar H–O, H–N, or H–F bond
- A nearby molecule with a lone pair on O, N, or F
Particle models show strong directional attractions between molecules.
Comparing Intermolecular Forces
| Type of IMF | Molecular Property Required | Relative Strength |
|---|---|---|
| London dispersion | All molecules | Weakest |
| Dipole–dipole | Polar molecules | Moderate |
| Hydrogen bonding | H–O, H–N, or H–F bonds | Strongest (IMF) |
Evaluating Particle Models of IMF
A correct model illustrating intermolecular forces must:
- Identify whether molecules are polar or nonpolar
- Show partial charges when polarity is present
- Represent stronger attractions with closer spacing or alignment
Models should not show covalent bonds breaking or forming between molecules.
Example
A particle model shows molecules with uneven charge distribution and opposite ends aligned. Identify the type of intermolecular force present and justify your answer.
▶️ Answer / Explanation
The intermolecular force is dipole–dipole attraction.
The molecules are polar, with partial positive and partial negative ends attracting each other.
Example
Two substances have similar molar masses, but one has a much higher boiling point. One substance contains O–H bonds, while the other does not. Evaluate how molecular properties explain this difference.
▶️ Answer / Explanation
The substance with O–H bonds can form hydrogen bonds, which are stronger intermolecular forces.
Stronger intermolecular forces require more energy to overcome, resulting in a higher boiling point compared to the substance lacking hydrogen bonding.
2.2.A.2 — Claims About the Type, Strength, and Origin of Intermolecular Forces
A scientific claim about intermolecular forces (IMF) explains which types of IMF are present in a substance, how strong they are relative to one another, and why they exist based on molecular properties.
All intermolecular forces originate from electrostatic interactions between charged particles (protons and electrons).
Origin of Intermolecular Forces
Intermolecular forces arise because electrons are not evenly distributed at all times. This creates regions of partial charge that attract one another.
- Temporary charge imbalances → London dispersion forces
- Permanent charge separation → dipole–dipole forces
- Highly polar H–O, H–N, or H–F bonds → hydrogen bonding
Although weaker than chemical bonds, IMF strongly influence physical properties.
Types of Intermolecular Forces
London Dispersion Forces (LDF)
- Present in all substances
- Caused by temporary fluctuations in electron distribution
- Only IMF present in nonpolar substances
LDF strength increases with molar mass, number of electrons, and surface area.
Dipole–Dipole Forces
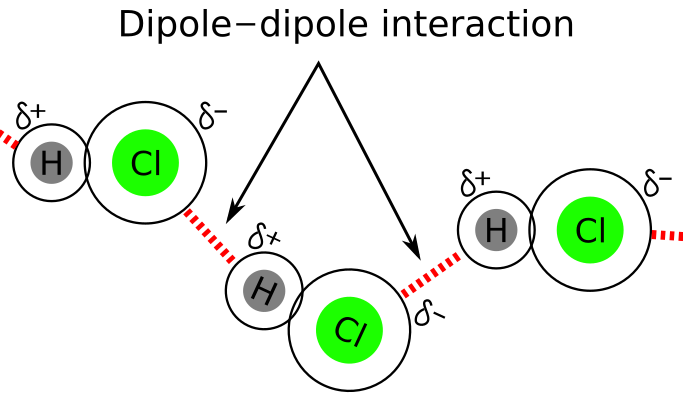
- Occur in polar molecules
- Caused by attraction between permanent partial charges
- Stronger than London dispersion forces for similar-sized molecules
Hydrogen Bonding
Hydrogen bonding is a particularly strong form of dipole–dipole attraction.
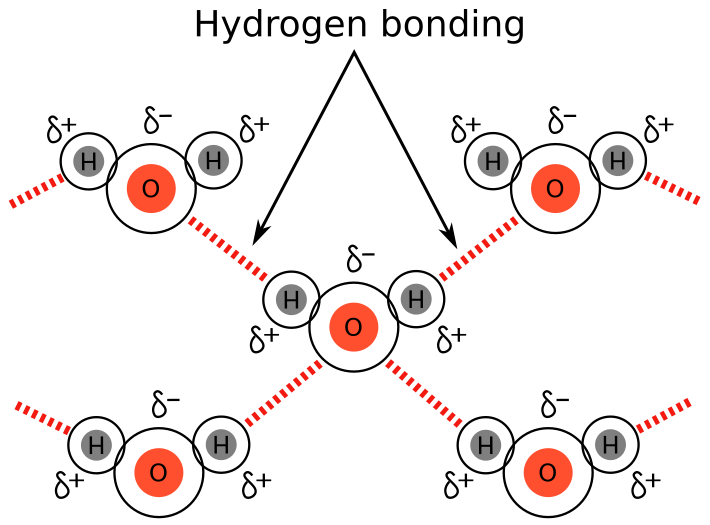
- Requires hydrogen bonded to O, N, or F
- Requires a nearby lone pair on O, N, or F
- Highly directional and relatively strong
Comparing Strengths of Intermolecular Forces
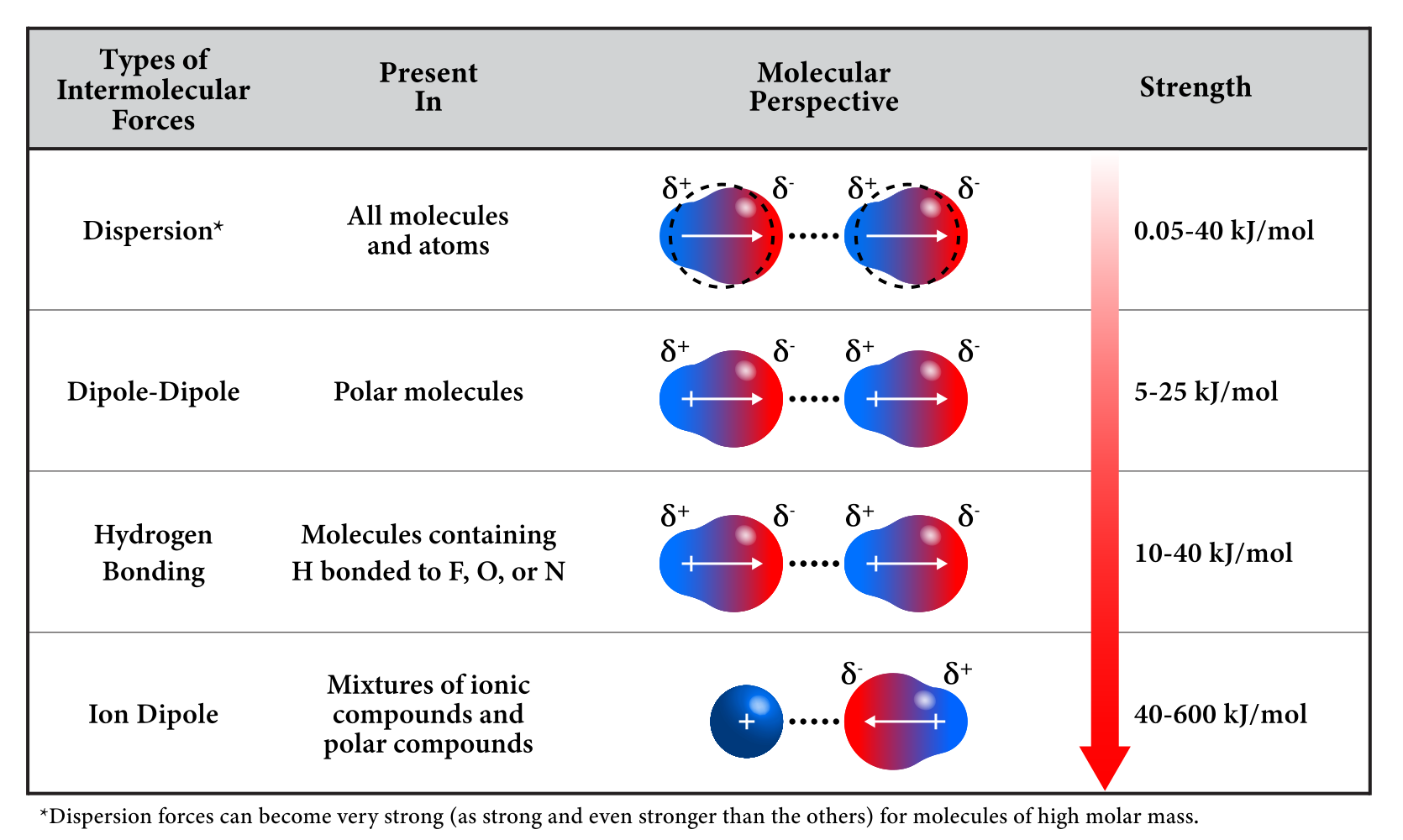
| Intermolecular Force | Origin | Relative Strength |
|---|---|---|
| London dispersion | Temporary electron motion | Weakest |
| Dipole–dipole | Permanent molecular polarity | Moderate |
| Hydrogen bonding | Highly polar H–O, H–N, or H–F bonds | Strongest (IMF) |
How to Construct a Correct Claim
A strong claim about intermolecular forces should:
- Identify all IMF present in the substance
- Compare their relative strengths
- Explain their electrostatic origin using molecular properties
Claims that ignore molecular polarity or electron distribution are incomplete.
Example
Create a claim describing the intermolecular forces present in carbon dioxide, CO₂, and explain their origin.
▶️ Answer / Explanation
Carbon dioxide experiences only London dispersion forces.
Although CO₂ contains polar bonds, the molecule is linear and nonpolar overall, so only temporary electron fluctuations create intermolecular attractions.
Example
Two substances have similar molar masses: methanol (CH₃OH) and hydrogen sulfide (H₂S). Create and evaluate a claim explaining which has stronger intermolecular forces and why.
▶️ Answer / Explanation
Methanol has stronger intermolecular forces than hydrogen sulfide.
Methanol experiences London dispersion forces, dipole–dipole forces, and hydrogen bonding due to its O–H bond.
Hydrogen sulfide lacks hydrogen bonding and relies mainly on weaker dipole–dipole and London dispersion forces. The stronger electrostatic attractions in methanol explain its higher boiling point.
