2.2C Periodic Trends- Pre AP Chemistry Study Notes - New Syllabus.
2.2C Periodic Trends- Pre AP Chemistry Study Notes
2.2C Periodic Trends- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
2.2.C.1 Describe trends in properties of elements based on their position in the periodic table and the shell model of the atom.
Key Concepts:
- 2.2.C The periodic table is an organizational tool for elements based on their properties.
a. Patterns of behavior of elements are based on the number of electrons in the outermost shell (valence electrons).
b. Important periodic trends include electronegativity and atomic radius.
2.2.C.1 — Periodic Trends, the Shell Model, and Element Properties
The periodic table organizes elements based on recurring patterns in their physical and chemical properties. These patterns can be explained using the shell model of the atom, which describes how electrons are arranged around the nucleus.
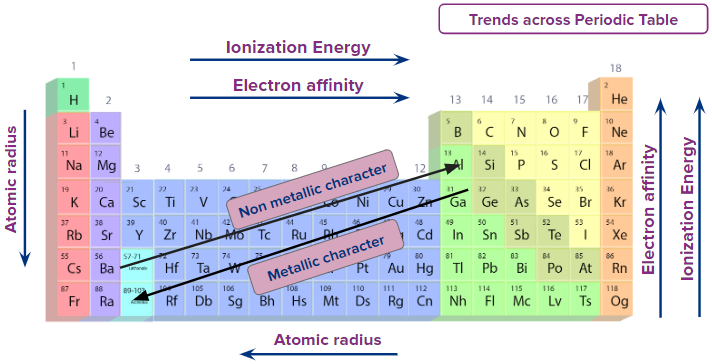
An element’s position on the periodic table reflects its number of valence electrons, which strongly influences reactivity and trends such as atomic radius, electronegativity, ionization energy, Electron Affinity , and metallic character.
The Shell Model of the Atom
The shell model represents electrons arranged in energy levels (shells) surrounding the nucleus.
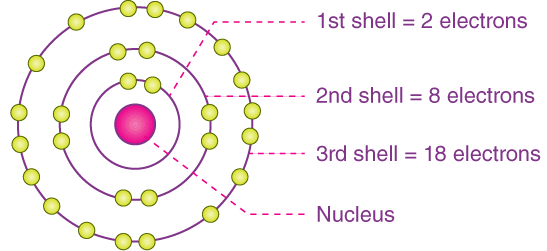
- Electrons closer to the nucleus experience stronger electrostatic attraction
- Outer shells are farther away and more shielded by inner electrons
- The outermost shell contains valence electrons
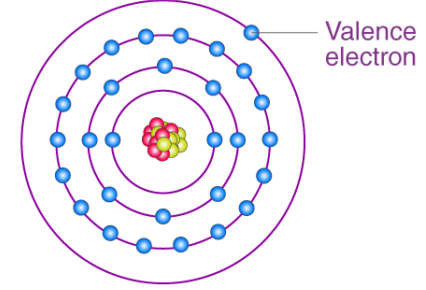
Valence electrons control bonding behavior, reactivity, and periodic trends.
Valence Electrons and Periodic Patterns
Elements in the same column (group) of the periodic table have the same number of valence electrons.
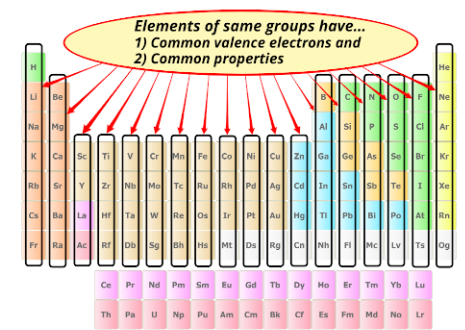
- Same valence electrons → similar chemical properties
- Predictable reactivity patterns within each group
This explains why alkali metals, halogens, and noble gases behave similarly within their groups.
Atomic Radius
Atomic radius is a measure of the size of an atom. It depends on the number of occupied shells and the attraction between the nucleus and valence electrons.
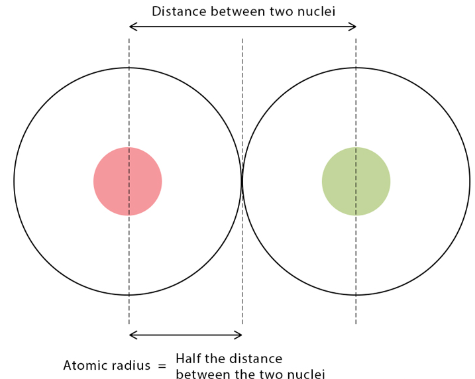
Across a Period (Left → Right)
![]()
- Same number of shells
- Increasing nuclear charge
- Electrons pulled closer to nucleus
- Atomic radius decreases
Down a Group (Top → Bottom)
![]()
- Additional electron shells added
- Greater distance from nucleus
- Atomic radius increases
Electronegativity
Electronegativity is the ability of an atom to attract electrons in a chemical bond.
![]()
Across a Period
- Nuclear charge increases
- Atomic radius decreases
- Electronegativity increases
Down a Group
- Valence electrons farther from nucleus
- Electronegativity decreases
Ionization Energy
Ionization energy is the energy required to remove a valence electron from a gaseous atom.
![]()
Across a Period
- Nuclear attraction increases
- Electrons held more tightly
- Ionization energy increases
Down a Group
- Valence electrons farther from nucleus
- More shielding
- Ionization energy decreases
Electron Affinity
Electron affinity is the energy change that occurs when a neutral gaseous atom gains an electron.
A more negative electron affinity means that more energy is released, indicating a stronger attraction for electrons.
![]()
Across a Period
- Nuclear charge increases
- Atomic radius decreases
- Atoms more strongly attract added electrons
- Electron affinity becomes more negative
Down a Group
- Increased distance from nucleus
- Greater shielding by inner shells
- Electron affinity becomes less negative
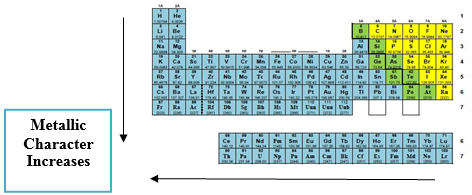
Metallic Character
Metallic character describes how readily an element loses electrons
 .
.
- Increases down a group
- Decreases across a period
Elements with low ionization energy exhibit strong metallic behavior.
Summary of Key Periodic Trends
| Trend | Across a Period | Down a Group |
|---|---|---|
| Valence electrons | Increase | Stay the same |
| Atomic radius | Decreases | Increases |
| Electronegativity | Increases | Decreases |
| Ionization energy | Increases | Decreases |
| Metallic character | Decreases | Increases |
Evaluating Claims Using the Shell Model
Accurate claims about periodic trends must:
- Reference valence electrons
- Explain nuclear attraction and shielding
- Use shell distance to justify trends
Claims based solely on position without particle-level reasoning are incomplete.
Example
Explain why atomic radius decreases from sodium to chlorine across Period 3.
▶️ Answer / Explanation
All Period 3 elements have the same number of electron shells.
As nuclear charge increases across the period, valence electrons experience stronger attraction, pulling them closer to the nucleus and decreasing atomic radius.
Example
Compare potassium and calcium in terms of atomic radius and ionization energy. Use the shell model to justify both trends.
▶️ Answer / Explanation
Potassium has a larger atomic radius than calcium because both elements are in Period 4, but calcium has a higher nuclear charge that pulls electrons closer.
Calcium has a higher ionization energy because its valence electrons experience stronger attraction to the nucleus, making them harder to remove.
